SCH4U - U1b Bonding and States of Matter Practice Test
1/97
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
98 Terms
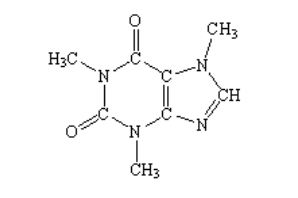
1. How many lone pairs are found on a molecule of aspartic acid (consider all atoms).
a. 0
b. 4
c. 6
d. 8
e. 12
d. 8
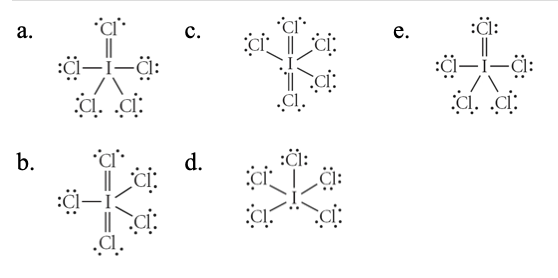
2. Choose the correct Lewis structure for ICl5.
a.
b.
c.
d.
e.
d.

3. Which of the following elements can form compounds with an expanded octet?
a. Se
b. C
c. Li
d. F
a. Se
in period 3 meaning it has access to the d orbital
4. Resonance structures are needed to describe the bonding in which of the following?
a. CO2
b. H2O
c. ClF3
d. HNO3
e. CH4
d. HNO3
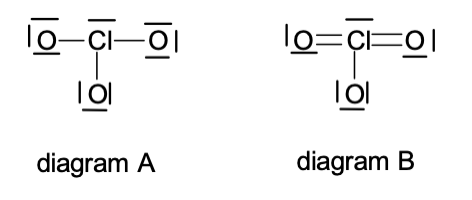
5. A Lewis diagram can be drawn for ClO3- as in diagram A. However, diagram B is the preferred structure. Why?
a. | Chlorine needs 12 electrons to satisfy its octet. |
b. | Each oxygen can only have 8 electrons. |
c. | The formal charge is better. |
d. | The chlorine atom behaves as a radical, pulling more electrons from oxygen. |
c. | The formal charge is better. |
6. According to VSEPR theory, molecules adjust their shapes to keep which of the following as far apart as possible?
a. | pairs of valence e-s | d. | mobile electrons |
b. | inner shell electrons | e. | lone pairs of electrons |
c. | bonding pairs of e-s | ||
a. | pairs of valence e-s |
According to VSEPR theory (Valence Shell Electron Pair Repulsion theory), the shape of a molecule is determined by the repulsion between pairs of valence electrons around the central atom. These electron pairs (both bonding and lone pairs) repel each other and arrange themselves to be as far apart as possible to minimize repulsion.
7. A molecule consisting of a central atom surrounded by two bonding pairs and two non-bonding (lone) pairs of electrons will be
a. | linear | d. | trigonal pyramidal |
b. | tetrahedral | e. | trigonal planar |
c. | bent | ||
c. | bent |

8. What would be the shape of a molecule containing a central atom attached to two other atoms with two lone pairs of e-s?
a. | bent | d. | see-saw |
b. | trigonal pyramidal | e. | square planar |
c. | trigonal planar | ||
a. | bent |

9. What would be the shape of a molecule containing a central atom attached to four other atoms with one lone pair of electrons?
a. | trigonal planar | d. | square pyramidal |
b. | see-saw | e. | octahedral |
c. | square planar | ||
b. | see-saw |

10. What would be the shape of a molecule containing a central atom attached to four other atoms with one lone pair of electrons?
a. | trigonal planar | d. | square pyramidal |
b. | see-saw | e. | octahedral |
c. | square planar | ||
b. | see-saw |

11. What is the shape of the nitrate ion, NO31-?
a. | linear | d. | square bipyramidal |
b. | trigonal planar | e. | trigonal pyramidal |
c. | tetrahedral | ||
b. | trigonal planar |
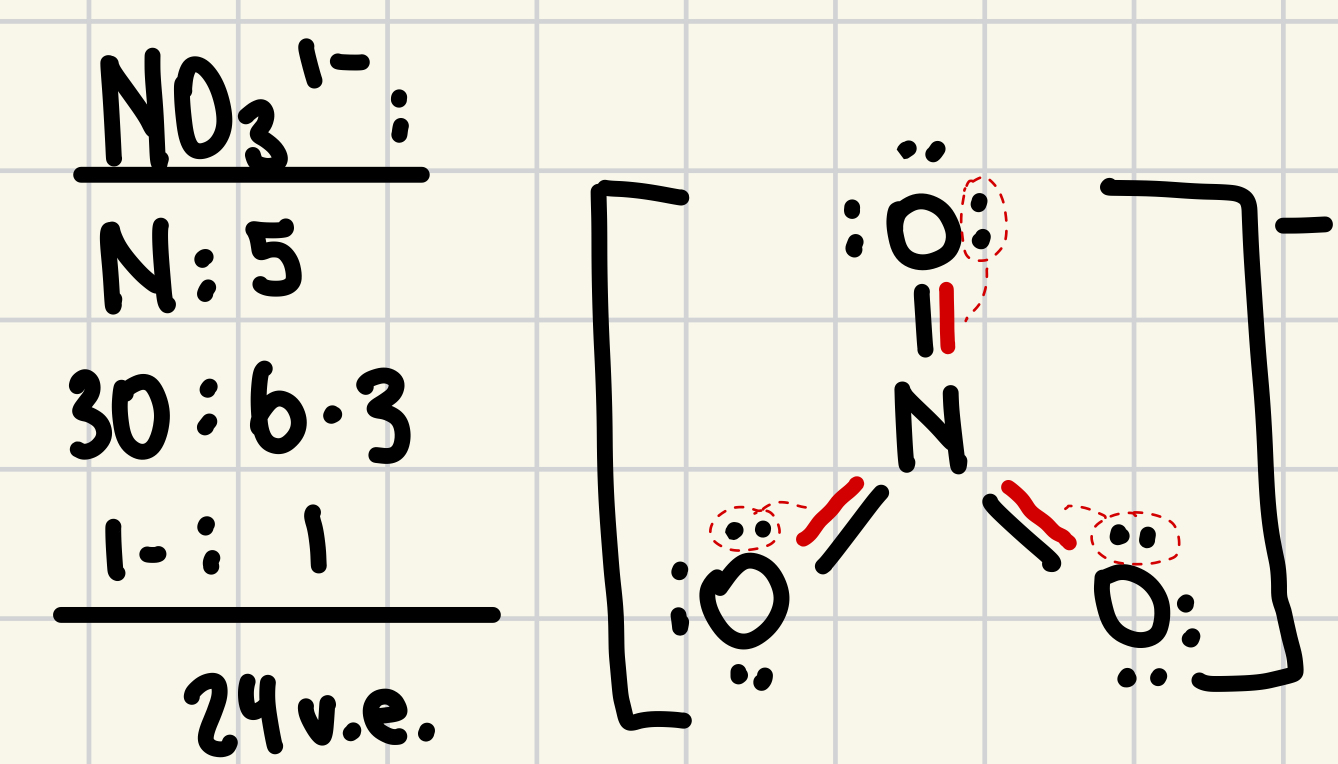
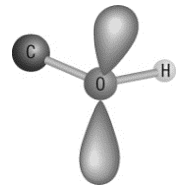
12. What molecular shape is shown?
a. | trigonal planar | c. | bent |
b. | tetrahedral | d. | trigonal pyramidal |
c. | bent |
13. Why does a central atom surrounded by 4 atoms have a tetrahedral shape instead of a square planar shape?
a. | Orbitals are never at right angles to each other. |
b. | The lone pairs around the central atom push the 4 atoms into this configuration. |
c. | The angle in a tetrahedron is larger than in a square planar arrangement. |
d. | The 4 atoms want to be as close together as possible. |
e. | None of the above. |
c. | The angle in a tetrahedron is larger than in a square planar arrangement. |
14. Six pairs of electrons surrounding a central atom will be arranged
a. | tetrahedrally | d. | octahedrally |
b. | spherically | e. | hexagonally |
c. | trigonal bipyramidally | ||
d. | octahedrally |
15. Which of the following molecular geometries exhibits the smallest bond angles?
a. | planar triangular | d. | trihedral |
b. | tetrahedral | e. | linear |
c. | octahedral | ||
c. | octahedral |
16. Give the approximate bond angle for a molecule with a trigonal planar shape.
a. 109.5
b. 180
c. 120
d. 105
c. 120
17. Give the approximate bond angle for a molecule with a tetrahedral shape.
a. 109.5
b. 180
c. 120
d. 105
a. 109.5
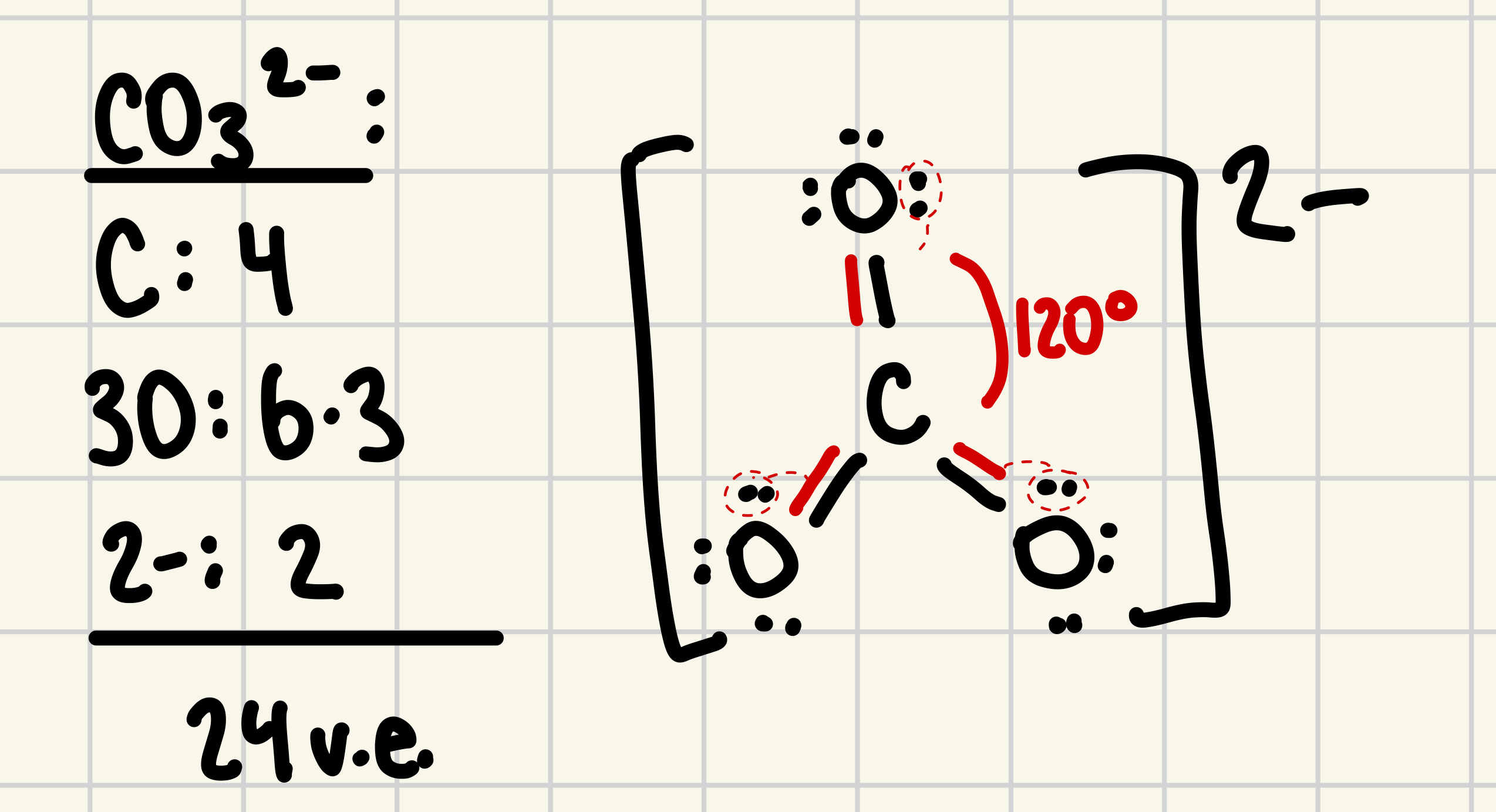
18. The bond angle in CO32- is expected to be approximately
a. 90
b. 109.5
c. 120
d. 145
e. 180
c. 120
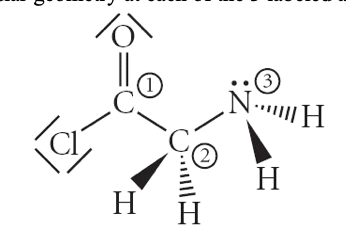
19. Consider the molecule in the image. Determine the molecular geometry at each of the 3 labeled atoms.
a. | 1=trigonal planar, 2=tetrahedral, 3=trigonal pyramidal |
b. | 1=tetrahedral, 2=tetrahedral, 3=tetrahedral |
c. | 1=trigonal planar, 2=tetrahedral, 3=tetrahedral |
d. | 1=tetrahedral, 2=tetrahedral, 3=trigonal planar |
a. | 1=trigonal planar, 2=tetrahedral, 3=trigonal pyramidal |
20. What is the expected hybridization of the central atom in a molecule of SiI4? This molecule is tetrahedral.
a. sp3d2
b. sp3d
c. sp2
d. sp3
e. sp
d. sp3

Predict the hybridization that occurs for the carbon atom in a methane, CH4, molecule?
a. sp3d2
b. sp2
c. sp3d
d. sp3
e. sp2
d. sp3

Predict the hybridization that occurs for the nitrogen atom in an ammonia, NH3, molecule?
a. sp3d2
b. sp2
c. sp3d
d. sp3
e. sp3d2
b. sp2
it makes sp3 so it can make 4 bonds BUT since only 3 atoms are bonded yoou only need three bonds and the other one remains unhybridized with a single electron
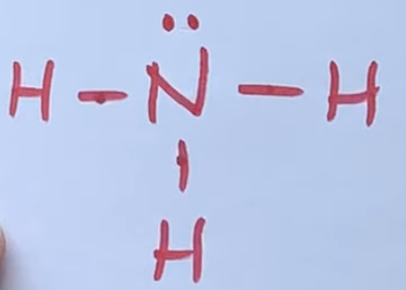
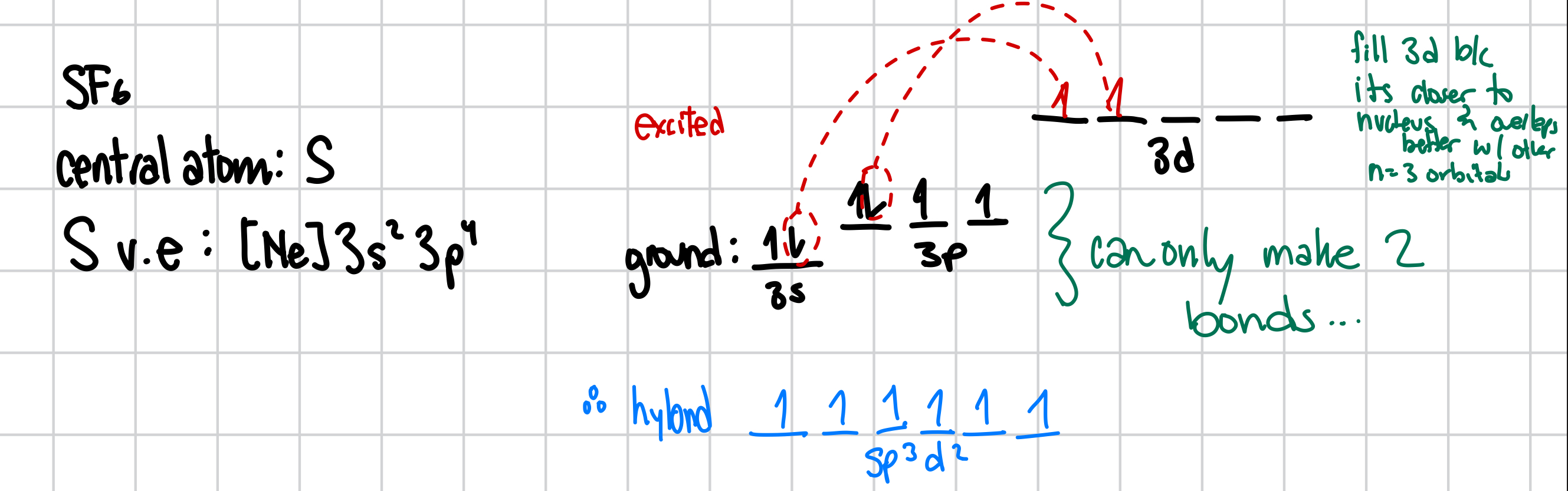
What is the hybridization of the central atom in SF6
a. sp
b. sp2
c. sp3
d. sp3d
e. sp3d2
e. sp3d2
24. A molecule containing a central atom with sp3 hybridization has a(n) __________ electron geometry.
a. | linear | d. | tetrahedral |
b. | trigonal bipyramidal | e. | bent |
c. | octahedral | ||
d. | tetrahedral |
4 places to bond…
25. Which of the following examples of bond hybridization best accounts for the shape of the molecule below?
a. sp
b. sp2
c. sp3
d. sp3d
e. sp3d2
a. sp
26. What is the correct shape of a molecule that demonstrates sp2 hybridization?
a. | bent | c. | inear |
b. | trigonal planar | d. | tetrahedral |
b. | trigonal planar |
27. What is the total number of π-bonds in (CO32-)?
a. 0
b. 1
c. 2
d. 3
e. 4
b. 1

28. Which of the following correctly describes a sigma bond?
a. | a bond formed when two orbitals overlap end to end |
b. | a bond formed when the nodes of two orbitals overlap |
c. | a triple bond |
d. | a double bond |
a. | a bond formed when two orbitals overlap end to end |
29. Which of the following most correctly describes a pi bond?
a. | a bond formed when the sides of the lobes of two orbitals overlap |
b. | a triple bond |
c. | a bond formed when the nodes of two orbitals overlap |
d. | a double bond |
a. | a bond formed when the sides of the lobes of two orbitals overlap |
30. Draw the Lewis structure for the molecule C3H4. How many sigma and pi bonds does it contain?
a. | 7 sigma, 1 pi | d. | 10 sigma, 0 pi |
b. | 8 sigma, 0 pi | e. | 8 sigma, 2 pi |
c. | 6 sigma, 2 pi | ||
c. | 6 sigma, 2 pi |
31. An electron in a(n) __________ subshell experiences the greatest effective nuclear charge in a many-electron atom.
a. 4f
b. 4p
c. 4d
d. 4s
e. 5s
d. 4s
32. Which of the following is the most electronegative?
a. cesium
b. nitrogen
c. chlorine
d. fluorine
d. fluorine
Place the following elements in order of increasing electronegativity.
19K 55Cs 15P
a.
P < K < Cs
d.
Cs < K < P
b.
K < P < Cs
e.
< Cs < K
c.
Cs < P < K
d. | Cs < K < P |
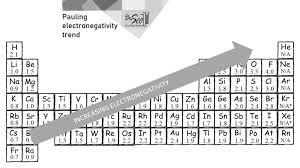
34. Use the accompanying table to predict which of the following substances will have the most polar covalent bond and the least polar covalent bond.
Pauling Electronegativities
H | F | Cl | Br | I |
2.1 | 4.0 | 3.0 | 2.8 | 2.5 |
a. | most: HI least: HF |
b. | most: HF least: HI |
c. | most: HCl least: HBr |
d. | most: HBr least: HCl |
b. | most: HF least: HI |
look at difference in EN
35. When is ionic bonding likely to occur between two atoms?
a. | When both atoms have low ionization energy and low electron affinity. |
b. | When both atoms have high ionization energy and high electron affinity. |
c. | When both atoms have high ionization energy and low electron affinity. |
d. | When one atom has high ionization energy and high electron affinity, while the other has low ionization energy and low electron affinity. |
e. | When one atom has a high ionization energy and low electron affinity, while the other has a low ionization energy and a high electron affinity. |
d. | When one atom has high ionization energy and high electron affinity, while the other has low ionization energy and low electron affinity. |
36. In general, why do nonmetals form covalent bonds with one another?
a. | They have similar atomic radii. |
b. | They have similar electronegativities. |
c. | Sharing of valence electrons is the most effective way to achieve stability. |
d. | They have similar atomic masses. |
e. | Answers b) & c) are correct. |
e. | Answers b) & c) are correct. |
b. | They have similar electronegativities. |
c. | Sharing of valence electrons is the most effective way to achieve stability. |
37. Which of the following statements is TRUE?
a. | A covalent bond is formed through the transfer of electrons from one atom to another. |
b. | A pair of electrons involved in a covalent bond are sometimes referred to as "lone pairs." |
c. | It is not possible for two atoms to share more than two electrons. |
d. | Single bonds are shorter than double bonds. |
e. | A covalent bond has a lower potential energy than the two separate atoms. |
e. | A covalent bond has a lower potential energy than the two separate atoms. |
38. Which of the following has ionic bonding?
a. | H2O | c. | NO2 | e. | CaCO3 |
b. | C6H12O6 | d. | Al |
e. | CaCO3 |
39. Which of the following factors are involved in formation of a covalent bond in hydrogen molecules?
a. | attraction of protons for electrons |
b. | force of repulsion between nuclei |
c. | distance between hydrogen atoms |
d. | all of the above |
d. | all of the above |
40. Which of these bonds is the most polar?
a. H-Cl
b. C-Br
c. H-F
d. H-I
c. H-F
41. Which of the molecules, CO2, NH3 and BCl3 will be polar?
a. | CO2, NH3 and BCl3 |
b. | CO2 and BCl3 |
c. | NH3 |
d. | CO2 and NH3 |
e. | BCl3 |
c. | NH3 |
CO2 is a linear molecule with a symmetrical structure. Although it has polar bonds, the dipoles cancel out, making the molecule nonpolar.
NH3 (ammonia) has a trigonal pyramidal shape with a lone pair on nitrogen. This causes an uneven distribution of electrons, making it a polar molecule.
BCl3 is trigonal planar and symmetrical. The polar bonds cancel out, resulting in a nonpolar molecule.
42. Which of the following is a non-polar molecule?
a. CCl4
b. CH3Cl
c. HCl
d. CH2Cl2
a. CCl4
symettrical
43. Polar covalent bonds form between atoms with
a. | equally high electronegativities |
b. | high but unequal electronegativities |
c. | equally low electronegativities |
d. | equally low ionization energies |
e. | low but unequal ionization energies |
b. | high but unequal electronegativities |
44. Rank the following in order of increasing bond polarity:
i. H-F ii. H-Br iii. F-F iv. Na-Cl
a. | i. < ii. < iii. < iv | c. | ii. < i. < iii. < iv. |
b. | iii. < i. < ii. < iv. | d. | iii. < ii. < i. < iv. |
d. | iii. < ii. < i. < iv. |
F-F < H-Br < H-F < Na-Cl
45. Why do ionic formula units usually have higher melting and boiling points than covalent compounds?
a. | The intermolecular forces in ionic formula units are weaker than those in covalent compounds |
b. | Covalent molecules have higher electron affinities than ionic molecules. |
c. | Ionic bonds are stronger than covalent bonds |
d. | The intermolecular forces in covalent compounds are weaker than those in ionic formula units |
e. | Covalent bonds are stronger than ionic bonds |
c. | Ionic bonds are stronger than covalent bonds |
46. Intermolecular forces are
a. | forces within covalent molecules that hold them together. |
b. | electrostatic forces between ions. |
c. | bonds between H and O atoms in water molecules. |
d. | attractive forces between separate covalent molecules. |
e. | covalent bonds within a network solid. |
d. | attractive forces between separate covalent molecules. |
47. Which isn’t an example of a van der Waals force?
a. i hydrogen bonding b. covalent bond c. London dispersion force d. dipole–dipole forces |
d. dipole–dipole forces
48. Which of the following are all intermolecular forces?
a. | Dipole-dipole, ion-dipole, and London dispersion |
b. | Covalent, metallic, and London dispersion forces |
c. | Dispersion forces, hydrogen bonding, and covalent |
d. | Dipole-dipole, hydrogen bonding, and dispersion forces |
e. | Metallic, dipole-dipole and ionic |
d. | Dipole-dipole, hydrogen bonding, and dispersion forces |
49. Which forces exist between ammonia, NH3, particles?
I. London forces
II. metallic bonding
III. hydrogen bonding
IV.dipole / dipole
a. | I only | d. | I, III and IV only |
b. | I and IV only | e. | I, II and III only |
c. | I and II only | ||
d. | I, III and IV only |
Which forces exist between methane, CH4 particles
I. London forces
II. metallic bonding
III. hydrogen bonding
IV. dipole / dipole
a. | I only | d. | I, III and IV only | |
b. | I and IV only | e. | I, II and III only | |
c. | I and II only | |||
a. | I only |
I. London forces
51. What is the difference between dipole-dipole forces and hydrogen bonds?
a. | Hydrogen bonds only form between molecules with O-H, N-H or F-H groups |
b. | dipole-dipole forces only exist between non-polar molecules |
c. | dipole-dipole forces occur between polar molecules |
d. | dipole-dipole forces result from the interaction of partial charges on different molecules |
e. | Answers a & d are correct |
e. | Answers a & d are correct |
a.Hydrogen bonds only form between molecules with O-H, N-H or F-H groups
d. dipole-dipole forces result from the interaction of partial charges on different molecules
52. What is the strongest type of intermolecular force present in NH2CH3?
a. | dispersion | d. | ion-dipole |
b. | dipole-dipole | e. | None of these. |
c. | hydrogen bonding | ||
c. | hydrogen bonding |
53. The strength of dispersion forces between like-molecules depends on __________ and __________.
a. | polarity, size |
b. | molecular mass, volatility |
c. | molecular mass, polarity |
d. | size, shape |
e. | metallic character, size |
d. | size, shape |
54. What type of intermolecular force causes the dissolution of NaCl in water?
a. | hydrogen bonding | d. | dispersion forces | |
b. | dipole-dipole | e. | None of these. | |
c. | ion-dipole force | |||
c. | ion-dipole force |
55. Choose the molecule or compound that exhibits dipole-dipole forces as its strongest intermolecular force.
a. H2 | b.SO2 | c.NH3 | d.CF4 | e.BCl3 |
b.SO2
SO₂ (Sulfur Dioxide) is a polar molecule because of its bent shape (due to lone pairs on the sulfur) and the difference in electronegativity between sulfur and oxygen. This polarity results in dipole-dipole interactions as its strongest intermolecular force.
56. The strongest type of intermolecular force in a sample of CH3OH is _______________.
a. | covalent | d. | ionic |
b. | dipole | e. | hydrogen bonding |
c. | London | ||
e. | hydrogen bonding |
57. Hydrogen bonding is present in all of the following molecular solids EXCEPT ____.
a.H2SO4 | b.NH3 | c.PH3 | d.HF | e.H2O2 |
c.PH3
H just wants to have FON
58. The strong intermolecular forces between molecules containing F−H, O−H, and N−H bonds result from
a. | covalent bonds | d. | ionic bonds | |
b. | hydrogen bonding | e. | London dispersion | |
c. | dipole-dipole force | |||
b. | hydrogen bonding |
59. Place the following substances in order of increasing boiling point. Ne Cl2 O2
a. | Ne < Cl2 < O2 | d. | Cl2 < Ne < O2 |
b. | Cl2 < O2 < Ne | e. | Ne < O2 < Cl2 |
c. | O2 < Cl2 < Ne | ||
e. | Ne < O2 < Cl2 |
comparing molar mass:
Ne (Neon) → Molar mass = 20.18 g/mol
O₂ (Oxygen) → Molar mass = 32.00 g/mol
Cl₂ (Chlorine) → Molar mass = 70.90 g/mol
when looking at the pt take the atomic weight and x2 for HOFBrINCl
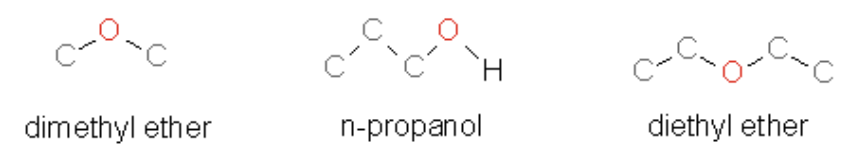
60. List the following molecules in order of increasing boiling point
a. | n-propanol < dimethyl ether < diethyl ether |
b. | diethyl ether < dimethyl ether < n-propanol |
c. | dimethyl ether < diethyl ether < n-propanol |
d. | dimethyl ether < n-propanol < diethyl ether |
c. | dimethyl ether < diethyl ether < n-propanol |
n propanol - note H bond
diethyl - more bonds compared to
dimethyl
61. Which of the following chemicals has the lowest boiling point?
a.F2 | b.NI3 | c.CH3I | d.H2O | e.H2O2 |
a.F2
F₂ (Fluorine) → London dispersion forces (LDFs)
Nonpolar molecule with weak LDFs.
62. Which of the following Group 14 hydrogen compounds has the highest boiling point?
a.CH4 | b.SiH4 | c.GeH4 | d.SnH4 |
d.SnH4
more electrons done the pt… more ldfs therefore higher boiling point
63. Identify the characteristics of a liquid.
a. | Indefinite shape and volume. |
b. | Indefinite shape, but definite volume. |
c. | Definite shape and volume. |
d. | none of the above |
e. | all of the above |
b. | Indefinite shape, but definite volume. |
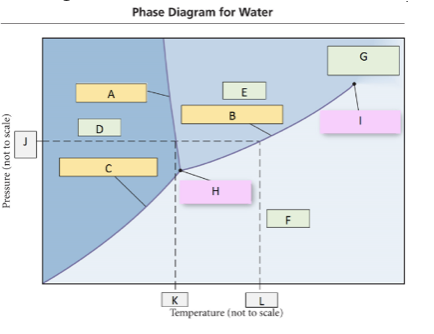
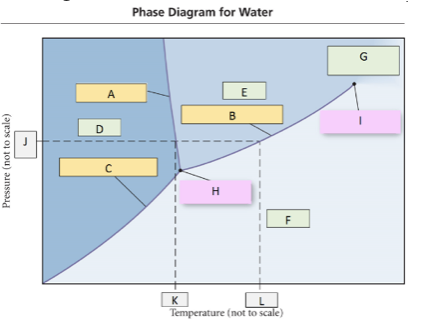
64. Use the phase diagram for water, what label should go in box H?
a. | solid | c. | gas | e. | critical point |
b. | liquid | d. | triple point |
d. | triple point |
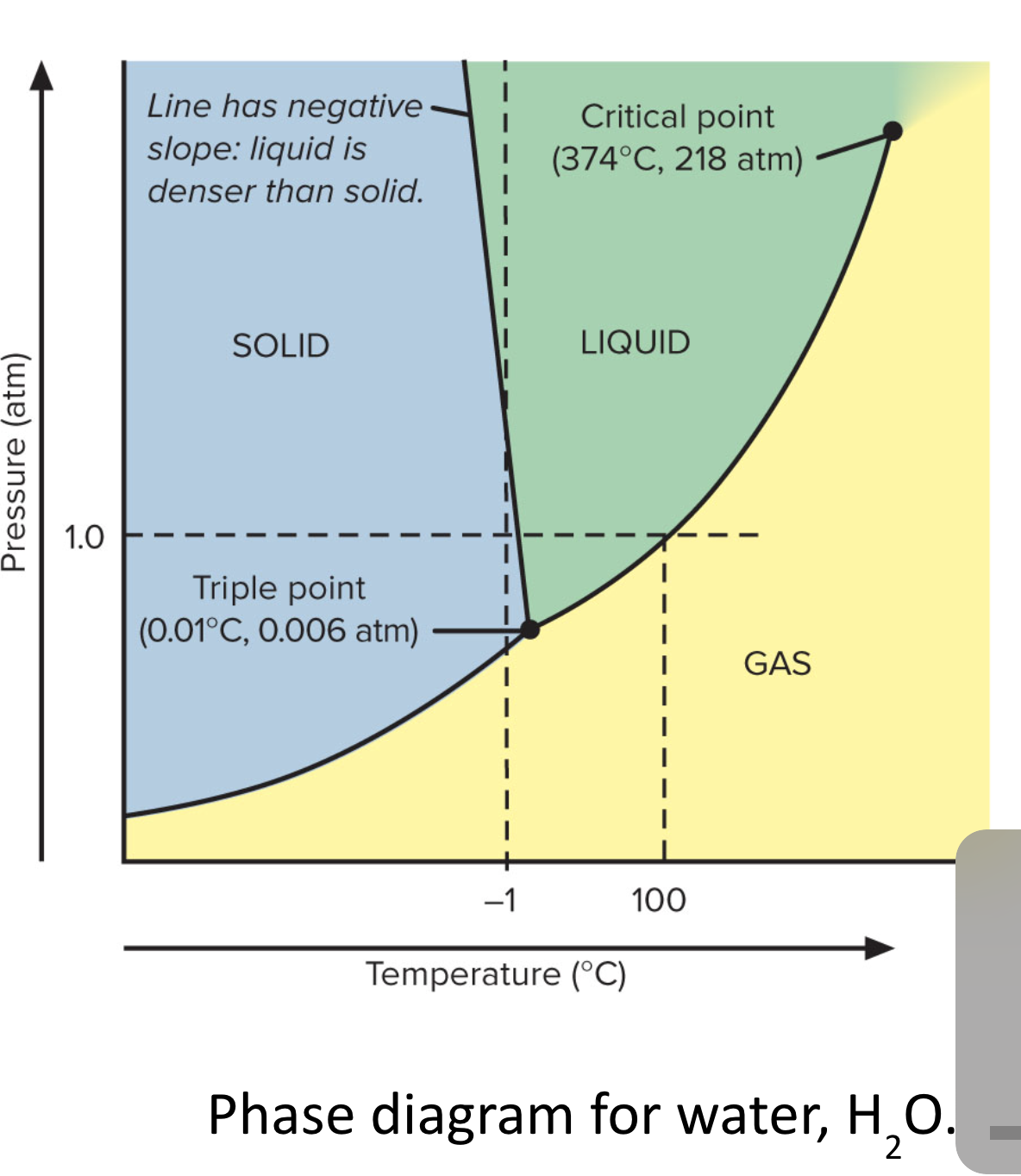
65. Use the phase diagram for water, what label should go in box F?
a. | solid | c. | gas | e. | critical point |
b. | liquid | d. | triple point |
c. | gas |
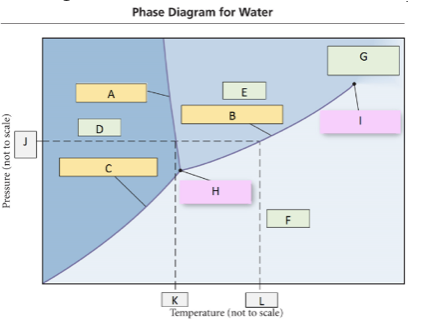
66. What change of state would occur going from D to E (crossing line A)?
a. | freezing | c. | boiling | e. | deposition |
b. | melting | d. | sublimation |
b. | melting |
67. The weakest attractions exist between particles of a _____ and the strongest attractions exist between particles of a _____.
a. | liquid, gas | d. | solid, gas |
b. | gas, liquid | e. | gas, solid |
c. | liquid, solid | ||
e. | gas, solid |
68. LiCl is a(n):
a. | non-polar molecule | d. | covalent compound |
b. | ionic formula unit | e. | Both a) & c) |
c. | metal | ||
b. | ionic formula unit |
69. Which of the following is an example of a covalent network solid?
a. | Graphite, C(s) |
b. | Sucrose, C12H22O11 |
c. | Ammonia, NH3 |
d. | Boron trifluoride,BF3 |
e. | Sulfur hexachloride, SCl6 |
a. | Graphite, C(s) |
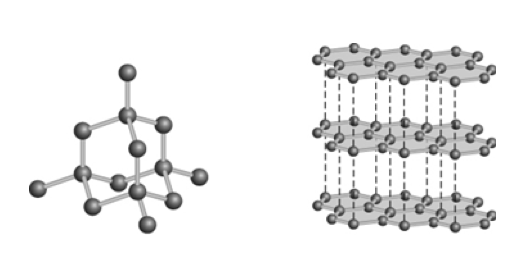
70. Diamond and graphite differ in that
a. | only graphite is composed of carbon atoms. |
b. | bonding hybridization is not the same. |
c. | only graphite burns in oxygen to give carbon dioxide gas. |
d. | diamond is less dense than graphite. |
e. | diamond is a compound. |
b. | bonding hybridization is not the same. |
71. What type of substance is graphite?
a. | ionic | d. | metallic |
b. | molecular | e. | None of these. |
c. | covalent network | ||
c. | covalent network |
72. Diamond and graphite differ in that
a. | only graphite is composed of carbon atoms. |
b. | only graphite conducts electricity. |
c. | only graphite burns in oxygen to give carbon dioxide gas. |
d. | diamond is less dense than graphite. |
e. | diamond is a compound. |
b. | only graphite conducts electricity. |
73. Diamond and graphite are allotropes. Why does one conduct electricity while the other doesn’t?
a. | sp3 hybrid bonding in diamond allows loose electrons to move. |
b. | sp2 hybrid bonding in graphite allows delocalized p electrons to ‘flow’. |
c. | Diamond’s electrons are delocalized and able to flow. |
d. | Diamond transmits light which allows electrons to flow. |
e. | The graphite electrons are highly localized. |
b. | sp2 hybrid bonding in graphite allows delocalized p electrons to ‘flow’. |
74. Which of the following substances is the best conductor of electricity?
a. | graphite, C | c. | quartz, SiO2 |
b. | diamond, C | d. | solid NaCl |
a. | graphite, C |
75. Why are diamonds so hard?
a. | Because they are made of carbon. |
b. | Because they are made of a three dimensional array of particles. |
c. | Because it is able to conduct electricity. |
d. | Because there are covalent bonds between particles. |
e. | None of the above. |
b. | Because they are made of a three dimensional array of particles. |
76. Which forces exist between iron, Fe, particles?
a. | dispersion | d. | dipole-dipole | |
b. | metallic bonding | e. | dipole-induced dipole | |
c. | hydrogen bonding | |||
b. | metallic bonding |
77. What is the basis of metallic bonding?
a. | The attraction of metal ions for delocalized electrons. |
b. | The attraction between neutral metal ions. |
c. | The neutralization of protons by electrons. |
d. | The attraction of oppositely charged ions. |
e. | The sharing of two valence electrons between two atoms. |
a. | The attraction of metal ions for delocalized electrons. |
78. Why is water an extraordinary substance?
a. | Water has a low molar mass, yet it is a liquid at room temperature. |
b. | Water is the main solvent within living organisms. |
c. | Water has an exceptionally high specific heat capacity. |
d. | Water has strong hydrogen bonding. |
e. | All of the above. |
e. | All of the above. |
Matching
a. | ionic bond | d. | sigma bond |
b. | metallic bond | e. | covalent bond |
c. | hydrogen bond | f. | pi bond |
1. A bond formed through the electrostatic attractive forces between delocalized electrons.
2. Two p orbitals laterally overlap to form this bond.
3. A bond formed through the sharing of electrons.
4. A bond formed through electrostatic attraction.
5. The strongest type of covalent bond; formed by overlapping between atomic orbitals
6. The strongest type of intermolecular force.
B metallic bond
F pi bond
E covalent bond
A Ionic bond
D sigma bond
C hydrogen bond
Matching
a. | covalent bond | c. | polar covalent bond |
b. | ionic bond | d. | coordinate covalent bond |
7. involves an unequal attraction of atoms for electrons in a molecule
8. a pair of bonding electrons originates from one atom
9. describes the bonding type in hydrogen chloride, HCl
10. most likely to occur between metals and non-metals
11. forms when electron orbitals overlap
12. occurs when electronegativity difference is greater than 1.7
C polar covalent bond
D coordinate covalent bond
C polar covalent bond
B ionic bond
A covalent bond
B ionic bond
a. | polar |
b. | non-polar |
c. | ionic |
13. lithium chloride, LiCl
14. ethane, C2H6
15. potassium bromide, KBr
16. carbon tetrachloride, CCl4
17. carbon dioxide, CO2
18. hydrogen bromide, HBr
C ionic
B non-polar
C ionic
B non polar
B non polar
A polar
a. | polar | d. | sp2 hybridized central atom |
b. | octahedral electron geometry | e. | sp hybridized central atom |
c. | nonpolar, but contains a polar covalent bond | f. | see-saw molecular geometry |
19. SF4
20. XeCl4
21. CH2F2
22. BCl3
23. BeF2
F see-saw molecular geometry
B octahedral electron geometry
A polar
C nonpolar, but contains a polar covalent bond
E sp hybridized central atom
1. Which of these compounds contain elements that DO NOT follow the octet rule? Explain.
a. NF3
b. PCl2F3
c. SF4
d. BH3
SF₄ (Sulfur Tetrafluoride)
Does not follow the octet rule because sulfur has 10 electrons in its valence shell, exceeding the octet rule. This is possible because sulfur is in Period 3, meaning it has access to the d-orbitals and can accommodate more than 8 electrons, forming an expanded octet.
BH₃ (Boron Trifluoride)
B only has 5 electrons and is thus described as electron deficient. Boron is an exception to the octet rule, often forming electron-deficient compounds. It remains stable with fewer electrons due to its small size and low electronegativity.
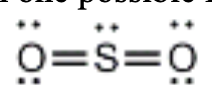
2. In molecules with more than one possible Lewis structure, formal charge is used to predict the most likely structure.
a) Determine the formal charge on each atom:
FC= v.e. - (1/2 bp e-s + lone pairs e-s)
For both (b/c theyre the same) Oxygens:
ve - 6
½ bp - 4/2 = 2
lone pairs (the electrons) = 4
FC = 6 - (2 + 4)
= 0
For Sulfur:
ve= 6
½ bp = 8/2 = 4
lone pairs = 2
FC = 6 - (4+2)
= 0
therefore the formal charge on each atom is 0

2. In molecules with more than one possible Lewis structure, formal charge is used to predict the most likely structure.
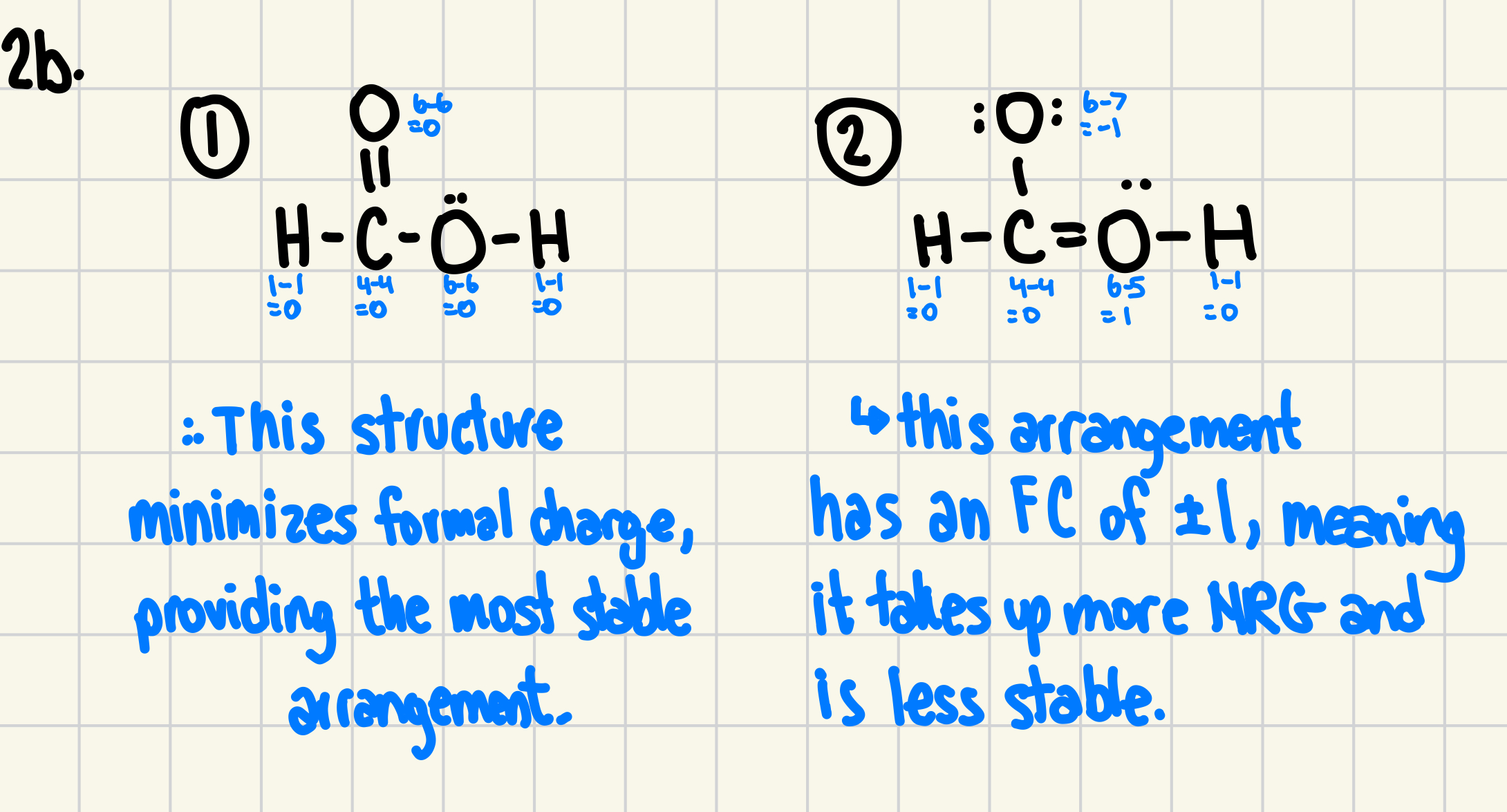
b) Formic acid has a formula of CHOOH, and has two possible Lewis structures. Use formal charges to predict the correct structure.
3. For each of the following compounds,
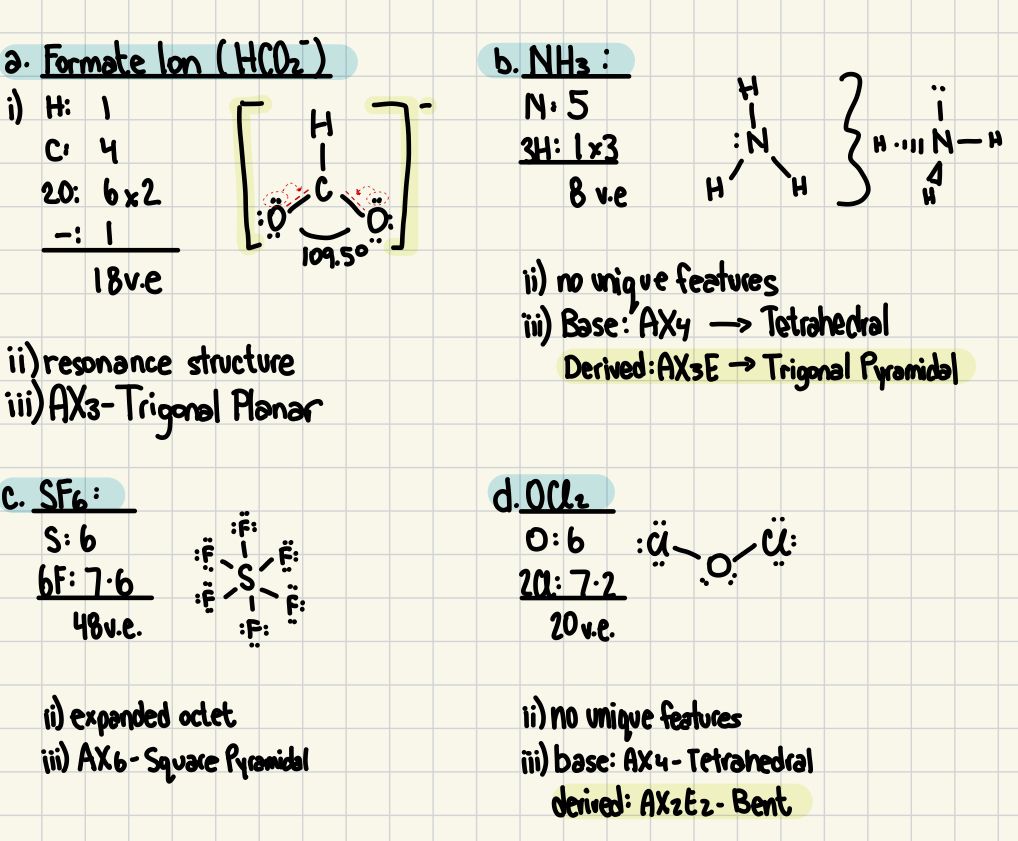
i. show all the steps and draw the Lewis structures.
ii. comment below each structure on any unique features of the structure.
iii. Give the VSEPR notation and predicted VSEPR shape.
a) formate ion (HCO2-)
b) NH3
c) SF6
d) OCl2
4. H2S has two hydrogen atoms bonded to a sulfur atom. Why isn’t the molecule linear?
H₂S is not linear because of the electron geometry around the sulfur atom. Sulfur has six valence electrons, with two forming covalent bonds with the hydrogen atoms and the remaining four existing as two lone pairs.
According to VSEPR theory, electrons pair around the central atom (sulfur) and arrange themselves to minimize repulsion. The two bonding pairs and the two lone pairs create an AX4 tetrahedral base. However, because lone pairs (which you cannot see) exert more repulsive force than bonding pairs, the molecule adopts a bent molecular shape.
5. Explain why the H2O angle in water is 104.5° even though the electron-pair geometry is tetrahedral and the tetrahedral angle is 109.5°?
In an H₂O molecule, the oxygen atom is sp³ hybridized, resulting in a tetrahedral configuration. Two of the hybrid orbitals form sigma bonds with hydrogen atoms, while the other two are occupied by lone pairs. Although the expected bond angle in a perfect tetrahedron is 109.5°, the actual bond angle in water is 104.5°. This reduction occurs because lone pair-lone pair repulsions are stronger than bond pair-bond pair repulsions, pushing the hydrogen atoms closer together.
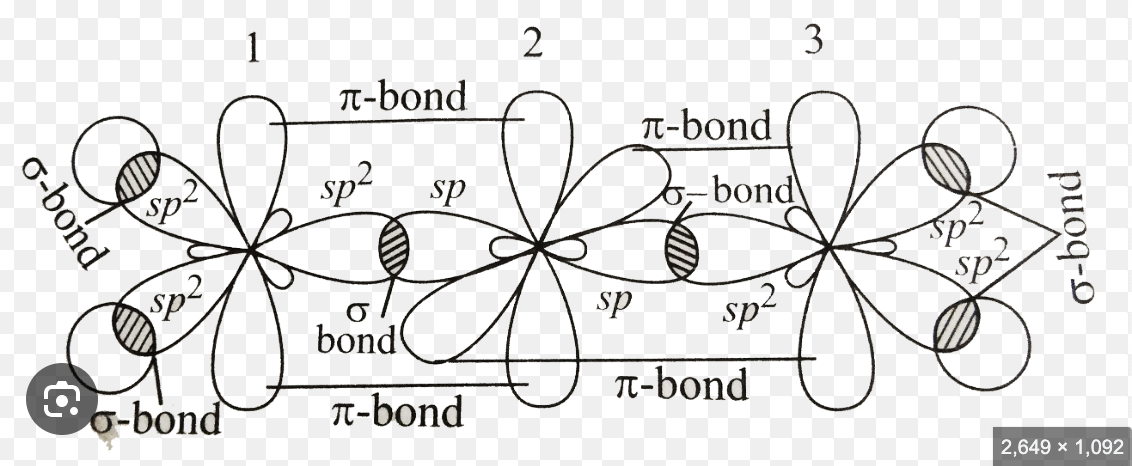
6. Use the progression developed in class (Lewis Dot ョ VSEPR Theory ョ Valence Bond Theory (hybrid orbitals) ョ sigma/pi bonding) to explain the bonding in a molecule of ethene, C2H4, (also known as ethylen e).
Based on the Lewis structure, ethene has a double bond between two carbon atoms, so each carbon needs to make three bonds. When carbon hybridizes, it typically forms four sp³ orbitals, but since only three hybrid bonding orbitals are needed, sp² hybrids form instead. This leaves one of the p orbitals unhybridized, containing a single electron. The unhybridized p orbitals on each carbon are perpendicular to the plane of the sp² orbitals, and they have a side-to-side overlap to form a pi (π) bond, completing the double bond.
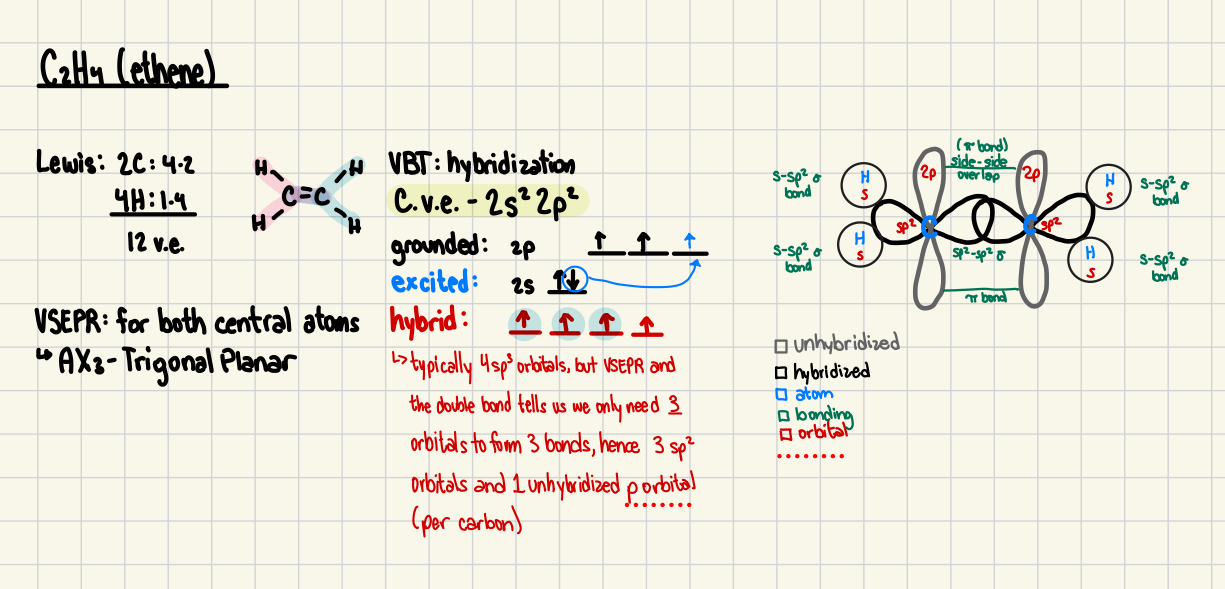
7. Compare and contrast the bonding arrangements in ethane(H3C-CH3), ethene (H2C=CH2) and ethyne(HCコCH) and give the theoretical description of the various bonds.
Ethane, ethene and ethyne are all hydrocarbons consisting of carbon-carbon bonds, but they differ in their bonding arrangements due to their distinct types of carbon-carbon bonds: single, double, and triple bonds.
In ethane, both carbon atoms are sp³ hybridized, forming a sigma (σ\sigmaσ) bond through the head-on overlap of sp³ orbitals, resulting in a tetrahedral geometry with bond angles of 109.5°.
Ethene’s carbon atoms are sp² hybridized, creating one sigma bond and one pi (π\piπ) bond from the sideways overlap of unhybridized p orbitals. This gives ethene a trigonal planar shape with 120° bond angles and prevents rotation around the double bond.
In ethyne, the carbon atoms are sp hybridized, forming one sigma bond and two pi bonds with unhybridized p orbitals. This results in a linear structure with 180° bond angles, making the triple bond rigid and preventing rotation. As the number of pi bonds increases, bond strength and rigidity also increase, influencing the molecular properties.
8. Predict the hybridization that occurs in PCl5. Explain how you reached your answer.
sp³d — • The steric number (number of electron domains) around phosphorus is 5.
• A steric number of 5 corresponds to sp³d hybridization (one s orbital, three p orbitals, and one d orbital mix to form five hybrid orbitals).
9. Compare and contrast ionic and covalent bonding using the terms “electronegativity” and “isoelectronic.”
ionic bonding: metal + nonmetal form a solid crystal lattice
Occurs between atoms with a large difference in electronegativity (typically >1.7).
One atom (usually a metal) loses electrons to become a cation, while another atom (usually a nonmetal) gains electrons to become an anion.
This creates opposite charges that create strong electrostatic attraction
Ionic compounds often involve atoms trying to achieve an isoelectronic state with the noble gases by gaining/losing e-.
Forms a crystal lattice structure with high melting and boiling points
covalent bonding: two nonmetals form a molecule
Occurs between atoms with a small or no difference in electronegativity (typically <1.7).
localized sharing in the region between nuclei of bonding atoms as each atom holds on to its own electrons (instead of transferring e-)
Atoms in a covalent bond do not necessarily become isoelectronic with noble gases, as they may share electrons rather than fully gain or lose them.
Form molecules
10. a) Use a diagram to show that water is a polar molecule.
b) Compare and contrast the terms “polar bond” and “polar molecule” with reference to methane, CH4.
a) The oxygen atom is more electronegative than the hydrogen atoms, meaning it pulls the shared electrons closer to itself, resulting in a partial negative charge (δ−) on oxygen and partial positive charges (δ+) on the hydrogens.
The bent shape of water (due to the two lone pairs and electron geometry wanting to minimize repulsion) creates an uneven distribution of charge, making it a polar molecule with a net dipole moment.
b) methane has polar C-H bonds since carbon is slightly more electronegative than hydrogen. However, methane’s shape is tetrahedral and symmetrical, causing the individual bond dipoles to cancel out. As a result, methane is a nonpolar molecule despite having polar bonds.
11.
a) Explain why CH3F is a polar molecule while CF4 isn’t.
b) How are molecules of AsF3 polar, whereas AsF5 are non-polar.
c) Predict whether the CH2F2 molecule is polar or non-polar. Justify your prediction with the appropriate data.
A) CH₃F) has a tetrahedral shape, with one fluorine atom, three hydrogen atoms, and a central carbon atom. Fluorine is highly electronegative, creating a strong dipole moment that pulls electron density toward it. Since the molecule is asymmetrical, the dipole does not cancel out, making CH₃F a polar molecule.
In contrast, carbon tetrafluoride (CF₄) also has a tetrahedral geometry, but all four surrounding atoms are fluorine. Since these fluorine atoms are symmetrically arranged, their dipoles cancel each other out. As a result, CF₄ has no net dipole moment and is a nonpolar molecule.
b) AsF₃) has a trigonal pyramidal shape due to the presence of a lone pair on the arsenic atom. This lone pair creates an asymmetry in the electron distribution, preventing the dipole moments from canceling out. Because of this asymmetry, AsF₃ is a polar molecule. On the other hand, arsenic pentafluoride (AsF₅) has a trigonal bipyramidal shape, with fluorine atoms arranged symmetrically around the arsenic atom. The symmetry causes the dipole moments to cancel out, making AsF₅ a nonpolar molecule.
C) CH₂F₂) has a tetrahedral molecular geometry, with two fluorine atoms and two hydrogen atoms attached to a central carbon atom. Fluorine is much more electronegative than hydrogen, creating strong dipole moments. However, because the molecule is asymmetrical—having two different types of atoms around the carbon—the dipoles do not cancel out completely. As a result, CH₂F₂ has a net dipole moment and is classified as a polar molecule.
12. Rank these compounds in order of increasing boiling point Br2, Cl2, I2. Explain your answer.
Cl2 (Chlorine) → Smallest, fewest electrons → Weakest LDFs → Lowest boiling point
Br2 (Bromine) → Medium size, more electrons than Cl2_22 → Moderate LDFs → Intermediate boiling point
I2 (Iodine) → Largest, most electrons → Strongest LDFs → Highest boiling point
Cl2<Br2<I2

13. a) Explain the term "sea of electrons" with respect to metallic bonding.
b) Use the following diagrams of Group 1 and 2 metal atoms to explain why metals are good conductors of electricity.
a) In metallic bonding, metal atoms release their valence electrons, which become delocalized and move freely throughout the metal lattice. This forms a "sea of electrons" that surrounds the positively charged metal ions (cations). The electrons are not bound to any particular atom, allowing them to move easily. This strong electrostatic attraction between the sea of electrons and the metal cations holds the structure together, giving metals their characteristic properties like malleability, ductility, and high thermal and electrical conductivity.
b) Metals are excellent conductors of electricity because of the presence of this sea of electrons. When an external voltage is applied, the free electrons can move rapidly through the metal, carrying electrical charge.
Group 1 Metals (e.g., Li, Na, K):
Group 1 metals have one valence electron per atom, which is easily lost to form a delocalized sea of electrons. While they conduct electricity well, the fewer free electrons mean they are generally less conductive compared to Group 2 metals.Group 2 Metals (e.g., Be, Mg, Ca):
Group 2 metals have two valence electrons per atom, contributing to a denser sea of electrons. This higher electron density results in stronger metallic bonding and better electrical conductivity compared to Group 1 metals. Additionally, the increased number of electrons allows for faster movement of charge through the lattice.
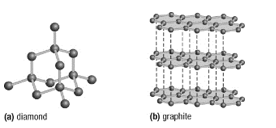
14.
a) Compare and contrast the physical properties of diamond and graphite. Explain how they differ and why they have different physical properties.
b) Refer to the accompanying diagrams (see below) to explain why graphite (b) is a good conductor of electricity and diamond (a) is not.
a)
Diamond and graphite are both made of carbon, but their different structures result in distinct physical properties. In diamond, each carbon atom forms four strong covalent bonds in a rigid, three-dimensional network with sp³ hybridization. This makes diamond extremely hard, with a high melting point of around 3800 °C. It is also an excellent thermal conductor but cannot conduct electricity because its electrons are tightly held in covalent bonds.
Graphite, on the other hand, has a layered structure with sp² hybridized carbon atoms arranged in hexagonal rings. Each carbon forms three sigma bonds and one delocalized pi bond, allowing electrons to move freely between the layers. The layers are held together by weak dispersion forces, making graphite soft and slippery. It has a similar high melting point but is a good electrical conductor due to its mobile electrons.
b) Electrical Conductivity Explanation
Diamond (a):
In diamond, each carbon atom forms four covalent bonds in a rigid, three-dimensional structure. Since all electrons are tightly held within these bonds, there are no free or delocalized electrons available to carry an electric current. This makes diamond an electrical insulator.Graphite (b):
Graphite has a layered structure with each carbon atom forming three sigma bonds and one pi bond. The fourth valence electron from each carbon remains delocalized and can move freely within the layers of graphite. This sea of delocalized electrons allows graphite to conduct electricity efficiently. The electrons move parallel to the layers, enabling electrical conductivity along the sheets. However, the weak dispersion forces between the layers prevent conductivity in the perpendicular direction.
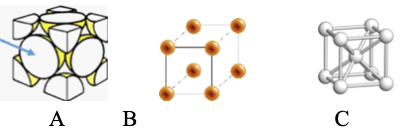
15.
a) The packing arrangement in A is called ___________while the packing arrangement in C is called __________.
b) Which arrangement is the least efficient packing system? ______
c) How many atoms belong to the packing arrangement in A? ______
d) The co-ordination number for a packing arrangement is the total number of atoms touching a central atom. What is the co-ordination number for an atom on a face in A (indicated by the arrow)? ______
a) The packing arrangement in A is called face centered cubic while the packing arrangement in C is called body centered cubic.
b) Which arrangement is the least efficient packing system? Simple Cubic
c) How many atoms belong to the packing arrangement in A? 4 atoms per unit cell (1/2 for the faces, 1/8 for the corners)
d) The co-ordination number for a packing arrangement is the total number of atoms touching a central atom. What is the co-ordination number for an atom on a face in A (indicated by the arrow)? 12