Chapter Twelve
1/67
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
Diols can be prepared from the reduction of what?
Diketones
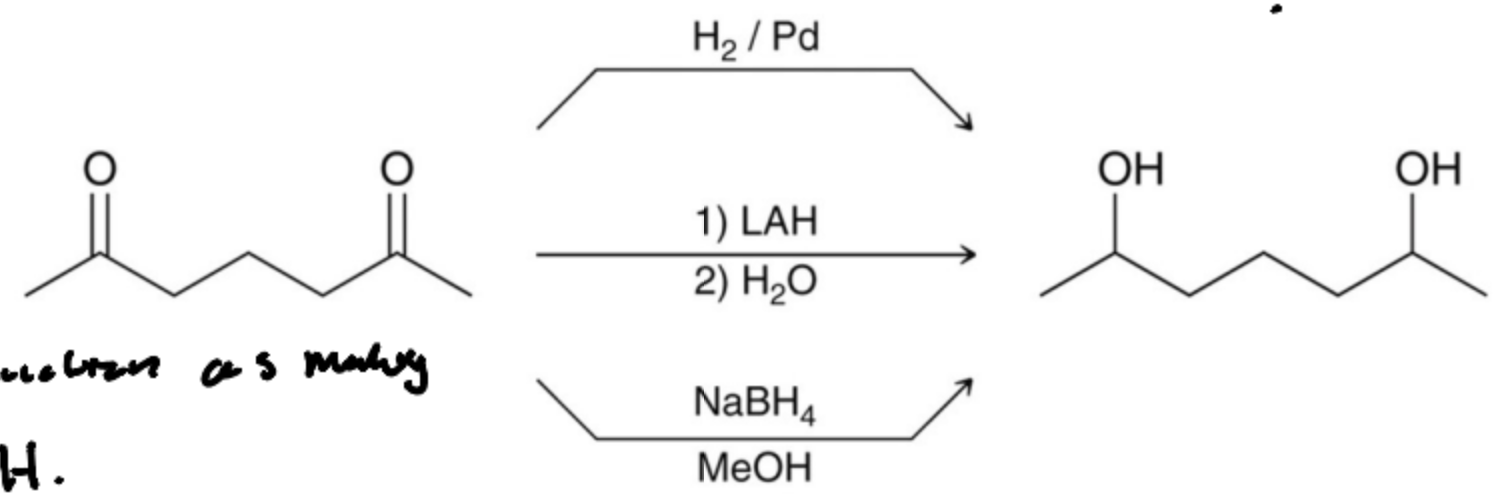
Due to the strength of LAH What can it reduce that NaBH4 cannot?
Esters and Carboxylic acids
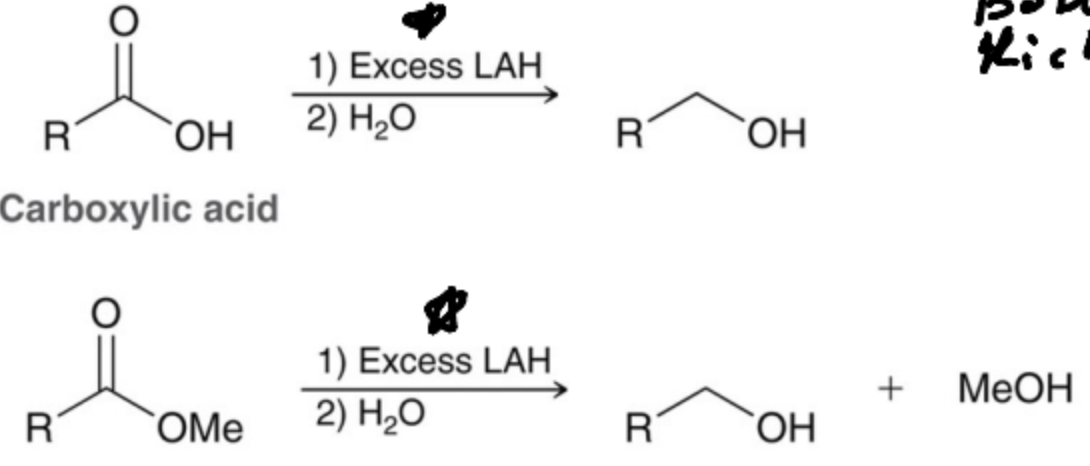
Why is excess LAH needed to reduce esters and Carboxylic acids?
A leaving group must be kicked off.
True or False
Hydride delivery agents are selective for carbonyl groups, where as hydrogenation is not.
True. Hydride delivery agents preferentially reduce carbonyl groups, while hydrogenation can reduce various unsaturated bonds.
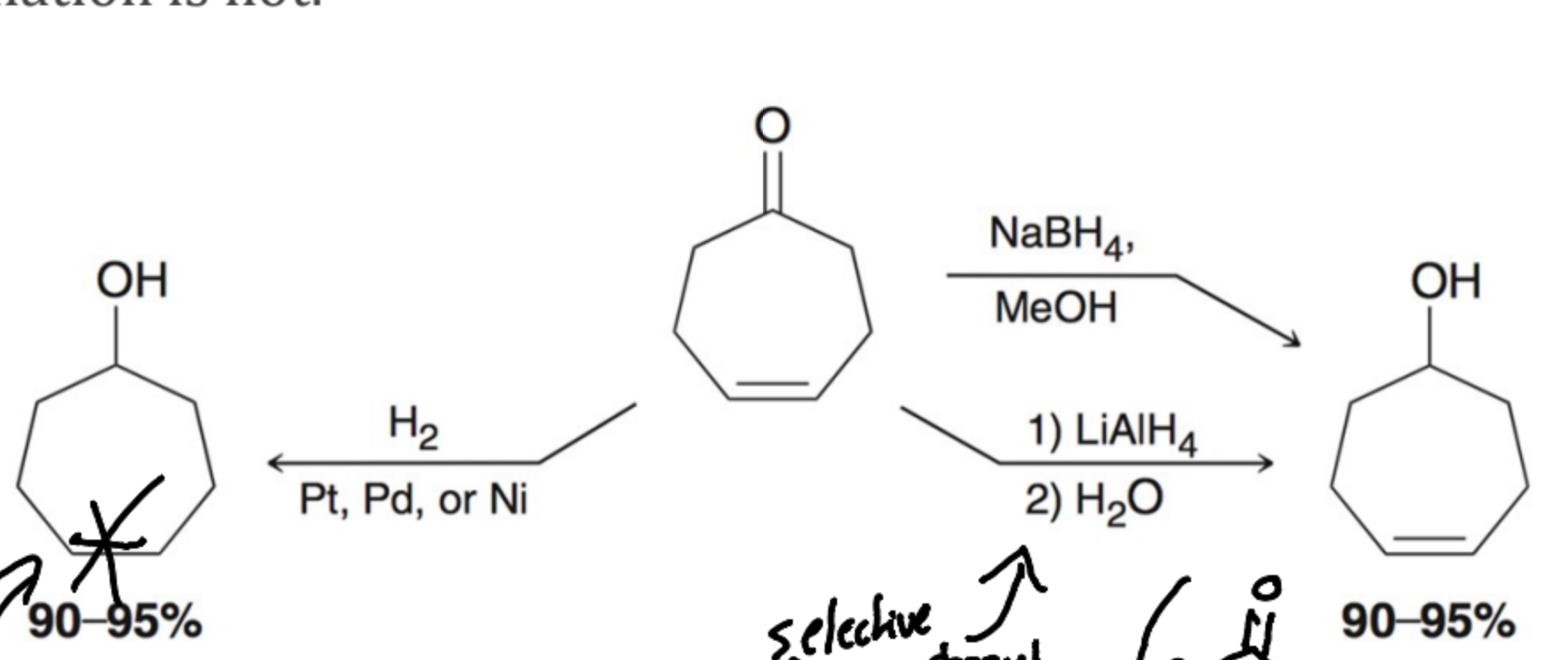
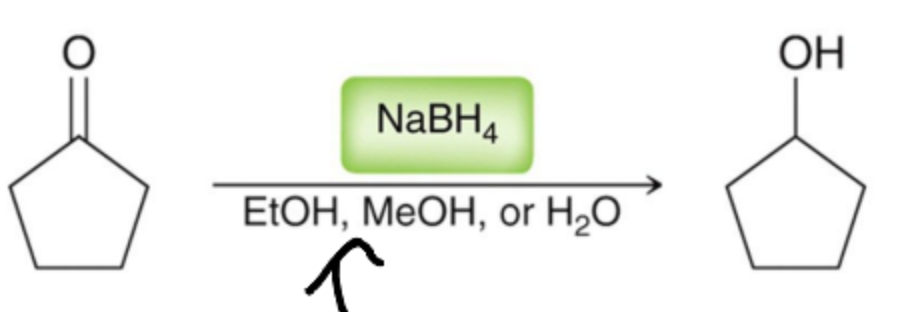
What is a common reductive agent for aldehydes and ketones?
Sodium Borohydride (NaBH4) in one step
Lithium Aluminum Hydride (LAH) is also a great Reductive agent for aldehydes and ketones, but What makes Lithium Aluminum Hydride different than sodium borohydride and why?
It is much stronger and must be done in two steps Because it can not be combines with a protic solvent.
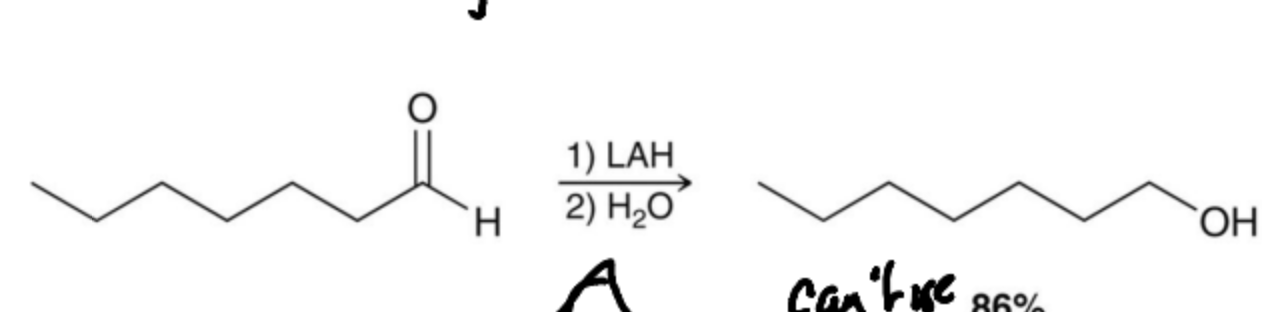
What is the first Reductive agent of an alcohol?
Catalytic Hydrogenation with is rarely used becasue of the requirement of harsh condition adn becasue it is not selective.
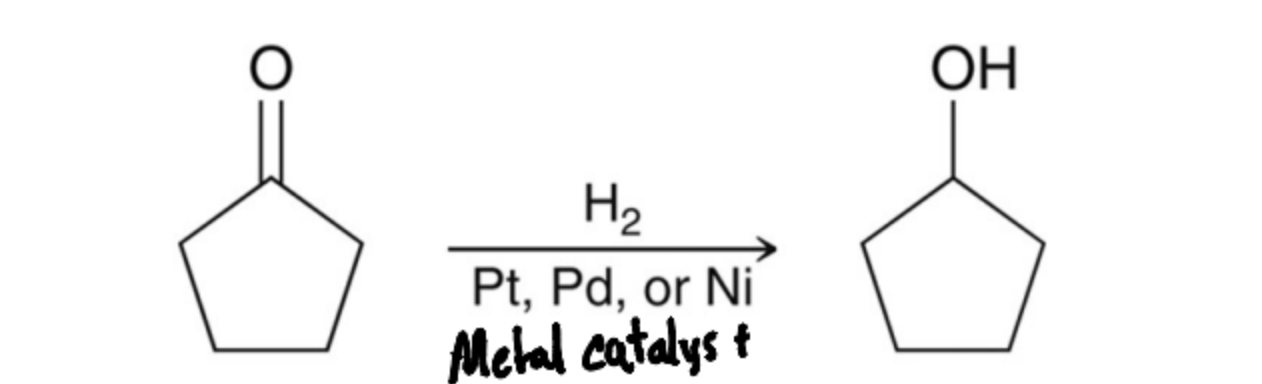
Since a ketone has trigonal Planar geometry what can Occur in a reductive reaction?
An Unsymmetrical Ketone has two R groups and thus can form a Chiral center with enantiomers, making a mix of products.
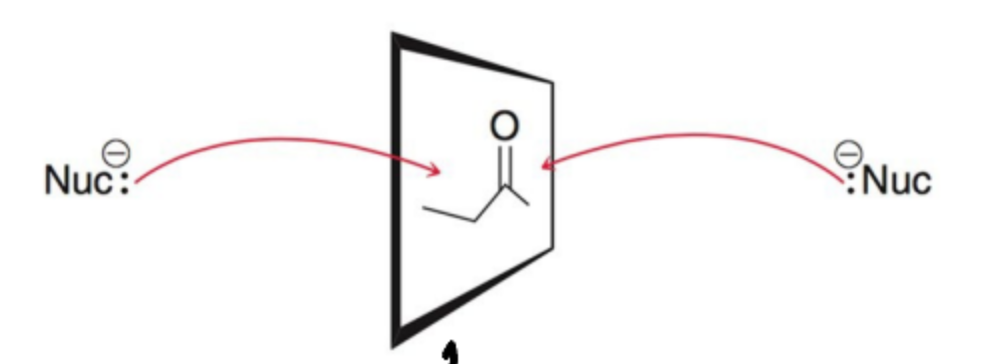
What can the oxidation states for a carbon range from?
-4 to +4 in even increments
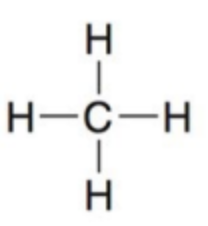
What molecule is this and what is the oxidation state?
Methane; -4
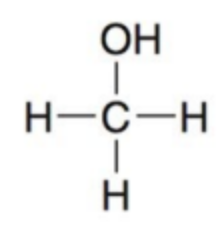
What molecule is this and what is the oxidation state?
Methanol; -2
What molecule is this and what is the oxidation state?
Formaldehyde; 0
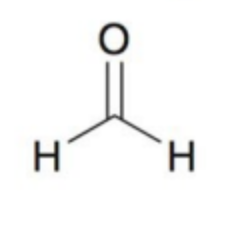
What molecule is this and what is the oxidation state?
Formic Acid; +2
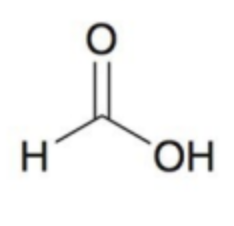
What molecule is this and what is the oxidation state?
Carbon dioxide; +4

In an oxidation reaction what happens to the carbon’s oxidation state?
It increases as electrons are lost.
In a recution reaction what happens to the carbon’s oxidation state?
It decreases as electrons are gained.
What is required to convert a carbonyl to an alcohol?
A reducing agent, such as lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4).
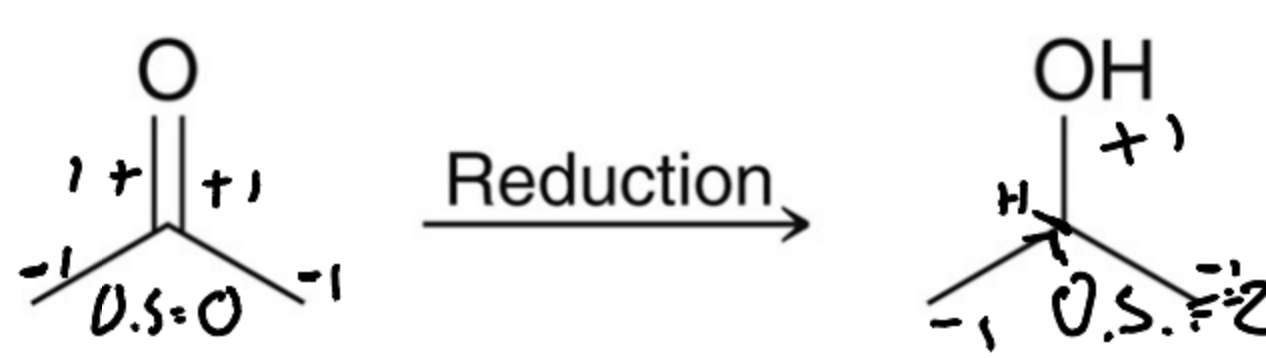
What is a third method to prepar alcohols?
A reduction of a carbonyl using a metal hydride or catalytic hydrogenation.
What do reduction involve?
A change in Oxidation state
to determine the oxidation state of an atom what do you do?
You imagine the electrons in a bond as a lone pair on the more electronegative atom
What is the second way to synthesize alcohols?
The second way to synthesize alcohols is through the hydrolysis of alkenes or alkynes, where water is added across the double or triple bond.
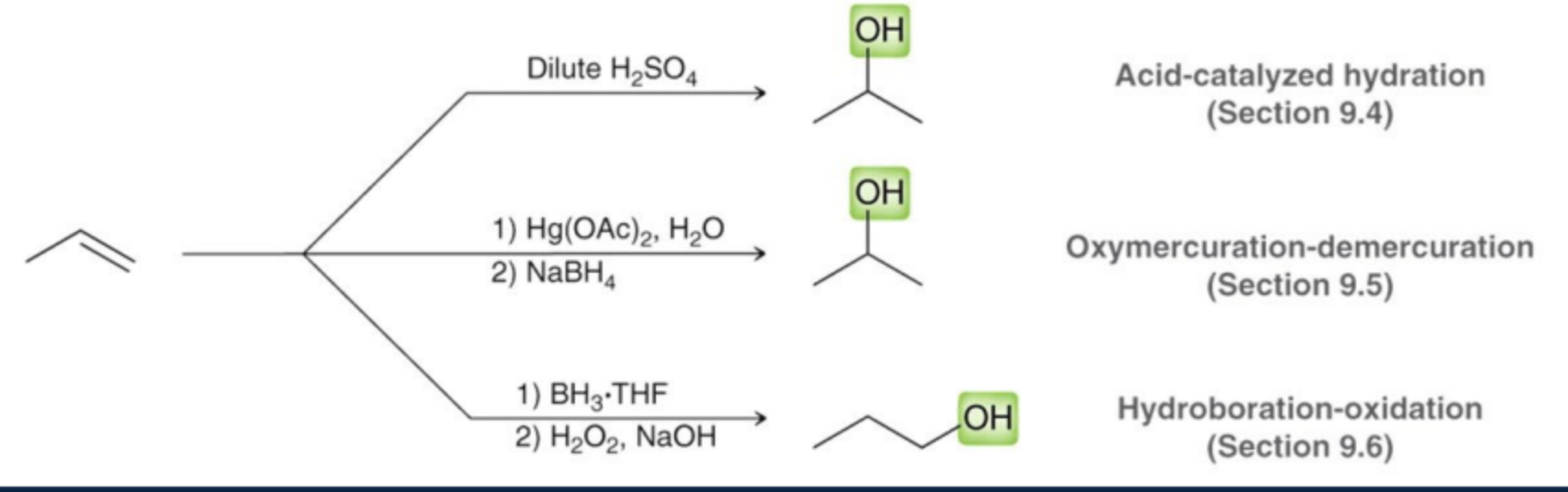
What is the first way to synthesize alcohols?
Substituting alkyl halides
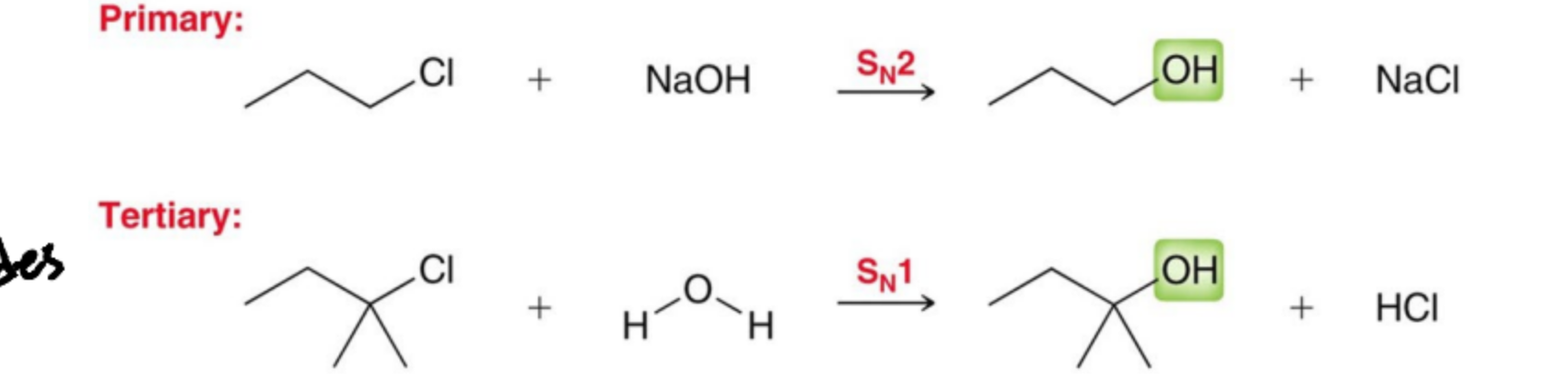
What is necessay to deprontate and alcohol?
A strong base
What is often used to regenerate the coresponding alkoxide
NaH
What is an alkoxide?
The conjugate base of a HOR
What metals are also good for deprotonation?
Na, K, or Li
What are smaller alkoxides that are easily solvated and stabilized by the solvent?
Weak Acids
Why is phenol more stable than cyclohexanol?
Resonance
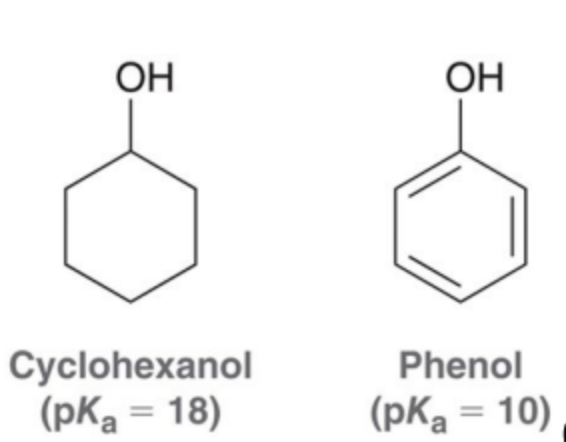
What is the cutoff of small bases?
ethoxide
What is the maximum amount of carbons that are miscible (dissolve) in water
three
What determines an alcohol’s potency as an antibacterial agent?
The sized of the hydrophobic group
Why are alcohols not all miscible in water?
Alcohols with larger hydrophobic groups are less miscible due to increased nonpolar character, which reduces their solubility in polar solvents like water.
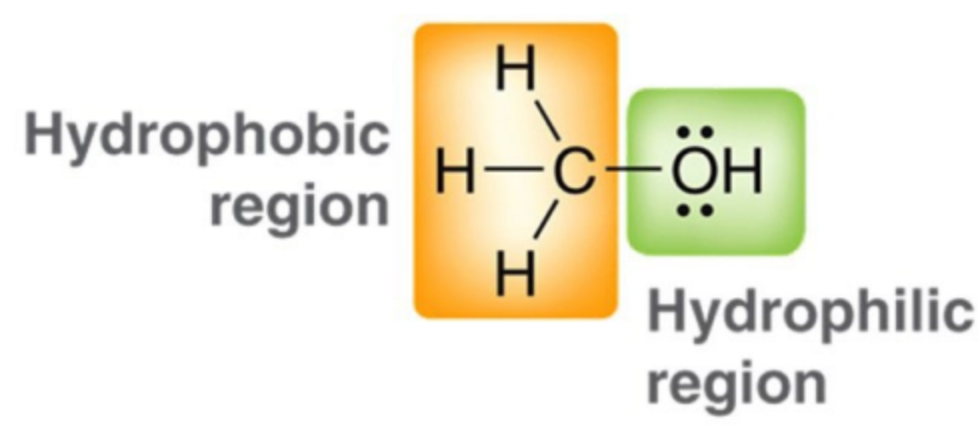
Why does size matter in alcohols for potency?
The alcohol needs some water solubility to travel through aqueous media to kill a bacterium, the alcohol should have a significant hydrophobic region to penetrate microbial membranes and disrupt their function effectively.
What is the difference between naming an alcohol and an alkane?
The parent chain suffix is “-ol” and alcohol must get priority
What is the rule of priority for when multiple alcohols, alkenes, and alkynes are present?
The rule of priority states that when naming a compound with multiple functional groups, the group with the highest priority is given the lowest possible number in the carbon chain, with alcohols taking precedence over alkenes and alkynes.
What do alcohols and phenol groups possess?
hydroxyl (-OH) functional groups.
What is the definition of Formal charge determination?
Treat all bonds as covalent and break them homolytically
What is the definition of oxidation state determination?
treat all bonds as ionic and break them heterolyticallly
What reagent is usually used in the synthesis of alcohols?
Grignard reagents
How is a grignard reagent formed?
An alkyl halide (Usually Br or Cl) is treated with Mg metal in an anhydrous ether solvent.
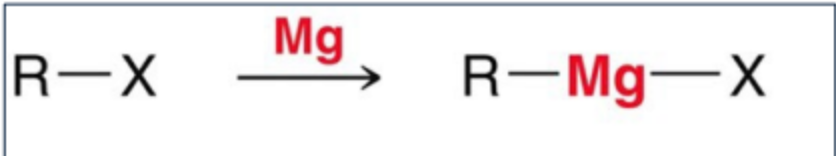
What type of Nu leophile and Base is a carboanion?
A carbanion is a strong nucleophile and a strong base due to its high electron density.
What is the key difference in the Grignard reaction versus the LAH reaction?
Water must be added after the creation of the Grignard Reagent to prevetnt eh creation of R-H.
What is the first reaction in a Grignard reaction?
The Dissociation and nucleophilic Attack of the R group to the Carbonyl ketone.
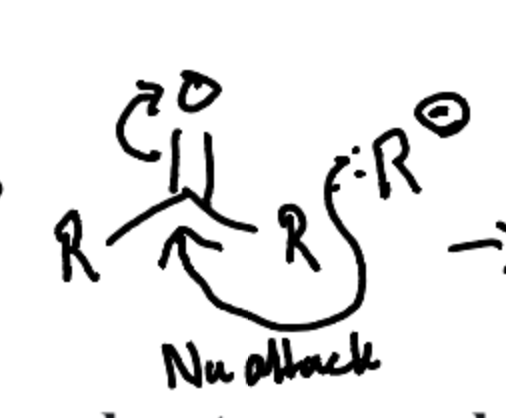
What is the second Reaction in the Grignard Reaction?
The formation of an alcohol by protonation with water after the nucleophilic addition to the carbonyl compound.
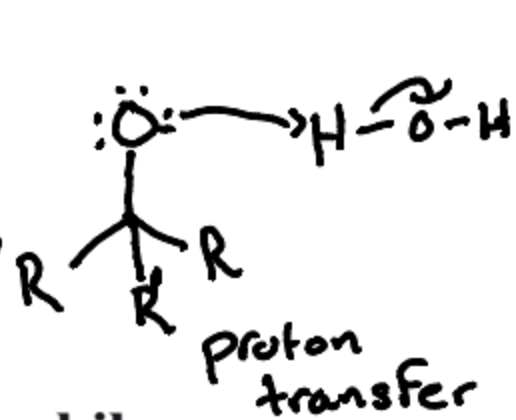
What must be avoided in the first step of grignard Reaction?
The presence of moisture or water/alcohol.
What are the steps to protect and synthesize alcohols?
Protect the -OH present
Perform Grignard Rxn
Deprotect -OH
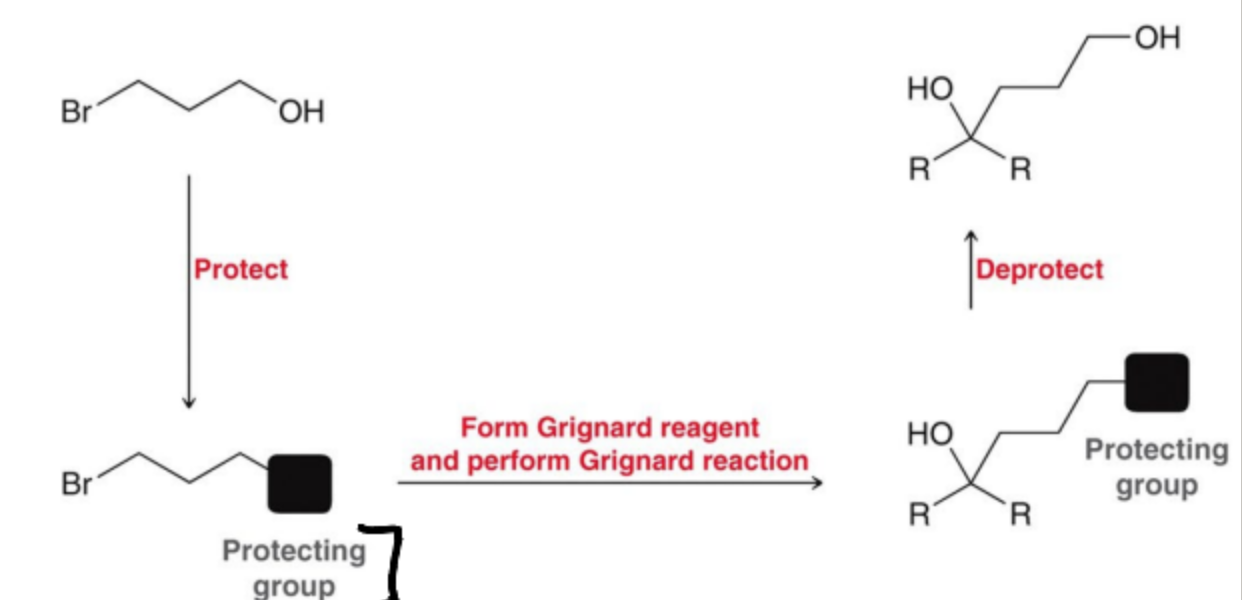
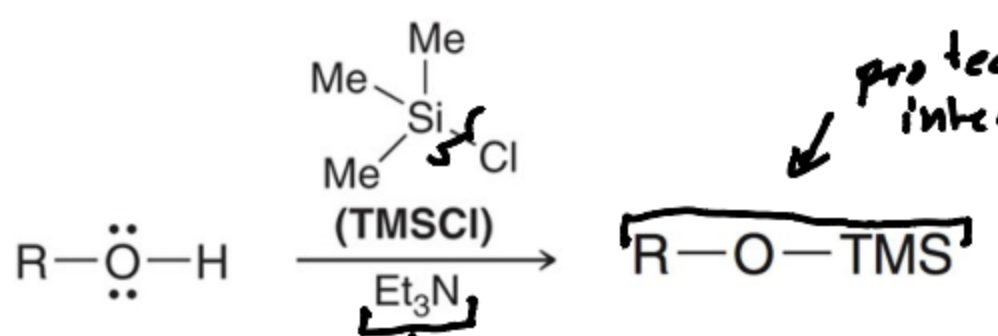
What is a protecting group generally used to protect alcohols?
Silyl ethers, such as trimethylsilyl chloride (TMSCl)
How does the substitution of a Silicon atom in TMS occur?
SN2 Mechanism
WHy can SN2 ocur with silicon but not Carbon?
Silicon is larger and open to backside attack
Why is Et3N needed in the first step of protecting alcohols?
A base is needed to neutralize HCL byproduct
Why is H3O+ pr TBAF needed in the second step of Protecting Alcohols?
Used to deprotect the alcohol
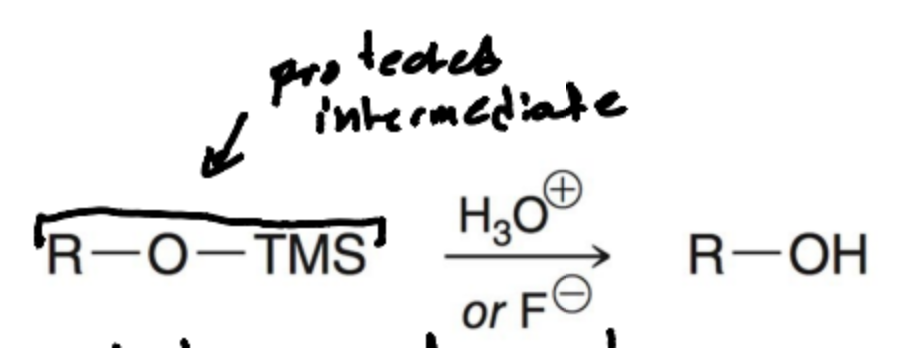
What is TBAF?
Tetrabutylammonium Fluoride is a reagent used in organic chemistry for deprotection and as a fluoride source.
If synthesizing alcohols and not needing to protet an alcohol what is teh process?
By using Grignard reaction in following order
MG
ketone
Water
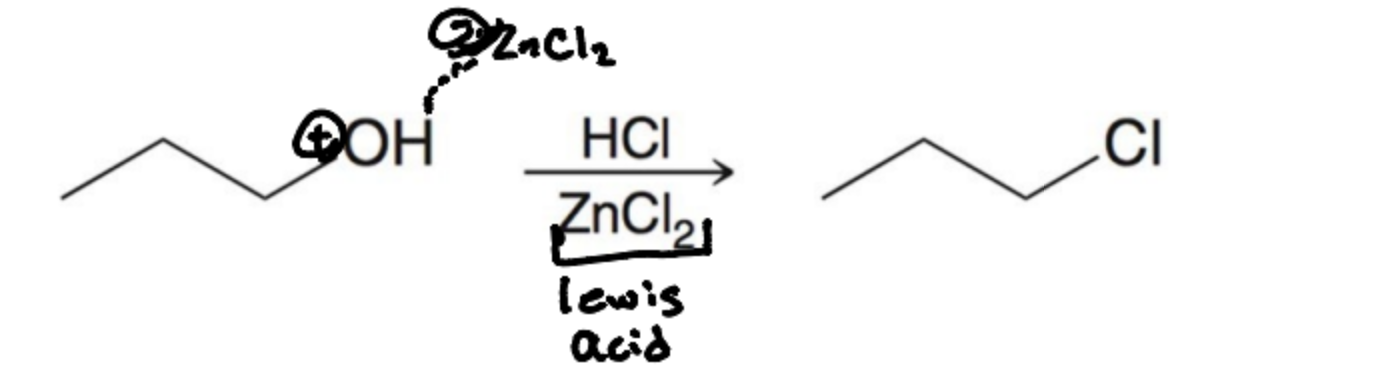
How are 3º alcohols converted to alkyl halides?
With HX in an SN1 rxn
What type of reaction converts 1º and 2º alcohols to alkyl halides?
SN2
What must be added to make an alkyl chloride?
ZNCl2 must be used wikth HCL to speed up the reaction
What reagent is specific for Alkyl Chlorine?
SOCl2/py
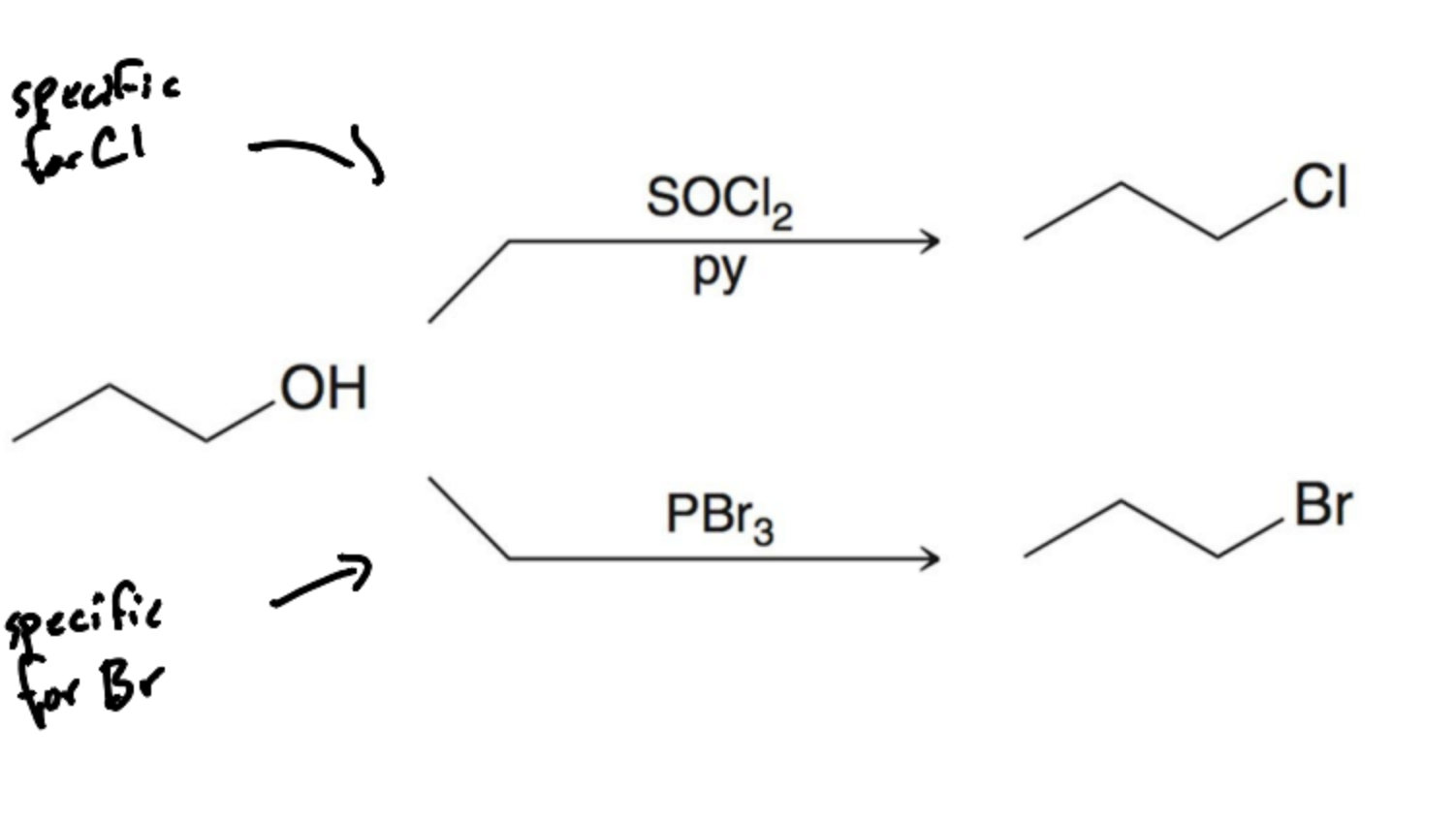
What reagent is specific for Alkyl bromine?
PBr3
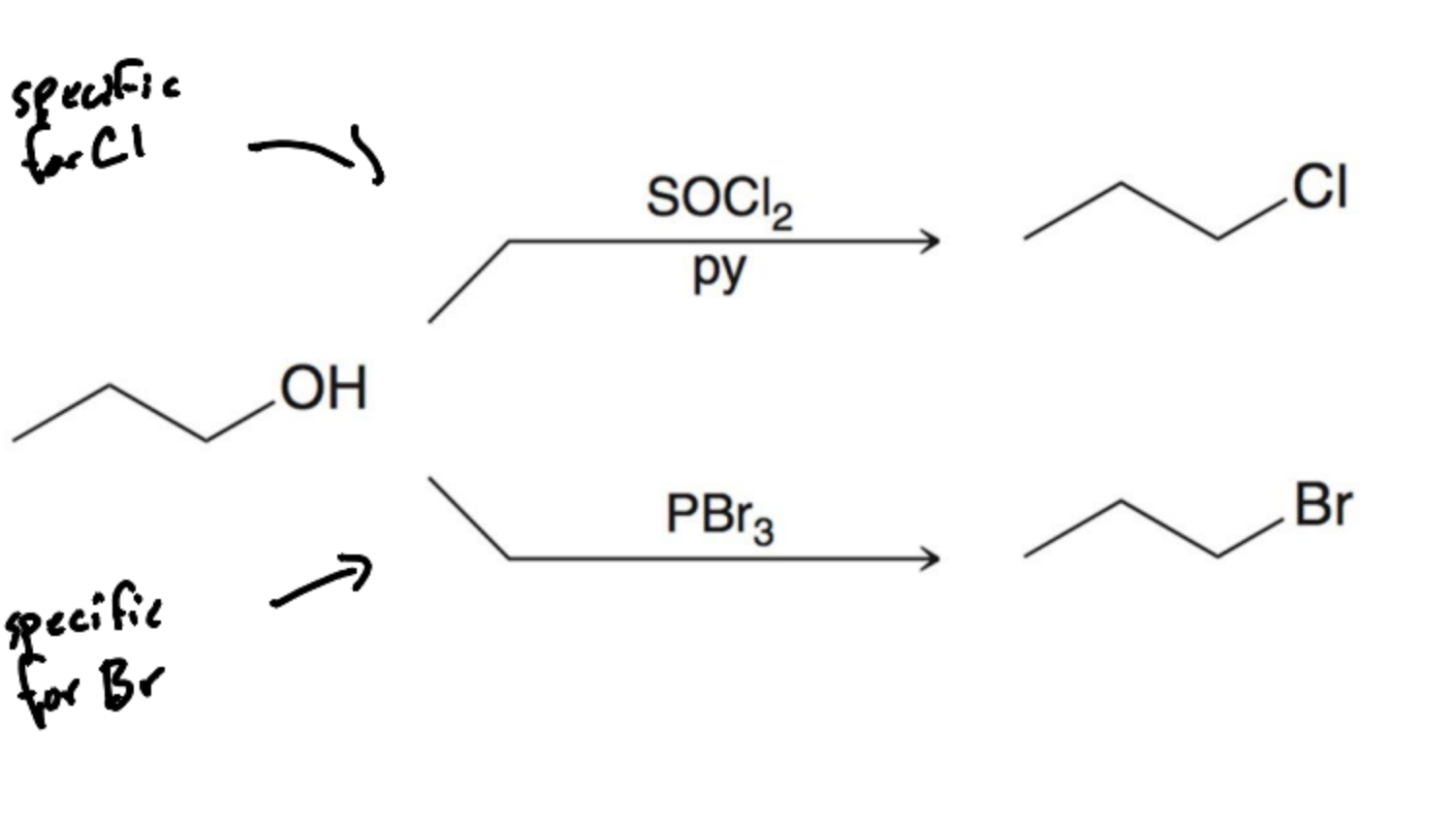
Alcohols can undergo elimination in acidic condition. generally through an E1 rxn but better leaving groups go through E2. What are the three steps?
Proton transfer
Loss of LG
Proton Transfer
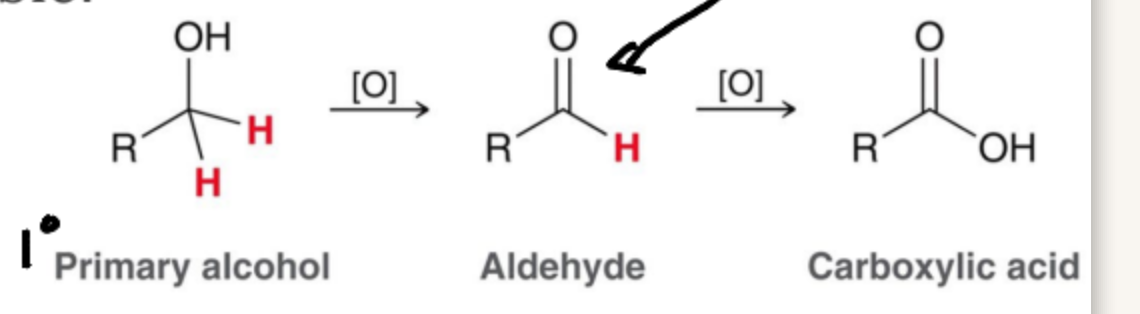
Alcohols can be formed by the reduction of a carbonyl but the reverse process, oxidation is also possible. What products can occur in a Primary Alcohol?
Aldehydes and Carboxylic acids
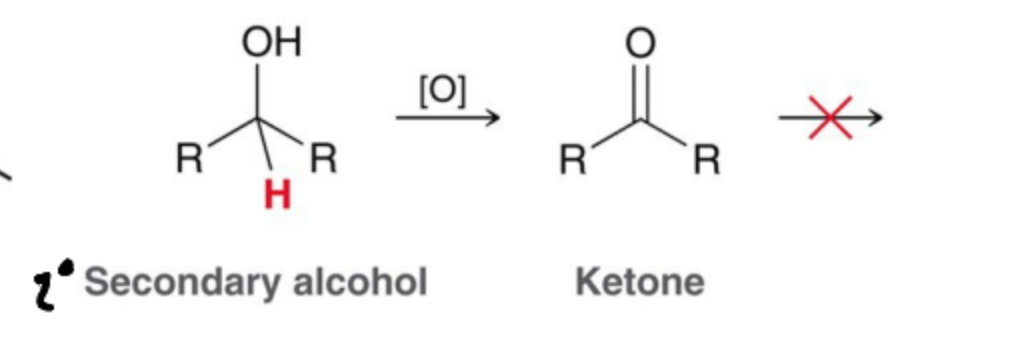
Alcohols can be formed by the reduction of a carbonyl but the reverse process, oxidation is also possible. What products can occur in a Secondary Alcohol?
Ketones
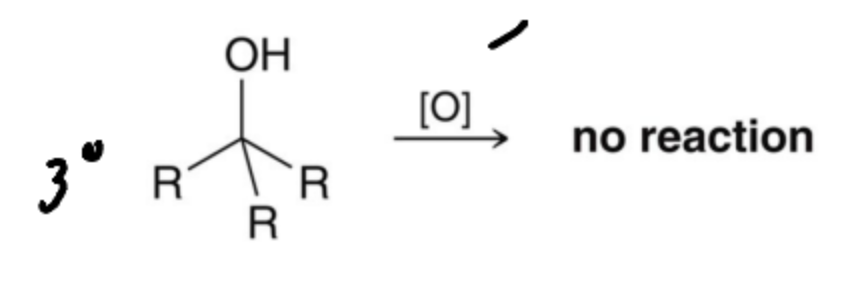
Alcohols can be formed by the reduction of a carbonyl but the reverse process, oxidation is also possible. What products can occur in a Tertiary Alcohol?
No Reaction
What oxidizing agent is formed from either CrO3 or Na2Cr2O7 in aqueous acid, and what is the product of 1º and 2º?
Chromic Acid; Carboxylic acid; Ketone
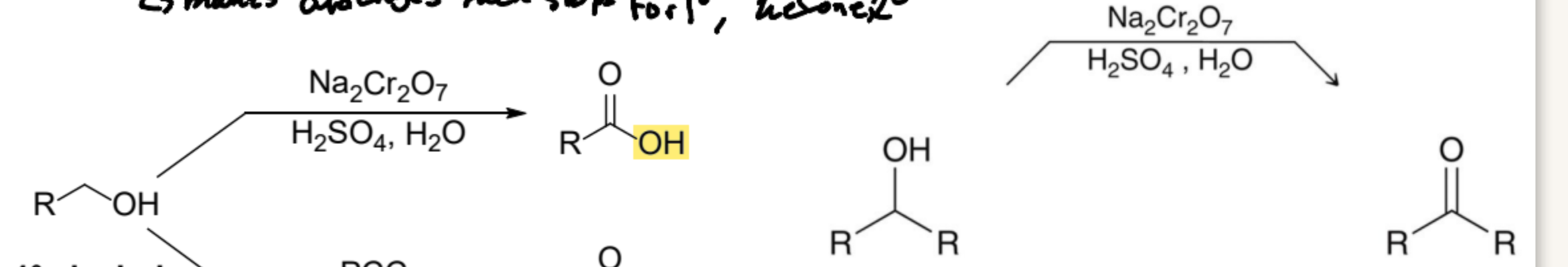
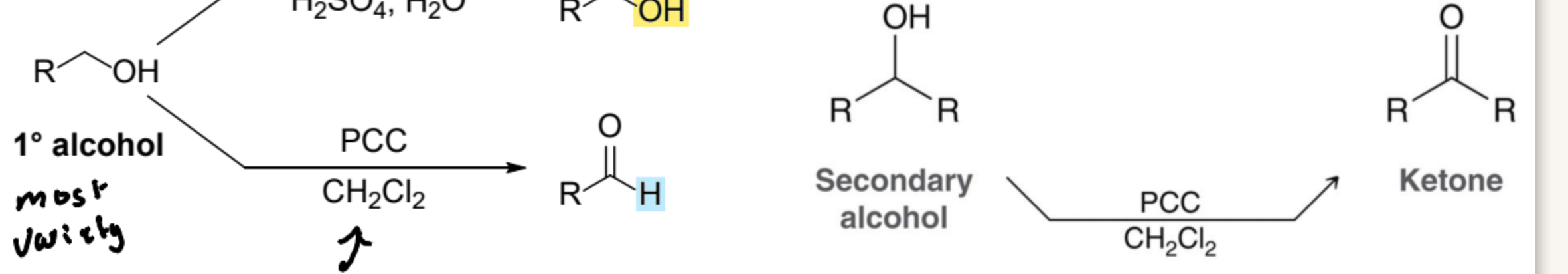
What Product is formed from the oxidizing agent, Pyridinium Chlorochromate (PCC) in 1º and 2º?
Aldehyde; Ketone
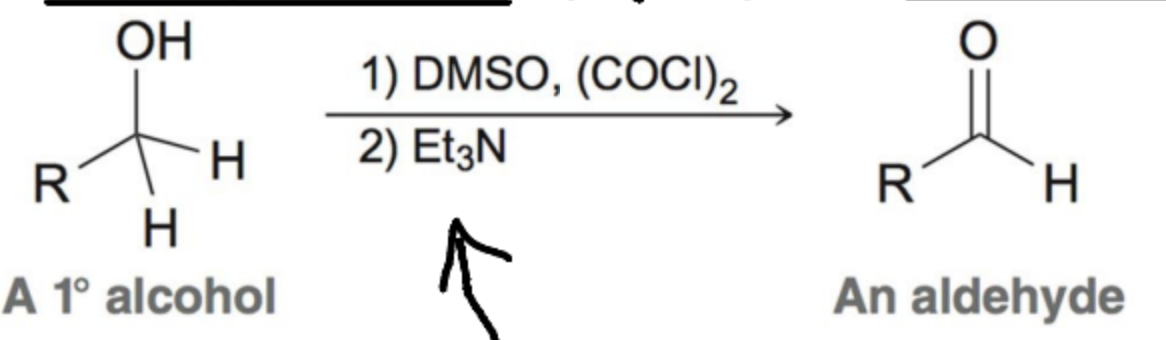
Chromium Reagents are toxix and horrible fro the environment. The swern oxidation is one such alternative, what are the reagents?
Dimethyl sulfoxide (DMSO) and oxalyl chloride form the active oxidant
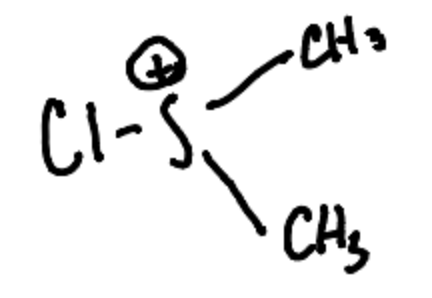
What is the Active oxidant in the Swern Oxidation?
Chlorodimethylsulfonium ion
Chromium Reagents are toxix and horrible fro the environment. The dess-Martin Periodane (DMP) oxidation is one such alternative, what are the reagents?
Dess-Martin periodinane and anhydrous solvent.
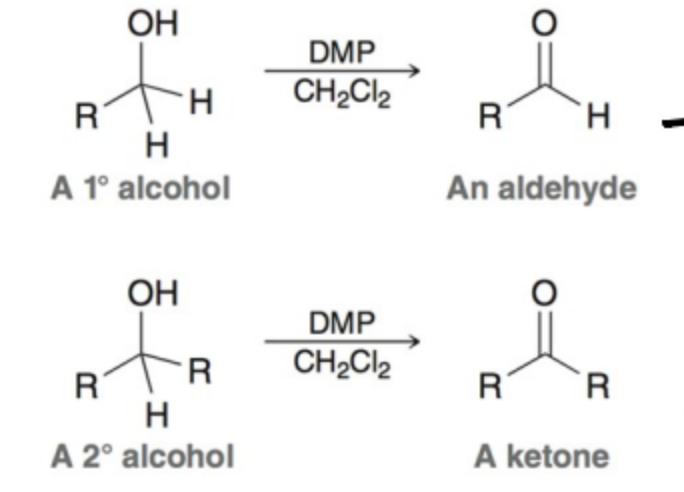
What do Swern and DMP Oxidation make as the product of 1º and 2º alcohol oxidation?
Aldehyde; ketone