Kraft Lignin Reaction
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
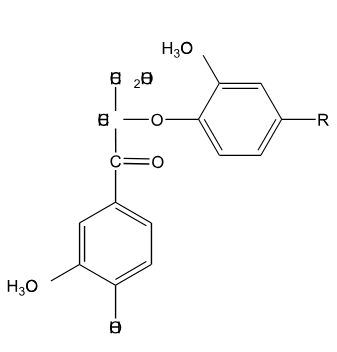
Is the following lignin compound split into two monomers through the action of NaOH at elevated temperature?
No, because of the alpha carbonyl, a double bond will form between the beta and gamma carbon. Addition of OH- will return the molecule to the original form so no cleavage.
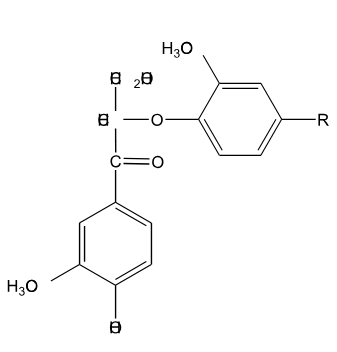
Is the following lignin compound split into two monomers through the action of NaOH at elevated temperature?
No, reaction with OH- results in the formation of a stillbene.
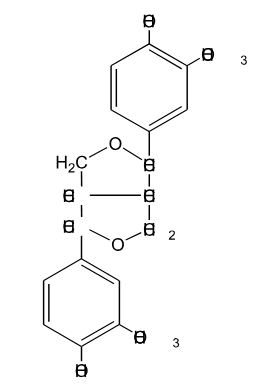
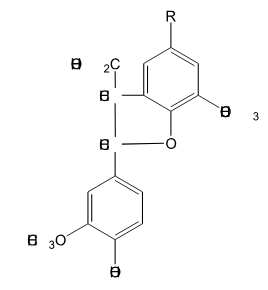
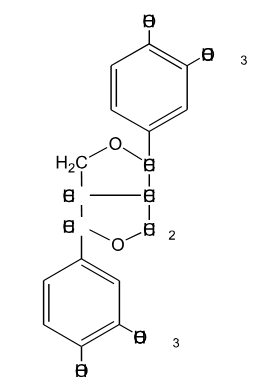
Is the following lignin compound split into two monomers through the action of HS- at elevated temperature?
No, It is very difficult to cleave carbon-carbon bonds during kraft pulping. This will not cleave.
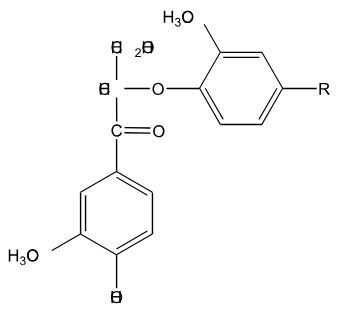
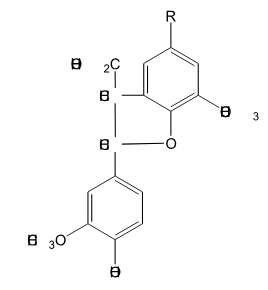
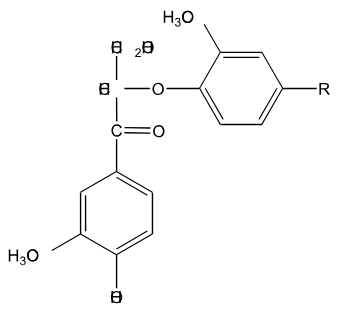
Is the following lignin compound split into two monomers through the action of HS- at elevated temperature?
Yes, this reaction involves the nucleophilic attack of the gamma carbon by HS- followed by a nucleophilic attack at the beta carbon by the S- attached to gamma carbon.
What is the important of etherified p-phenolic structure containing β-aryl ether bonds?
the β-aryl ether linkage is cleaved by hydroxide ions
this reaction promotes efficient delignification by fragmenting the lignin
generates new free phenolic hydroxyl groups
Which one is a stronger nucloephile?
OH- or HS-?
The hydrosulfide ion (HS⁻) is a stronger nucleophile than the hydroxide ion (OH⁻).
HS⁻ is a stronger base than OH⁻
The sulfide ion (HS⁻) has a larger atomic radius compared to the oxygen in OH⁻
Which of the following carbohydrates are lost to a higher degree during kraft cooking?
a) Xylans
b) Glucomannas
Glucomannans are much more highly degraded during kraft pulping. This can be seen in the table on slide #6. Of course this is more important with softwoods as there is a much higher level of glucomannans.
What is saponification?
a) The consumption of alkali through reactions with acidic groups such as carboxylic acids
b) The formation of internal esters (lactones) through the reaction between carboxylic acids and alcohols
c) The alkaline cleavage of fats resulting in the formation of fatty acids and glycerol along with consumption of alkali
c) the alkaline cleavage of fats resulting in the formation of fatty acids and glycerol along with consumption of alkali
Simple definition. The important thing here is that fats consume alkali so additional is needed for pulping.
Why is the fact that wood species can contain different levels of fats important to kraft pulping?
a) Variable levels of fats can cause problems with the EA because of alkali consumption due to saponification reactions
b) High levels of fats can cause liquor penetration.
c) High levels of fats can cause foaming problems in the recovery furnace
a) Variable levels of fats can cause problems with the EA because of alkali consumption due to saponification reactions.
A tree with higher levels of fats can require more alkali to pulp than one with lower fat levels. Of course there are also other variables to be concerned with such as the level of acidic extractives, etc.
Are pine trees acidic, basic, or neutral?
a) Acidic
b) Basic
c) Neutral
a) Acidic
There are a large number of acidic extractives in pine trees which will lower the pH. In fact, most pine trees have a pH of around 5. Once again, these acidic extractives will consume alkali.
The kraft pulping process has been divided into 3 chemical phases. In which of these phases in the majority of lignin removed?
a) Initial
b) Bulk
c) Residual
b) Bulk
Even though the removal of lignin is slow in this phase, the majority of lignin comes out in this phase. This can be seen in slide 15.
Is lignin more susceptible to reaction during the initial or bulk phase of the kraft cook?
a) Initial
b) Bulk
a) Initial
This is the portion of the cook where lignin reacts very rapidly. There is not enough lignin reactions to cause large lignin removal but there are lignin groups available in the beginning of the cook that are very reactive.