GCC Exam II Lecture VIII
1/51
Earn XP
Description and Tags
Edery Lecture 11: mRNA vaccines, from COVID to personalized cancer treatment
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Today’s Lecture
Today’s lecture
Go over immune system and its regulation by the clock
Discuss SARS-CoV2 mRNA vaccines
Discuss possible application of mRNA vaccines to cancer
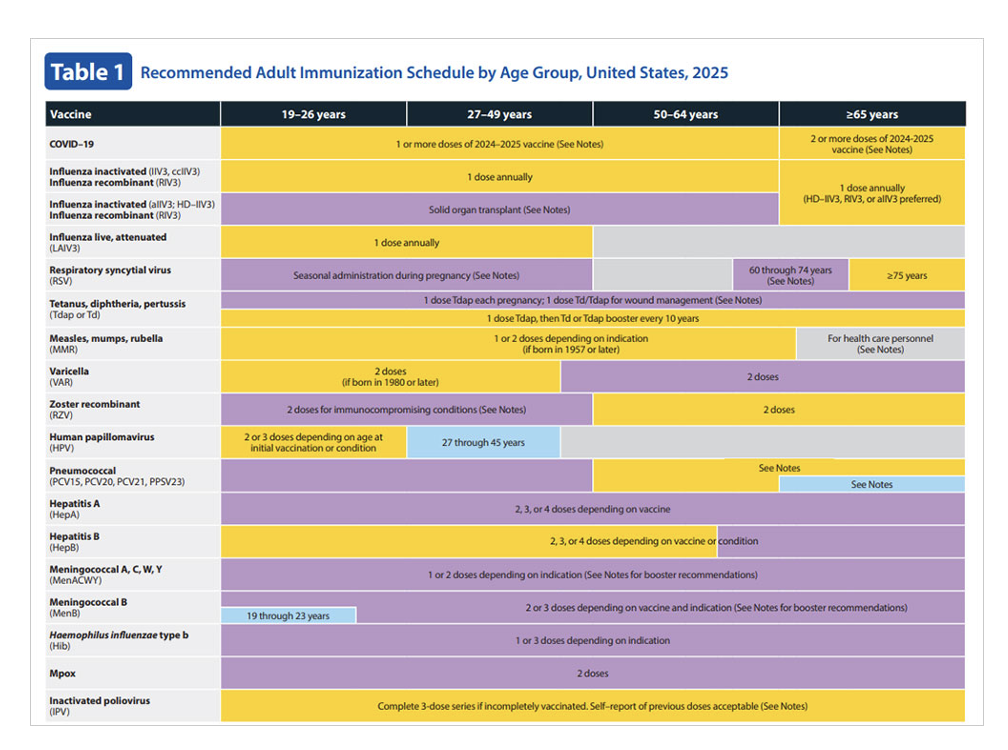
Recommended Child and Adolescent Immunization Schedule
for ages 18 and younger

Recommended Adult Immunization Schedule
By Age Group
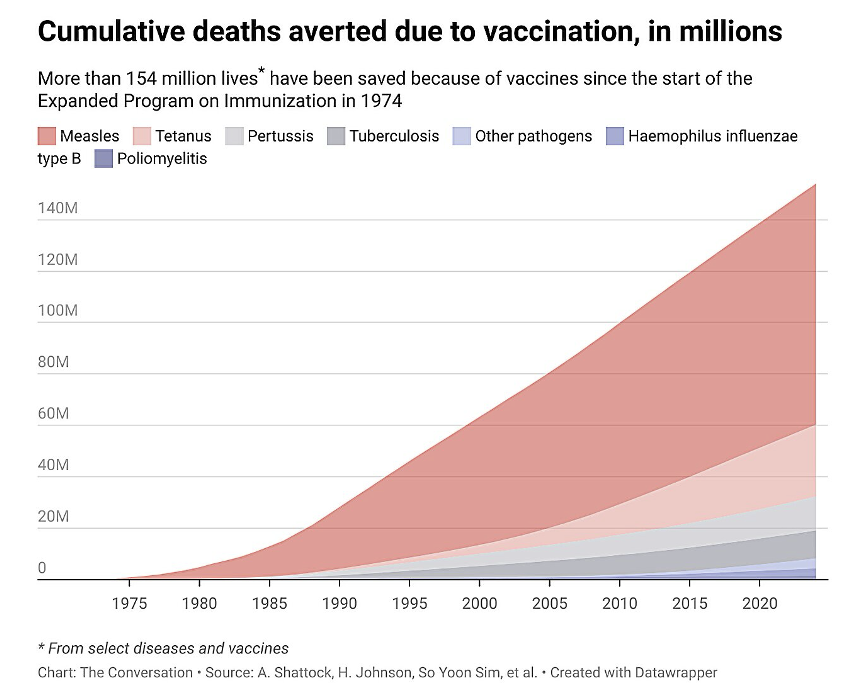
cumulative deaths averted due to vaccinations in millions
more than 154 million lives have been saved because of vaccines since the start of the Expanded Program on Immunization in 1974
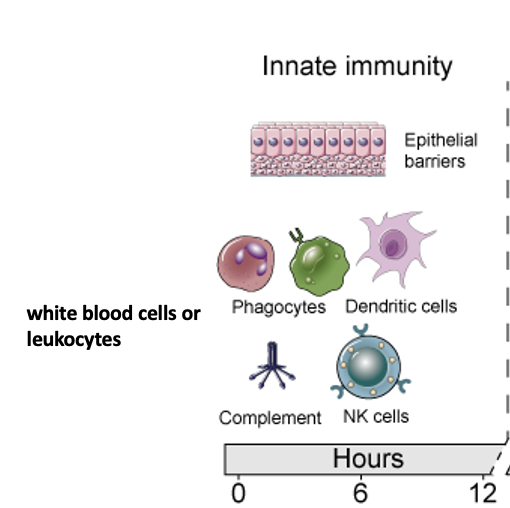
Many organ systems contribute to immunity and fighting pathogens;
ranging from immune specialized cells such as phagocytes to skin barrier and ciliated lung airways.
Innate Immunity
Innate (or “non-specific”) immunity refers to immunity present in the body designed for protection even in the absence of an antigen.
Adaptive Immunity
Adaptive (or “acquired”) immunity refers to immunity that is activated by a specific antigen. Before adaptive immunity becomes activated, the antigen has to be processed and analyzed. Adaptive immunity takes longer to develop.
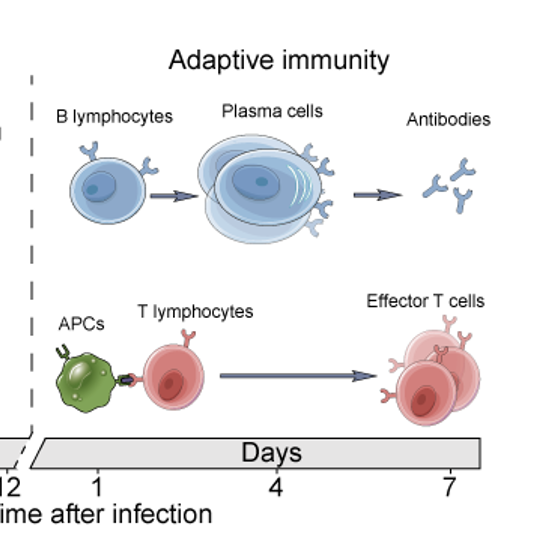
Circadian clocks regulate
all aspects of immunity and host defense mechanisms
In addition, other systemic factors (as we saw in lecture 7 and 8) can affect daily fluctuations in immune pathways. These daily changes can affect disease symptoms, susceptibility and treatment efficacy.
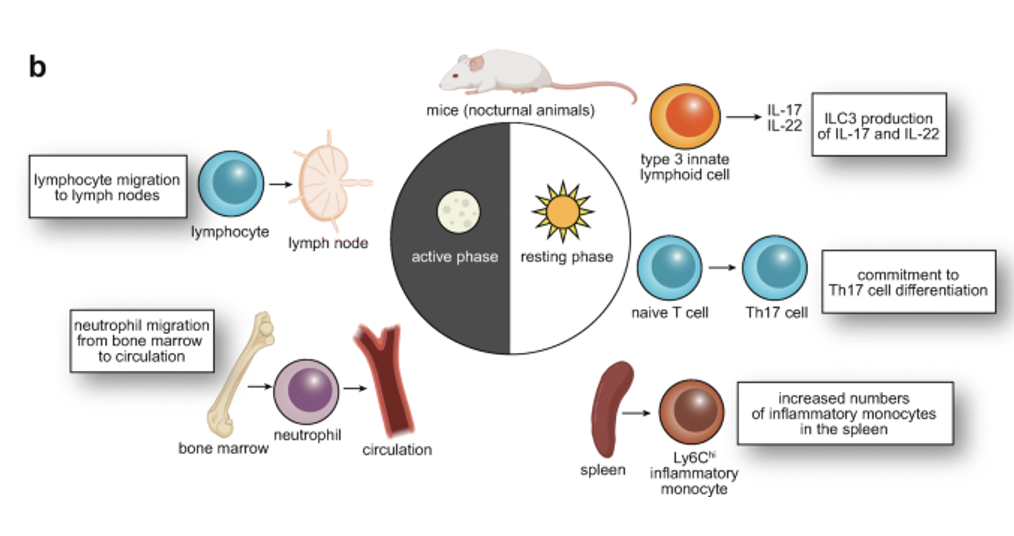
In addition, there are age and sex-linked differences in the immune system that underlie many diseases
In general, women have higher levels of circulating antibodies then men, which might enhance autoimmunity
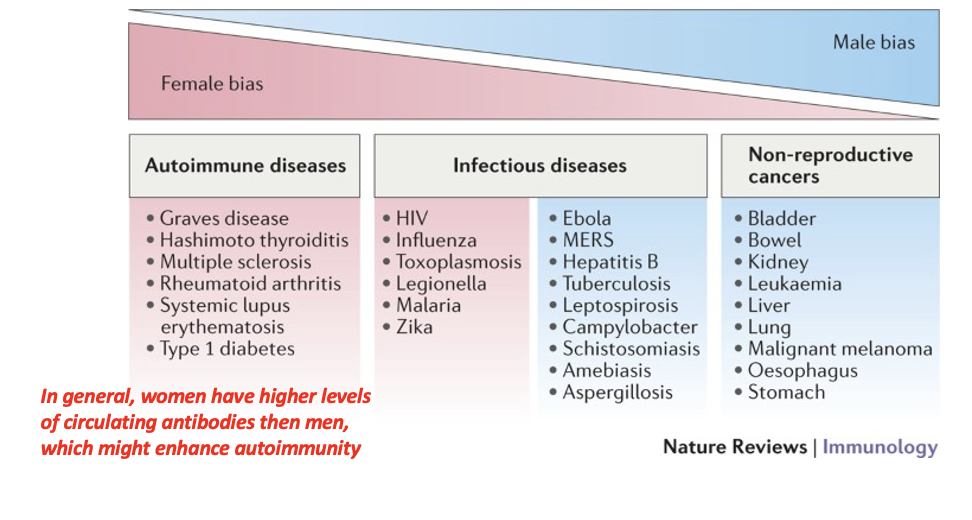
Additionally, females show increased susceptibility to
to autoimmune disease development and males show increased susceptibility to non- reproductive malignant cancers.
Reproductive status, including pregnancy, as well as immune-mediated pathology contributes to female-biased infectious diseases, whereas pathogen-associated damage, including delayed clearance, is associated with male-biased infectious diseases.
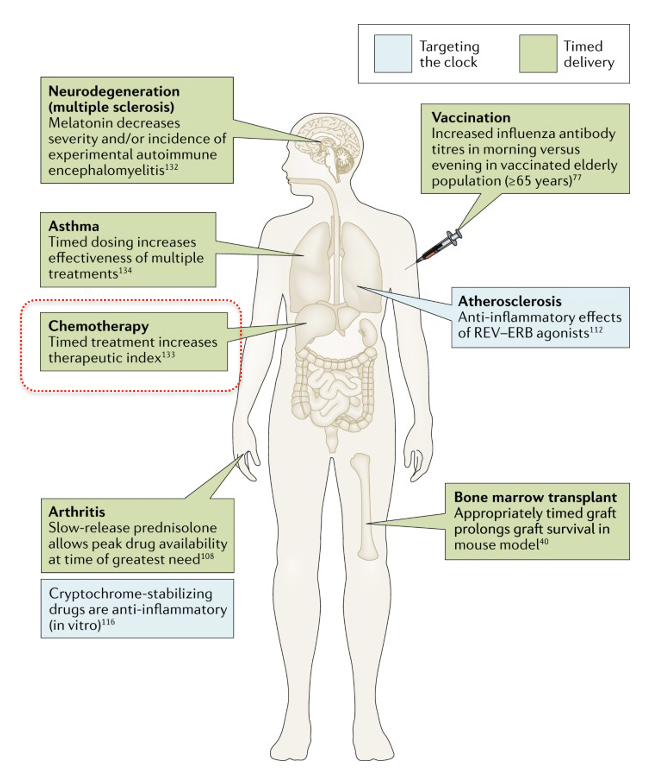
Targeting clock genes and/or timing of treatments can
improve immune responses and improve health outcomes
Examples of known cases where either targeting a clock gene (light blue), or daily timing of treatment (light green) has shown to be beneficial in combating immune/autoimmune based disorders. Some of the results shown come from mouse models, others from human studies.
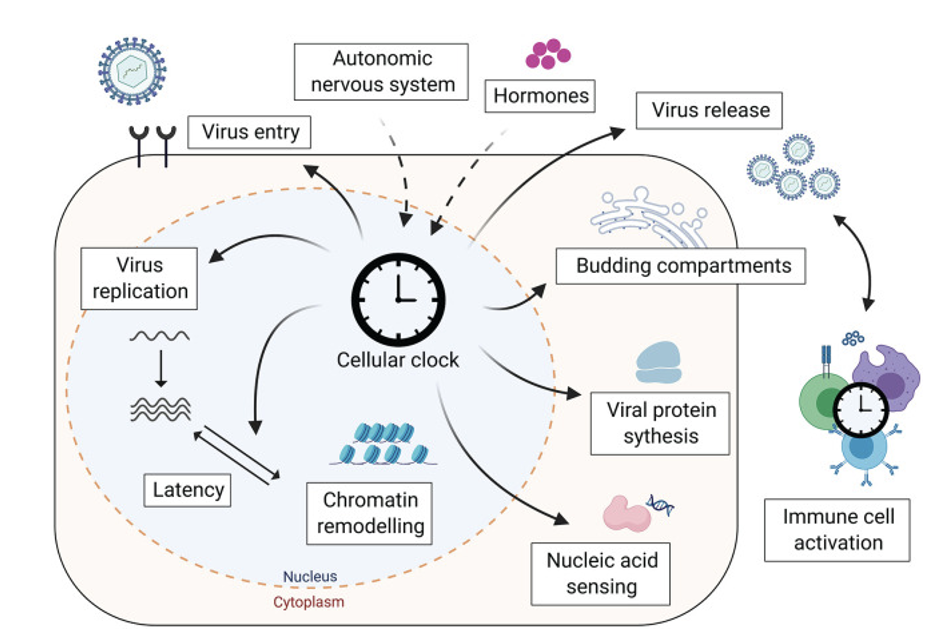
Cellular clocks affect the life cycle
of viruses at many levels
(below, shown for virus entering nucleus—DNA containing virus)
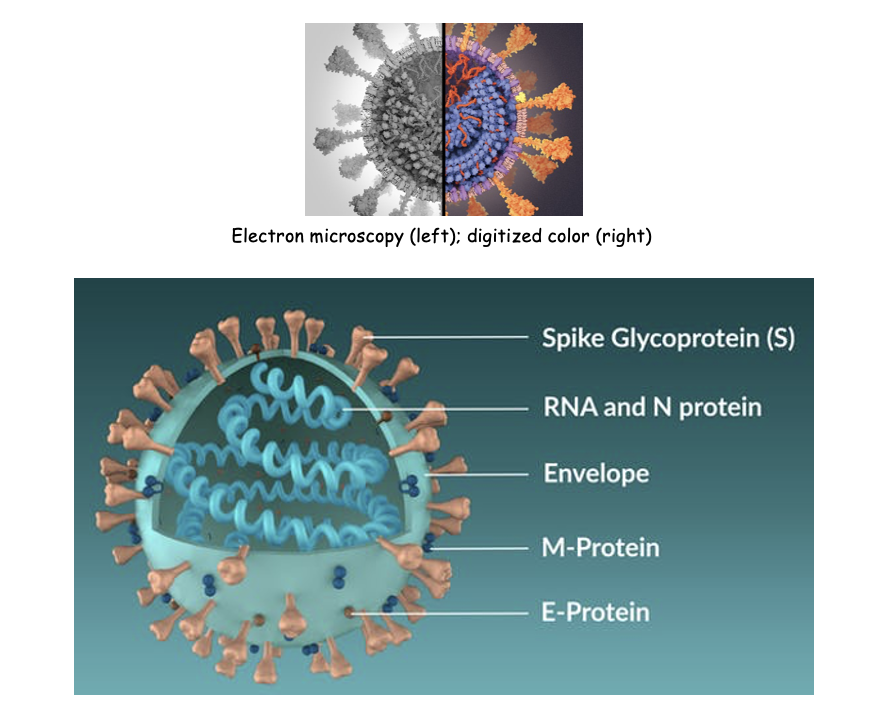
SARS-CoV-2
Electron microscopy (left); digitized color (right)
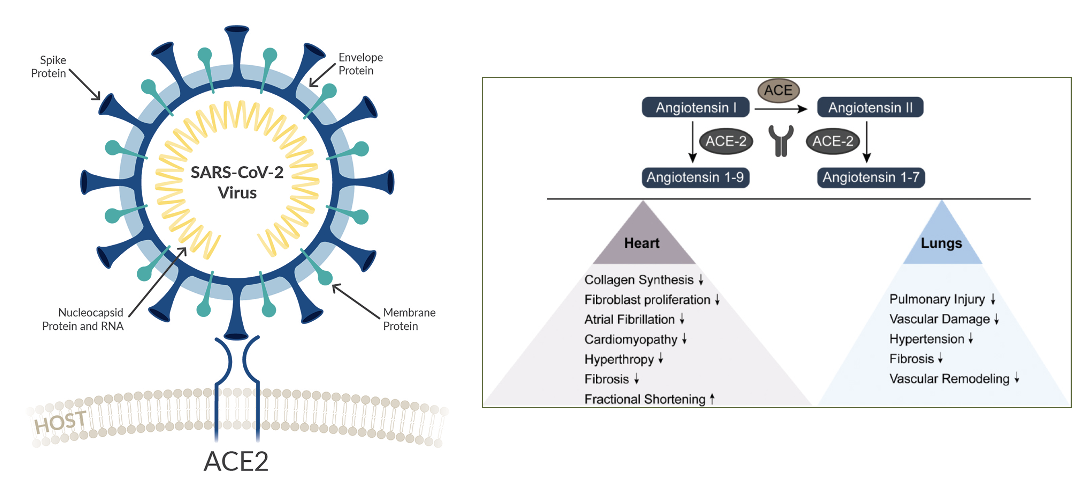
SARS-CoV-2 infects
cells by binding to the human ACE2 (angiotensin-converting enzyme2) found on the plasma membrane of many organs
ACE2 lowers blood pressure by converting angiotensin I/II (vasoconstrictor) to a vasodilator
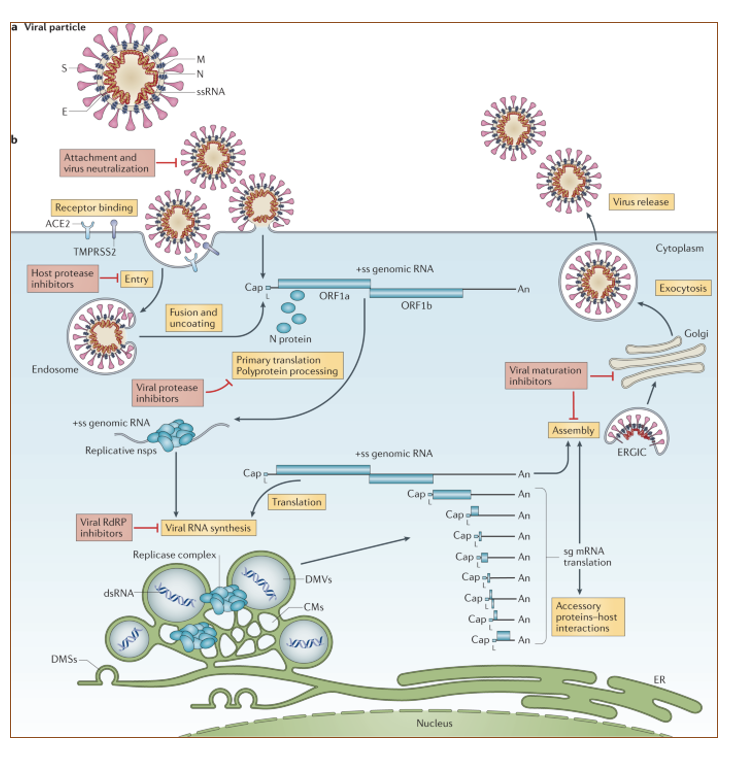
Overview of SARS-CoV-2 infection. There are different kinds of viruses depending on their genetic content, DNA and RNA viruses.
For RNA viruses they can be (+) or (-) single strand , double strand or retroviruses. SARS-Cov-2 is a (+) RNA virus.
In this case, the genetic material is the positive strand RNA that can be translated and used for replication and nested transcription.
Primary infection in lungs by SARS-CoV-2 can have secondary effects
effects on other organs via hyper-inflammation (cytokine spike)
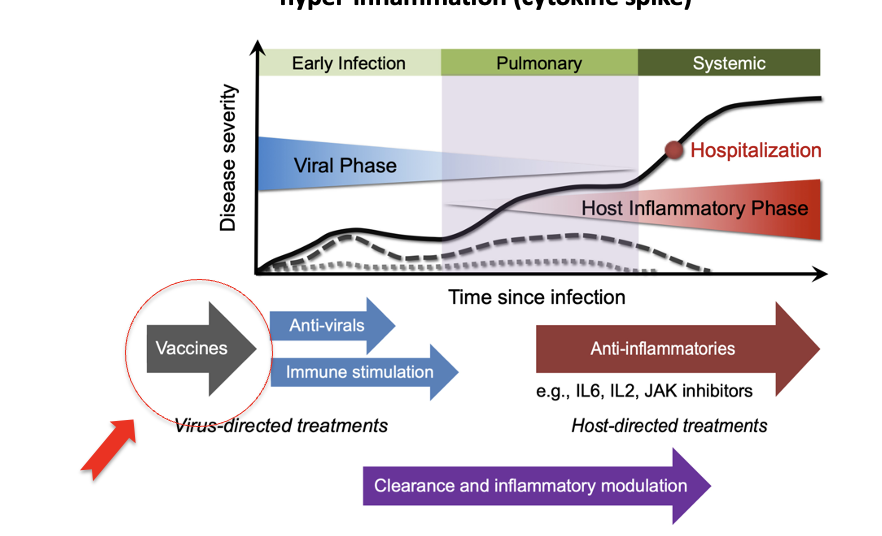
Treatment Options:
Treatment options:
Prior to infection: vaccines
Phase 1: Early phase; anti-virals
Phase 2: Pulmonary phase; liquid in lungs
Phase 3: Systemic hyper-inflammation; anti-inflammatories
A good vaccine elicits
production of memory B- and T-cells
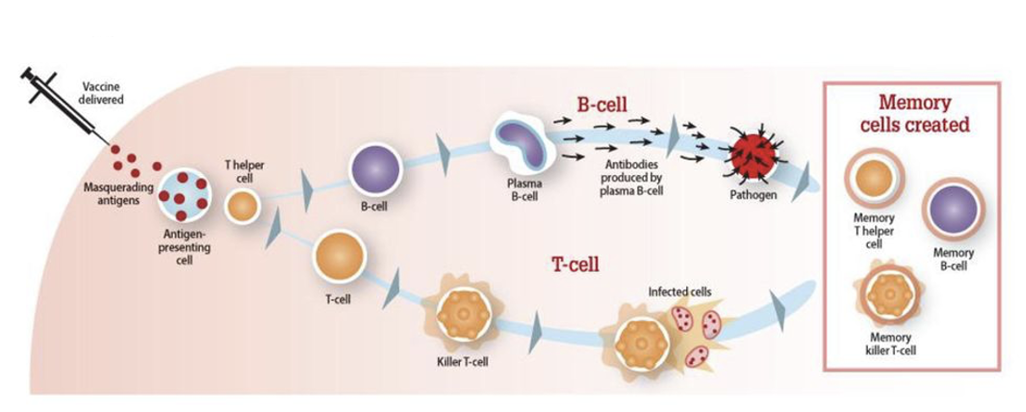
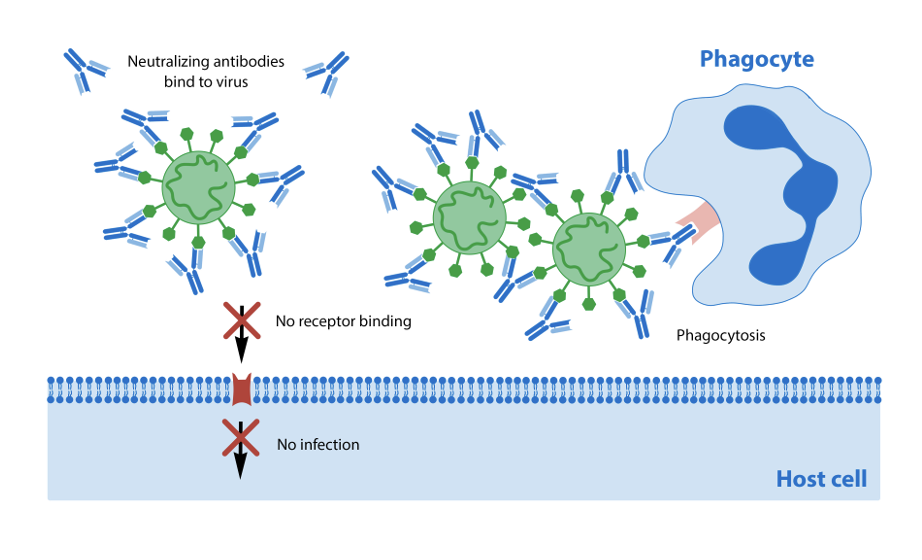
In particular, the best vaccines elicit the production of neutralizing antibodies
as they ”neutralize” viral entry into the cell. This is why so many vaccines target the viral proteins on the surface that trigger binding to the host cell
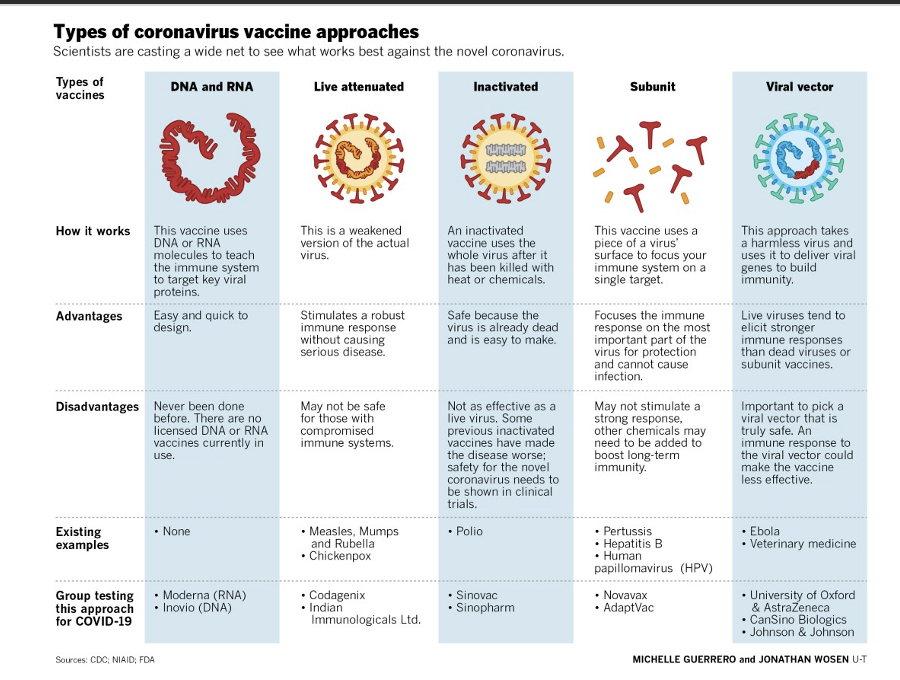
Numerous strategies have been used to generate vaccines against SARS-Cov-2,
from the more classic of using inactivated or attenuated viruses to new technologies using RNA-based methods. We will discuss the Moderna vaccine. There are advantages and disadvantages with each strategy
The rapid time-line for first-generation COVID-19 vaccines
vaccines is unprecedented
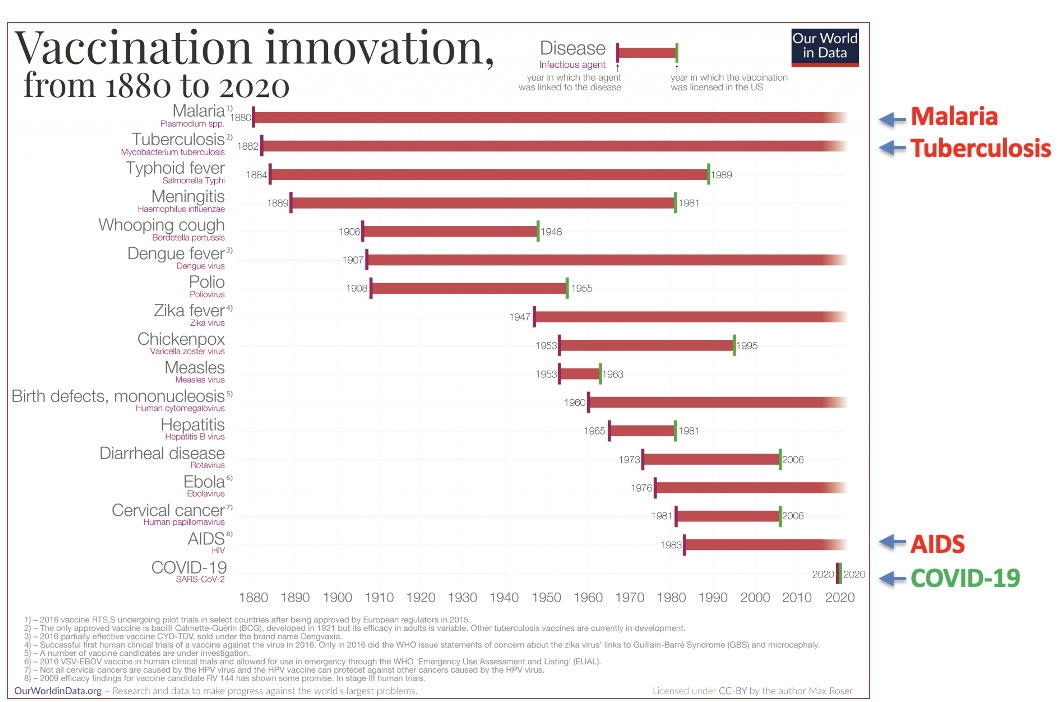
How did Moderna make such an effective and fast vaccine?
Moderna already had a long history and indeed was established to use mRNA as a new category of medicines.
The idea was simple, generate mRNA in vitro to a certain target; embed the mRNA inside a particle, inject particle, mRNA get taken up by host cell and gets translated like any other mRNA in the cell.
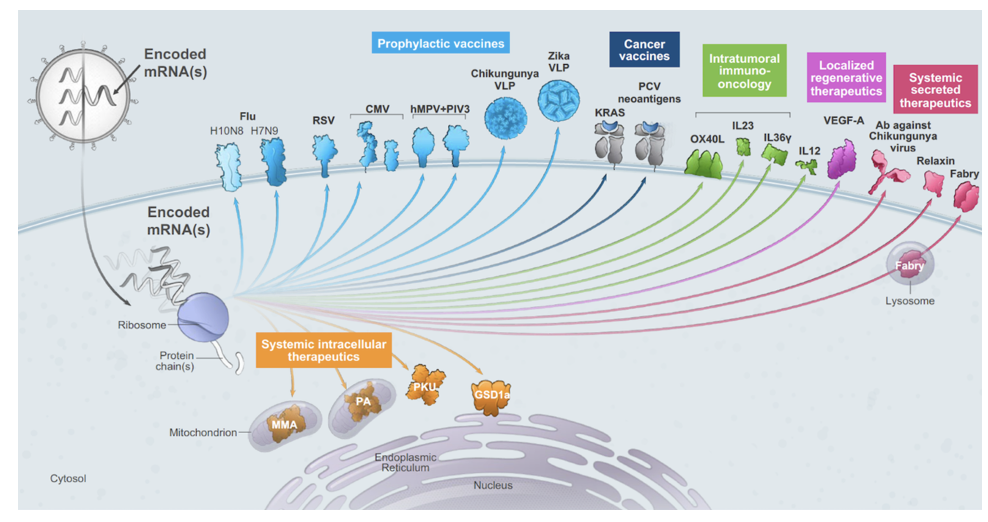
Advantages of mRNA
Can be rapidly produced in large quantities
DISATVANTAGES of mRNA:
Very unstable
Problem: mRNA is much more
unstable than DNA
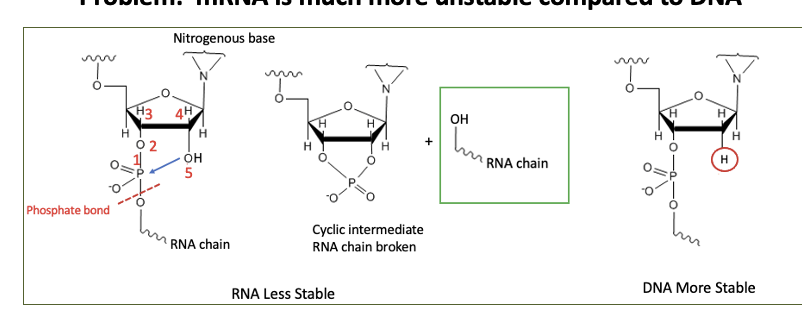
HOWEVER, can stabilize mRNA against immune detection
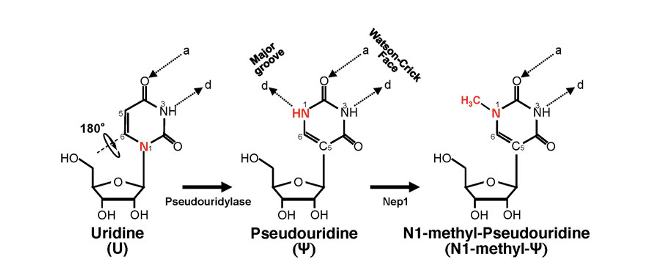
by modifying bases. 5-methylcytidine (m5C), and pseudouridine (ψ) are the most preferred base-pair alterations because they lower immunogenicity while increasing translation efficiency.
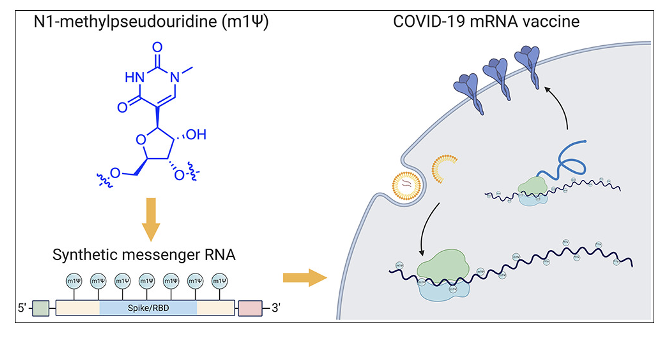
Other modifications to increase mRNA stability
NOTE: mRNA purification is required after in vitro transcription to eliminate immunogenicity. HPLC, anion exchange and affinity chromatography, are used in the purification process to remove truncated transcripts.
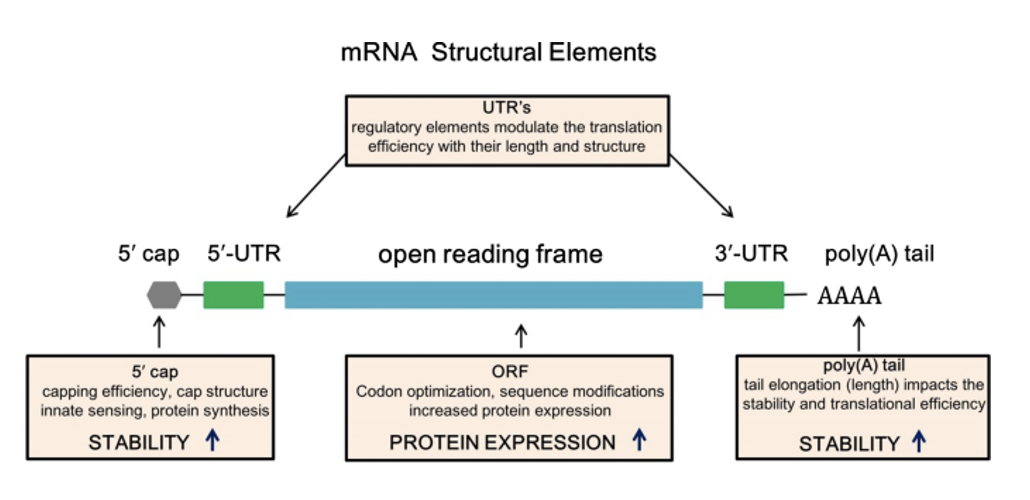
Major advances that allowed for mRNA vaccines (Moderna & Pfizer “secret sauces”)
ability to make synthetic mRNA in a form that is stable and will not trigger immune response
ability to package and deliver mRNA
Problem: how to make a synthetic mRNA stable and evade immune response
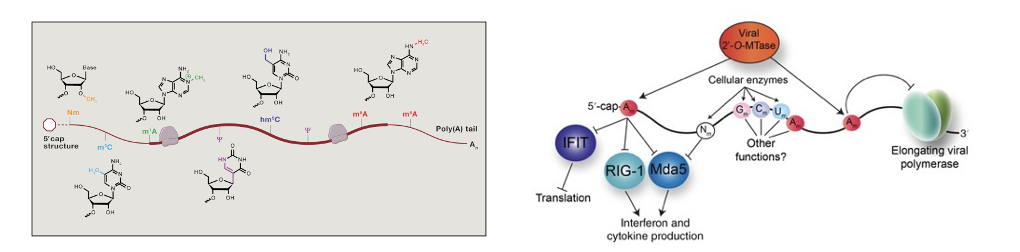
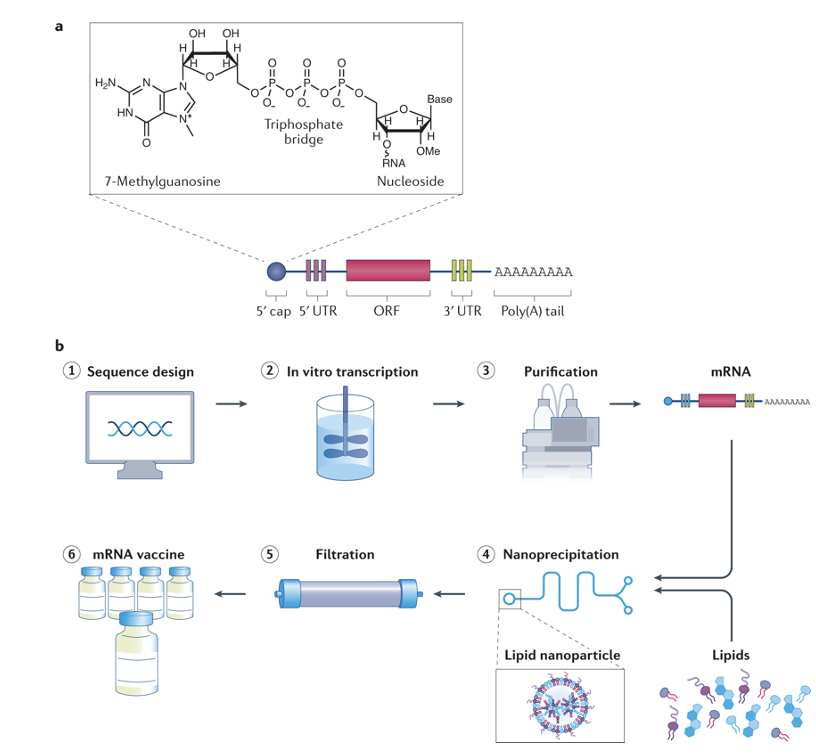
Natural mRNAs undergo a range of modifications, from the 5’ cap structure to m6A methylation (left).
Cap structure and polyA tail were known to stabilize mRNA and enhance translation. Additional modifications are also important for many aspects of mRNA stability and translation.
Without these modifications, the immune system can sense these RNAs as foreign and target them for destruction (right).
Many cellular AND viral RNAs are highly modified to avoid triggering immune response. In SARS-Cov-2 they encode enzymes to cap the mRNA and modify internal nucleotides (right). mRNAs without a cap structure are recognized by RIG to trigger an immune response (right).
Ground Breaking Discoveries
Katalin Kariko and Drew Weissman in a series of papers showed that including modified bases (such as m6A) helped evade immune detection.
Derrick Rossi picked up on these advances and applied it getting a new source of embryonic stem cells, adding mRNAs encoding key cell identity factors.
MODERNA
Usage of pseudo-uridine was a key
in the success of the Moderna and Pfizer vaccines
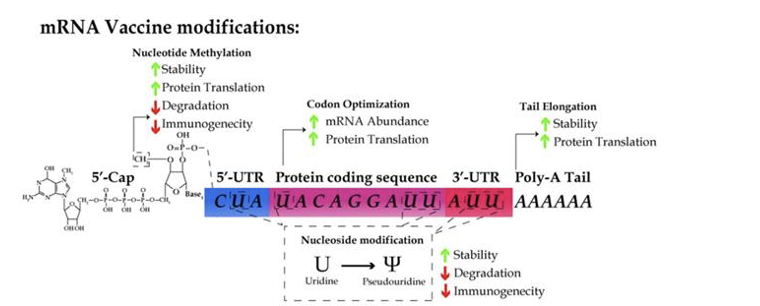
In vitro transcribed mRNAs for vaccines
vaccines are highly modified to increase translation, stability and decrease innate immunity.
A significant modification is using pseudouridine instead of uridine
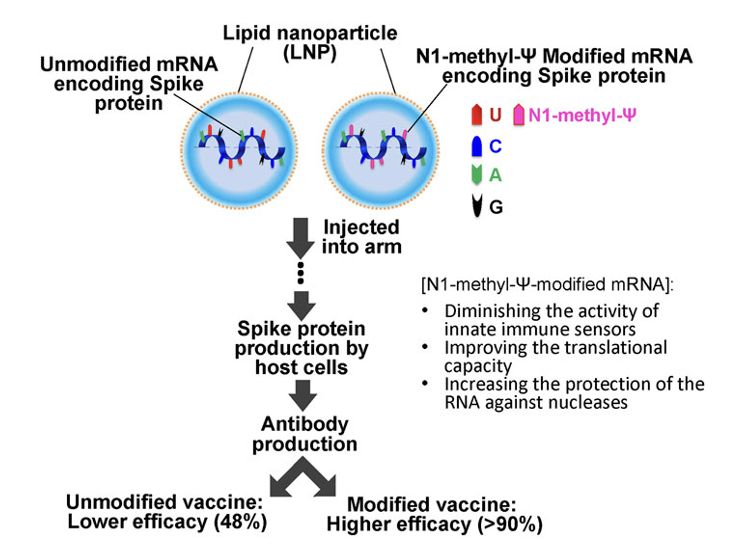
Pfizer-BioNTech and Moderna Therapeutics (comirnaty® and spikevax®, respectively) were the first ones presenting efficacies higher than 90%.
%. Both consisted of N1-methyl-pseudouridine-modified mRNA encoding the SARS-COVID-19 Spike protein and were delivered with a lipid nanoparticle (LNP) formulation. CVnCoV vaccine had the same sequence and delivery method but did not use modified nucleotides. Clinical trial results showed only 48% of efficacy against any severity of the disease
How to deliver the synthetic mRNA (the “secret” sauce)?
Problem. mRNA is three to four orders of magnitude larger than molecules that readily diffuse into cells; in addition, the dense negative charge of mRNA electrostatically repulses the anionic cell membrane, preventing its uptake.
Therefore, mRNA vaccines require a delivery vehicle that not only protects the nucleic acid from degradation but allows the mRNA to get into cells.
BioNTech/Pfizer’s and Moderna’s mRNA vaccines both use lipid nanoparticles as mRNA carriers. Much prior work had been done on generating lipid nanoparticles.
This knowledge has certainly contributed to the rapid development of COVID-19 mRNA vaccines.
There are many steps involved from mixing different factors and purifying particles
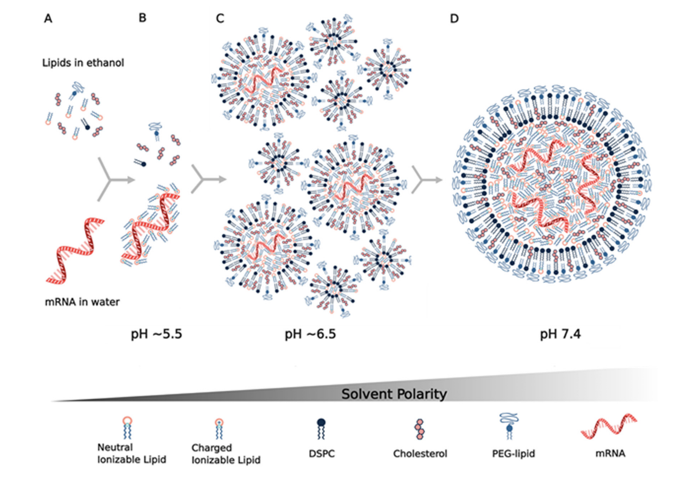
overview of mRNA vaccine production
Sequence Design
In vitro transcription
Purification
Nanoprecipitation
Filtration
mRNA vaccine
a rapid, one-step method used to produce polymer nanoparticles and nanocapsules with a narrow size distribution, high reproducibility, and good stability.
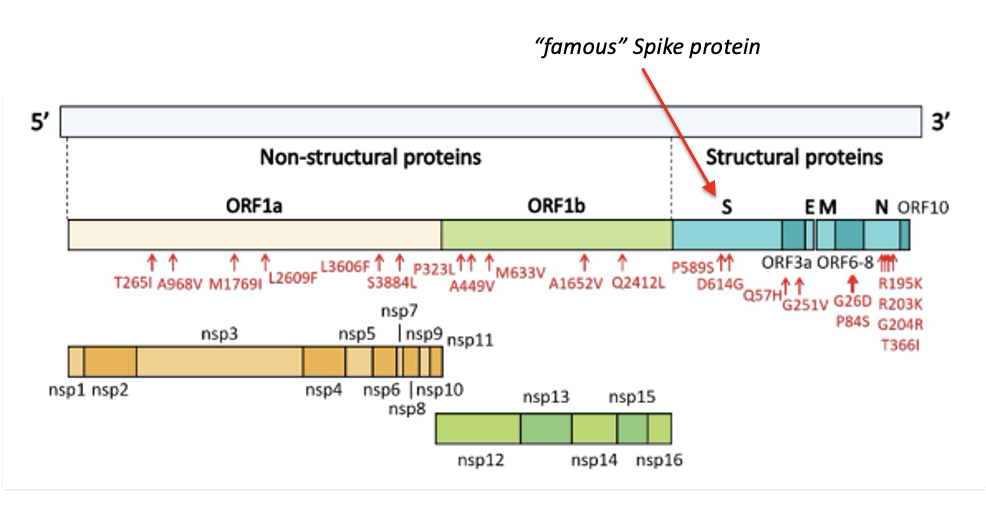
Final issue: what sequence to put in the mRNA vaccine?
Genomic organization of SARS-CoV2: Like many viruses, most of the protein products are made by cleavage of larger proteins (viruses have to pack in much information into a small genome)
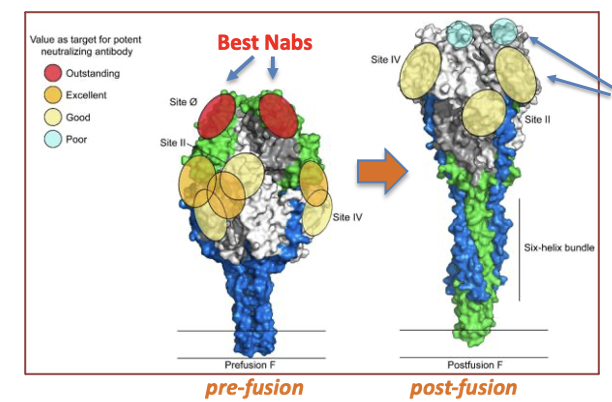
Prior work on respiratory syncytial virus (RSV) showed importance of pre-fusion/post-fusion conformational changes in producing a vaccine that elicits stronger production of neutralizing antibodies
Strong neutralizing antibodies (Nab) were found for RSV
These antibodies bound the top of the F protein, required for binding to cells
The structure of the F protein changes spontaneously and after binding to cells, adopting a post-fusion conformation
The pre-fusion to post-fusion conformational change destroys Nab recognition
Strategy to improve vaccine for RSV
change the structure of the F protein so that it is locked in the pre-fusion structure
They found that changing 2 amino acids to prolines, locked the structure
Since many of these viral proteins have similar overall structures, based on this prolines were introduced into other viral proteins, leading to stabilized pre-fusion structures (next slide)
Pre-fusion vs. Post-Fusion F protein
Strong Nabs lost after conformational change of F protein in RSV
In the post-fusion conformation, only weak Nabs are still able to bind, the strong Nabs no longer recognize the new conformation
Conformational change of SARS-CoV-2 spike protein
(from trimer to dimer)
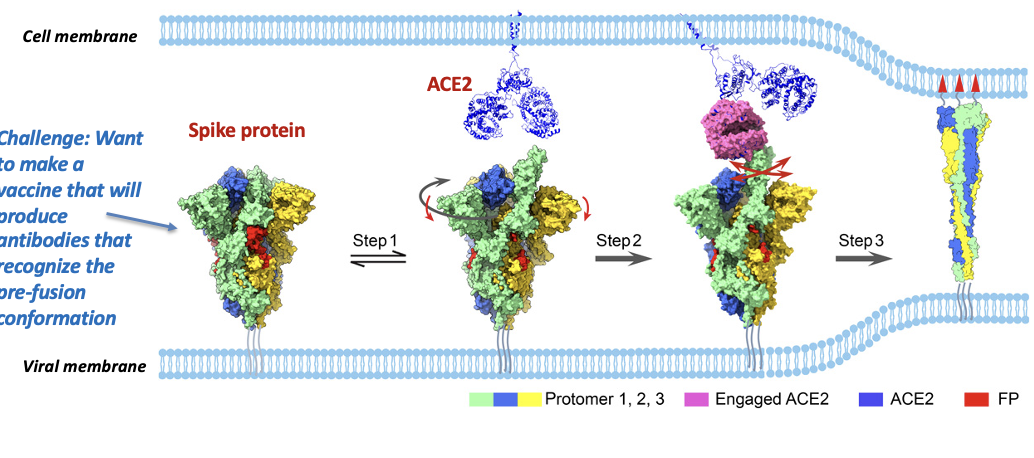
Challenge: Want to make a vaccine that will produce antibodies that recognize the pre-fusion conformation
Conformational transitions from the closed ground prefusion state
(with packed FP, in red) to the
transiently open state (step 1) with an untwisting motion (highlighted in dark gray arrow) associated with a downward movement of S1 (red arrow), from the
open state to the dynamic ACE2 engaged state (step 2), and
then all the way to the refolded postfusion state (step 3).
Thus, once the sequence of the SARS-Cov-2 was published,
Moderna and collaborators at NIH designed a 2 proline spike protein to test—they got its structure, put it into the Moderna RNA pipeline and did initial studies on neutralizing antibody responses.
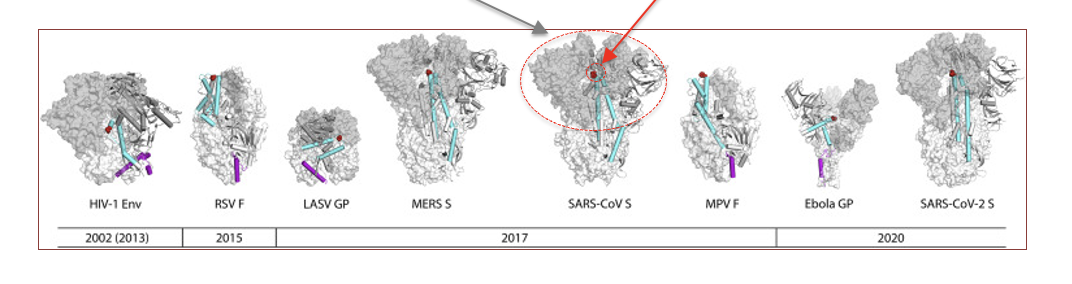
Examples of viral surface proteins where
changing 2 sites to prolines stabilized pre-fusion structures, enhancing vaccine efficacy by stimulating production of stronger neutralizing antibodies (prolines are red circles). Nabs antigenicity shown in grey.
Grey area shows where neutralizing antibodies bind
Switching 2 aa for prolines, locks many viral surface proteins involved in fusing to cell into the pre-fusion conformation. This allows raising antibodies that will act as neutralizing; i.e., prevent attachment to cell.
mRNA versus making the protein as a vaccine (hours to months difference)
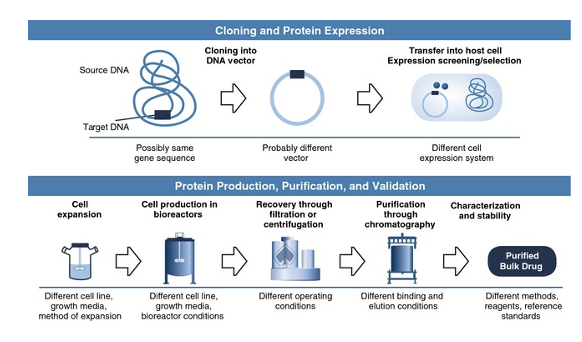
For large scale production of target proteins, they need to be synthesized in cells grown in large bioreactors.
Cells are required because protein synthesis requires ribosomes.
A major challenge is to purify the target protein from all the other proteins, etc in the cells. This is less of a problem for proteins that can be secreted into the culture media.
Also, if the protein needs post-translational modifications the host cell has to be appropriate for that. This entire process can take weeks to months. Thus, the process has to be optimized for each target protein.
Large scale production of a target mRNA is much
faster, uses less components and everything can be done in vitro.
A plasmid or PCR product that contains the coding region and an upstream promotes is basically mixed with 1 transcription factor and A, U, G, C ribonucleotides in a tube. The mix also contains the ability to add a 5’ cap structure and modified nucleotides (like m6A).
DNAse treatment gets rid of the DNA leaving the desired mRNA, which is easily purified away from the transcription factor and unused nucleotides in one step.
The PURIFIED mRNA can then be packaged into lipid nanoparticles for delivery. Since all the steps are the same irrespective of DNA sequence there is little issue with optimization.
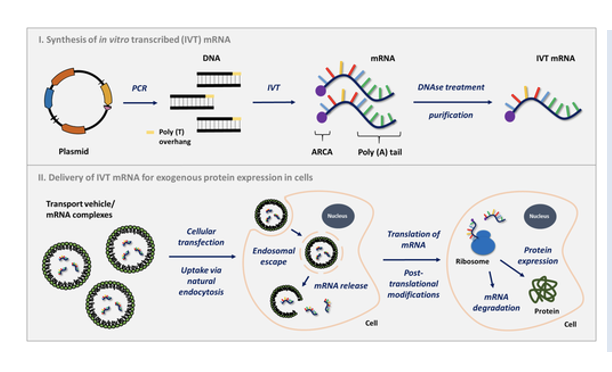
Safety: mRNA vaccines
cannot produce DNA or integrate into the genome
Can’t make more mRNA lipid nanoparticles
mRNA and protein eventually degrade
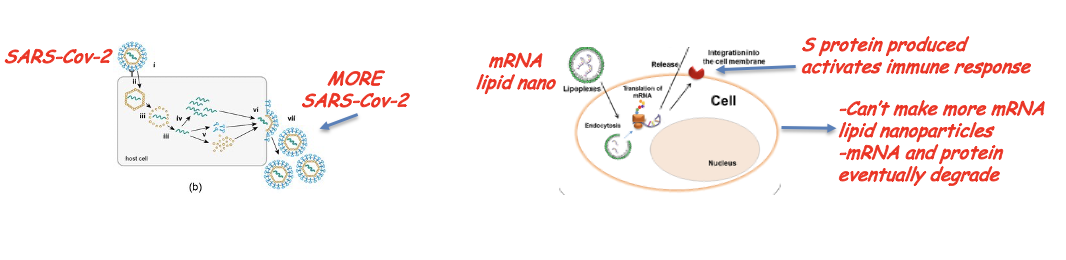
why is it impossible for the mRNA vaccine to be integrated into the genome
There is no reverse transcriptase or integrase in the vaccine
Even with all the modifications on the mRNA vaccine they only last 10-14 days (no protein is produced from them after that time)
Because the mRNA vaccine is injected into the arm muscle and stays local, it is almost impossible for it to get past the blood brain barrier EVEN THOUGH, SARS-Cov-2 as it is replicating and producing more viral particles has been shown to cross the BBB.
This does not mean there are no effects of SARS-COV2 virus
Increased risk of myocarditis, especially in adolescent and young men, especially after second dose (small pox vaccine also had this issue). But apparently, the risk form COVID for myocarditis is some 5x higher compared to vaccine BUT this might not be the case for very young (e.g., 2-5 yrs).
Increased risk of cerebral venous thrombosis—rare blood clot--, especially in women (but again, more risk of CVT from SARS-CoV2 infection). Bigger problem with AstraZeneca. Uses Adenovirus to deliver DNA that is transcribed in nucleus, which could lead to aberrant splicing and misfolded spike proteins!
Possible scenario for vaccine to affect ACE2 receptor:
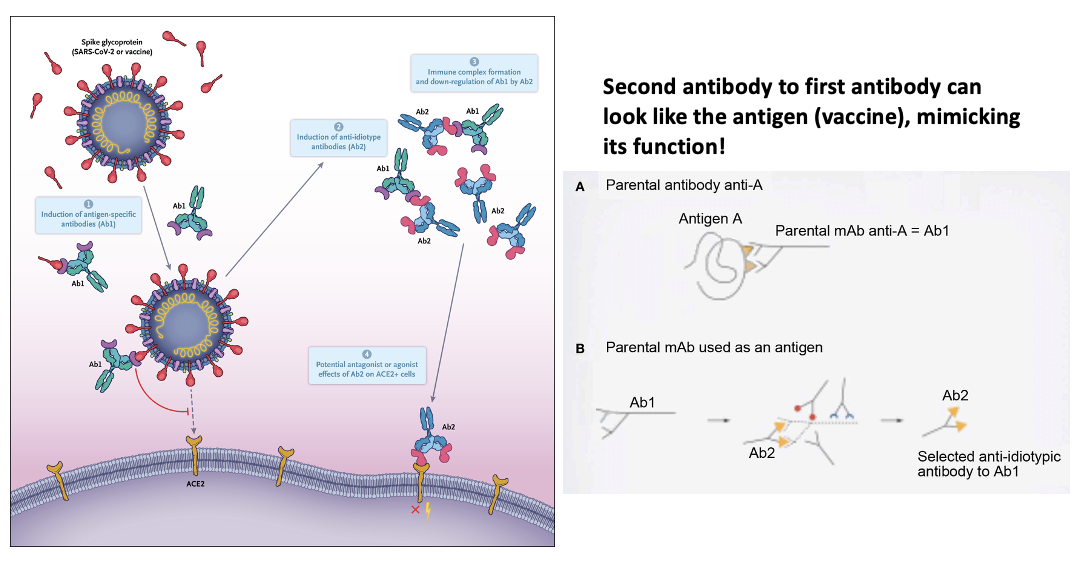
Anti-idiotype Antibodies and SARS-CoV-2
The idiotype portions of Ab1 that bind and neutralize the spike protein have distinctive sequences in complementarity-determining region 3 (CDR3), and those antibody-binding regions can themselves elicit antibody responses called anti-idiotype (Ab2) antibodies as a means of down-regulation.
Second antibody to first antibody can look like the antigen (vaccine), mimicking its function!
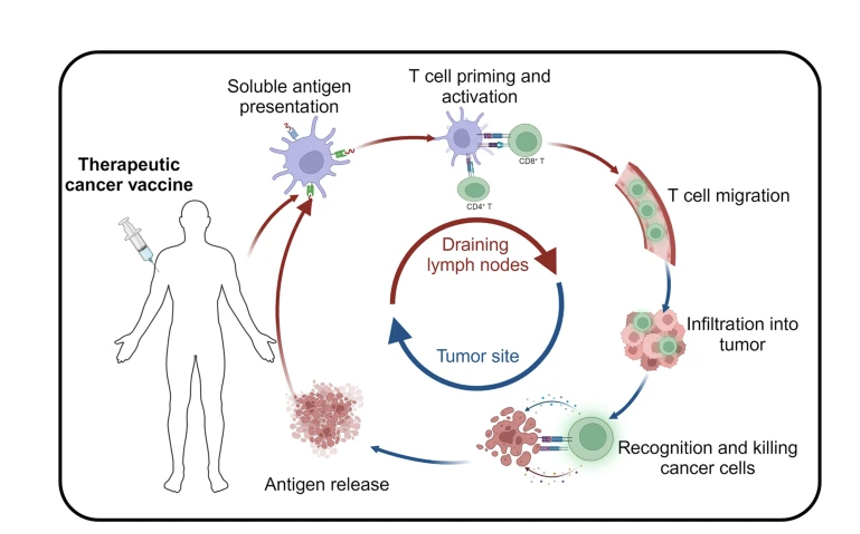
mRNA vaccines for fighting cancer?
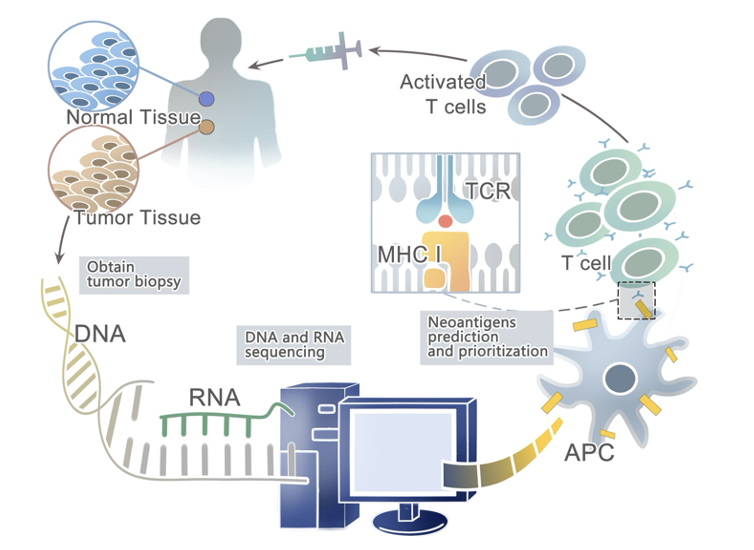
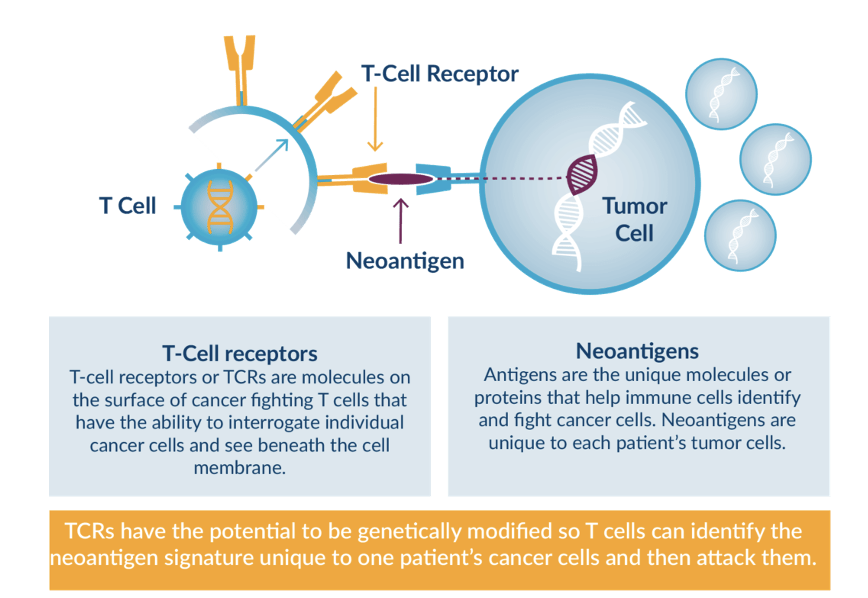
The goal of a cancer vaccine is to safely expose the patient’s immune system to tumor-specific antigens (neoantigens) to induce cytotoxic T-cell activation to enable the immune system to elicit a more effective antitumor response.
Cycles of tumor neoantigen presentation:
The mechanism of cancer vaccine in vivo.
After the tumor antigens migrate into the body in different forms, they are phagocytosed, intracellularly expressed, and efficiently processed by specialized antigen-presenting cells (APCs).
The major histocompatibility complex (MHC) of dendritic cells presents antigens to their surface, and the MHC complexes activate antigen-specific T-cells by binding to T-cell receptors (TCR) on the surface of T-cells, therefore safely, persistently, and specifically destroying tumor cells and inhibiting tumor growth
A framework for the identification and prioritization of neoantigens from computational analysis for an individual cancer sample
Recently, technological innovations have made neoantigen discovery possible in a variety of malignancies, thus providing an impetus to develop novel immunotherapies that selectively enhance T cell reactivity for the destruction of cancer cells while leaving normal tissues unharmed