Materials Vocabulary (KCC Yr 9 Science 2025)
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Element
A pure substance made of only one kind of atom. Elements are the building blocks of other substances and can be found on the periodic table.
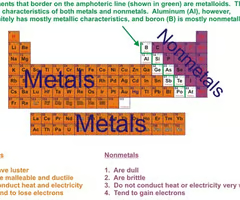
Metals
Elements that are good conductors of electric current and heat. On the left of the periodic table.
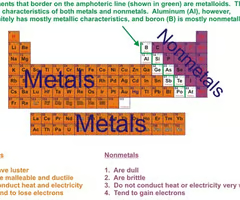
Non-metals
elements that are usually dull in appearance, poor conductors of heat and electricity, gases at room temperature. On the right of the periodic table.
Metalloids
Elements that have properties of both metals and nonmetals. Between the metals and non-metals on the periodic table.
Examples of substances that are elements
Gold, pure water, pure oxygen.
Examples of metals
Iron, copper, sodium, magnesium.
Examples of non-metals
Oxygen, carbon, chlorine
Examples of metalloids
boron
Monatomic elements
Elements that exist in nature as single atoms. Eg. helium and argon.
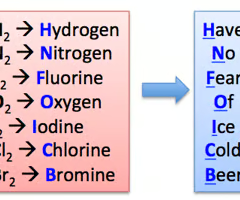
Diatomic elements
Elements that exist in nature as two atoms of the same bonded together. Eg. chlorine (Cl2) and nitrogen (N2).
Lattice Elements
Elements that arrange themselves in large, complex crystal structures. Eg. diamond.
Compounds
Substances that contain atoms of two or more different elements chemically bonded in a fixed ratio.
Mixtures
Two or more pure substances (either elements or compounds) combined but not chemically bonded together. Alloys are mixtures of a metal and other metals or non-metals.
Pure substances
Substances that are either elements and compounds.
Impure substances
Substanes that are homeogenous and heterogenous mixtures.
Solids
A state of matter in which the object has a fixed shape and volume.
Liquids
A state of matter in which the object has a fixed volume but a variable shape.
Gas
A state of matter ion which the object has a variable shape and variable volume.
Diffusion
The process of gases expanding to fill their container.
Melting
State change from solid to liquid
Evaporation
State change from liquid to gas.
Condensation
State change from gas to liquid.
Freezing/solidification
State change from liqiud to solid.
Sublimation
State change from solid to gas.
Deposition
State change from gas to solid.
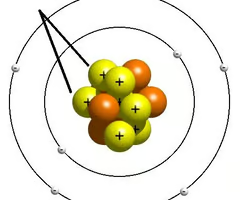
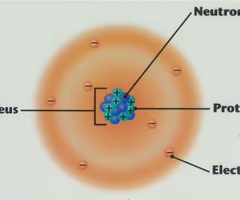
Proton
positively charge subatomic particle found in the nucleus.
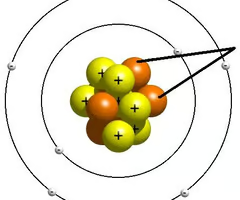
Neutron
neutral subatomic particle found in the nucleus.
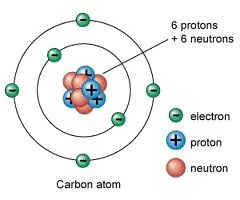
Electron
negatively charged subatomic partcile found orbiting the nucleus

Atomic number
the number of protons in an atom. Found on the periodic table.
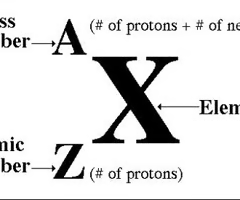
Mass number
the sum of the number of neutrons and protons in an atomic nucleus.
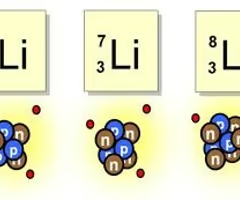
Isotope
A form of an element that has the same number of protons but a different number of neutrons
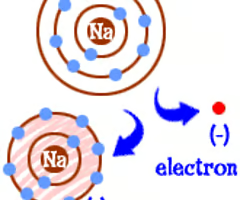
Ion
A form of an atom that has a positive or negative charge due to electrons and protons not being equal
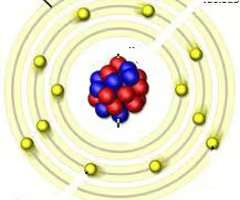
Valence shell
The outermost electron shell in an element
Electron configuration
the arrangement of electrons in an atom
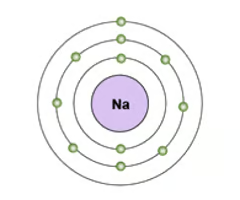
Electron shell diagram
Diagram showing electrons in their shells around the nucleus of an atom.
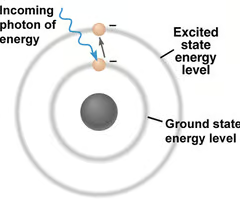
Ground state of an atom
The lowest possible energy state of the electrons in an atom.
Annealing
Heating a metal to a specified temperature where recrystallisation occurs and allowing it to cool down slowly to room temperature
Quenching
Heating a metal to a specified temperature where recrystallisation occurred and quickly cooling it down in water or oil bath
Tempering
Heating a quenched metal to below recrystallisation temperature and allowing it to cool down in the air
Metals
Dense, ductile, malleable substances with high melting and boiling points that are generally good thermal and electrical conductors
Ductile
A term used to describe a material that can be pulled out into a long wire.
Malleable
capable of being beaten into shape
Lustrous
Shiny
Metal Alloys
Mixture of at least one metal and one other substance to improve its properties.
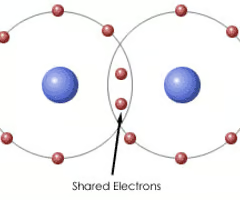
Covalent bond
A bond between non-metal elements that involves the sharing of electrons to gain a full outer shell.
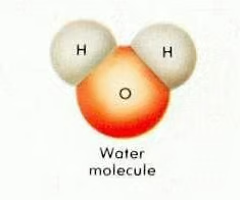
Covalent molecular compounds
A compound composed of two or more nonmetal elements bonded in individual sub-units (e.g. water, carbon dioxide)
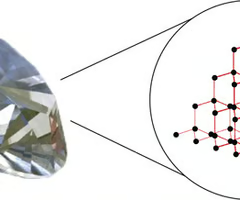
Covalent network compounds
Non-metal elements or metalloids covalently bonded in a huge 3-D network (e.g. diamond, sulfur, silicon)
Allotrope
Different physics forms of the same element (e.g. graphite, charcoal and diamond)
Properties of covalent compounds
These vary but, most have low melting and boiling points (but there are lots of exceptions), most are poor conductors of electricity, cakey and crumbly (if solid) and dull with no shine or lustre.
Properties of ionic compounds
Brittle, hard with very high melting and boiling points. Conduct electricity when dissolved in water but not as solids.
Examples of ionic compounds
Sodium chloride (NaCl), copper sulfate (CuSO4), potassium nitrate (KNO3).
Examples of covalent compounds
Water (H2O), carbon dioxide (CO2), sulfur (S8).