5. Psychometry & Convective drying
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Food preservation by drying
Drying is a method of food preservation that inhibits the growth of bacteria, yeasts, and mould through the removal of water
Why not sun drying?
Not safe: micro-organisms may grow during slow drying
Unpredictable; big losses
Not realistic in many temperate regions
Solutions is the use of hot air
How much of the total industrial energy consumption goes to drying?
15-20% of total industrial energy consumption
Relative humidity formula

Properties of vapor and air
Vapor and dry air mix well
Hot air can contain more water vapor
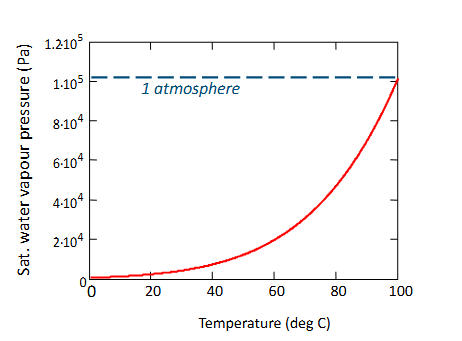
What does a sorption isotherm tell you?
It gives the moisture content/change in mass on the y axis needed to reach a certain water activity
When does hysteresis occur?
Hysteresis is the difference between adsorption and desorption isotherm
The effect does not occur in solutions; only in porous products, such as foods, hysteresis occurs
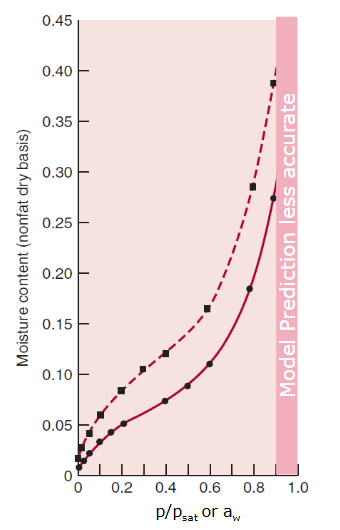
Absolute humidity (need to know!)
molar fraction of moisture in air
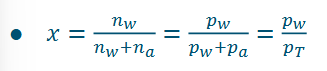
Humidity ratio (or moisture content) (need to know!!)
i.e. kg of water per kg dry air
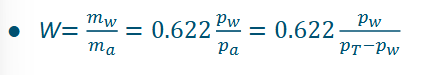
Relative humidity definition (need to know!!)
Relative humidity is the relative amount of vapor in air compared to the maximum saturation at a specific temperature

Dry and wet bulb temperature (need to know!)
Dry bulb: Basically the normal air temperature
Wet bulb: Temperature measured with a thermometer wrapped with a truly wet cloth exposed to the air flow
Wet bulb temperature is the temperature that a wet surface will take upon fast evaporation of the water
The wet bulb temperature is:
Not dependent on air flow rate
Twet bulb«Tdry bulb
Dependent on RH
Why are at 100% RH the wet and dry bulb temperatures equal?
At 100% Relative Humidity (RH), the air is saturated, meaning it is at its maximum capacity to absorb moisture at its current temperature. Because the air is already saturated, the wet and dry bulb temperatures become identical
Dew point (need to know)
The dew point indicates the temperature at which water vapor starts to condense when cooling air
Adiabatic saturation
This is a process where air contacts a wet product and takes up moisture without exchanging heat with the environment.
In this state, the air's sensible heat is converted into latent heat to fuel evaporation, causing the air to cool down as its humidity increases.
The air will cool down if you contact hot air with a wet product adiabatically
Adiabatic = without heat loss
Of the air:
The sensible heat decreases
The latent heat increases
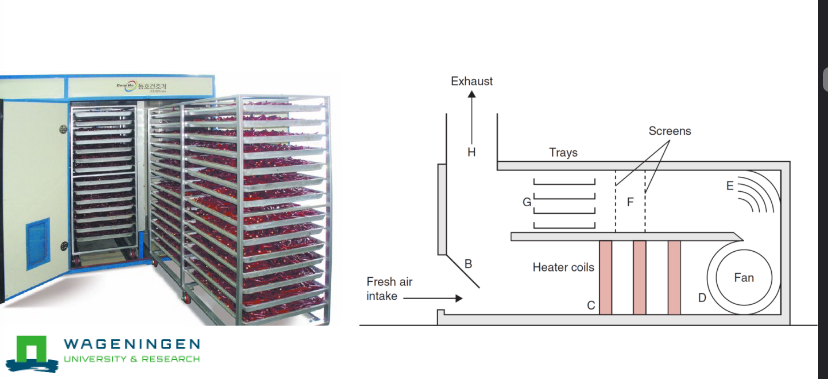
Tray dryer
Allows for larger products
Longer drying times
e.g. raisins, dates, plums, figs, apricots
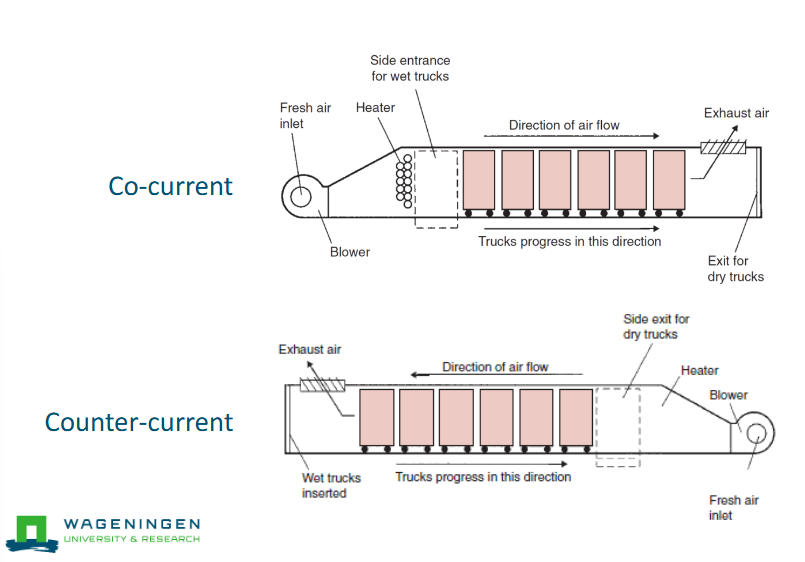
Continuous tray dryer
Continuous throughput
Carts enter through a sluice, and exit through another one
More efficient use of drying air
Co-current and counter-current drying
Counter current is more efficient but also heats up more and therefore can potentially damage the product.
Spray dryer
For liquid products only
Product is dispersed into very small droplets, in hot, dry air
Drying time < 1-2s
Droplet moisture evaporates, air cools down
e.g. milk powder, whey protein isolate, cream; lactose
Fluidized bed drying
Moist particles are bubbling in the continuous upward flow of air
Used for drying with longer residence time (falling rate period)
Used for agglomeration into larger particles (granulation)
Used for cooling
Feed = ‘moist’ granular product
Can be batch or continuous
Three zones in continuous fluidized bed drying
Agglomeration
Drying
Cooling
Agglomeration
Done by fluidized bed drying
Individual (~spherical) particles are glued to each other
Agglomerates/granules are created
Porous, powder density is low
Excellent flowability
Sometimes a compaction step may be added
Dissolution behavior of powders (dispersion)
Because powder materials are typically soluble and hygroscopic, they suck up liquid into their internal pores through capillary action, a process called wicking
Fast swelling/dissolution can lead to lumps, e.g. in hot water
Addition of lecithin controls wettability and dispersion
Dissolution behavior of powders (disintegration)
As water wicks into the pores, mechanical forces (like stirring) act on the powder bed to break it into many small pieces.
Agglomerated powders excel here because they allow water to separate clusters before they become a solid mass
dissolution behavior of powders (dissolution)
This is the final stage where primary particles actually dissolve.
Interestingly, increasing stirring speed does not necessarily accelerate this stage once particles are suspended, as they are too small to be affected by convective turbulence (eddies).
The rate of dissolution is primarily driven by the specific surface area; therefore, irregular, cracked, or highly porous particles dissolve the fastest.
influence of spray drying on solubility of milk powder
Insolubility is hypothesized to be caused by protein aggregation, which occurs when milk components are exposed to high temperatures while still within a specific moisture rang
Insoluble particles are only formed between 10 to 30 w/w% moisture and the rate depends on temperature
If the particle is still containing significant internal moisture while its temperature is rising into this high range, the proteins (particularly caseins and whey proteins) can denature and aggregate. These aggregated proteins form a dense, hydrophobic network that water cannot easily penetrate or break apart during later reconstitution
Stages of droplet drying kinetics
First stage: ideal shrinkage till locking point
Second stage: collapse of droplet → powder morphology development
Drying of moist solid products such as vegetables can be characterized in a similar way
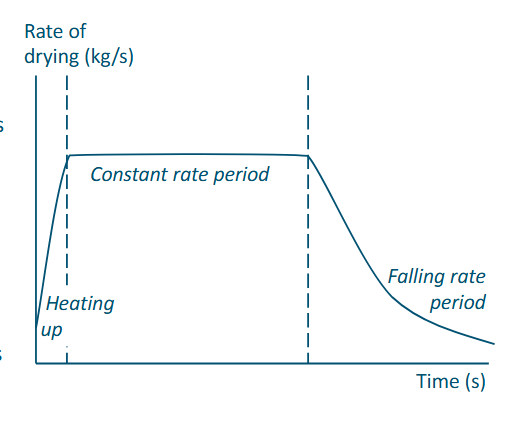
Drying rate curve
Two periods:
Constant rate period
Particle is still very wet
Evaporation of moisture is externally limiting
Temperature low
The ratio of heat transfer to mass transfer is constan
Falling rate period
Particle is getting dry at the surface
Diffusion inside product is limiting
Temperature higher
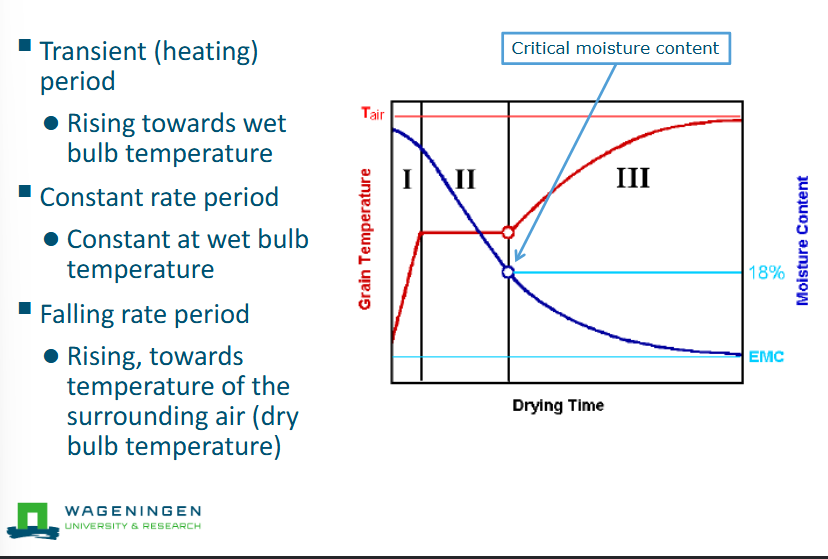
Change in temperature during drying
Red line = product, plateau = wet bulb temperature
When temperature changes, does relative humidity also change?
Yes but the moisture content stays the same
What machines can or cannot be used when preventing protein aggregation?
Spray drying: This is generally a milder process because the high surface area of small droplets allows for rapid drying at the wet bulb temperature. However, if the air is too hot or the particles reside too long in the dryer, the "critical moisture range" overlap with high heat leads to the formation of insoluble components
Drum Drying: This method is much more aggressive because the milk slurry is in direct contact with a metal wall heated to 120–160 °C. This intense heat causes widespread protein denaturation and gives the powder a "cooked" or caramelized flavor, resulting in poorer solubility compared to spray-dried version
What is HL?
HL = Hv - Hc
What is PT
Total pressure of the moist air (atmospheric pressure)