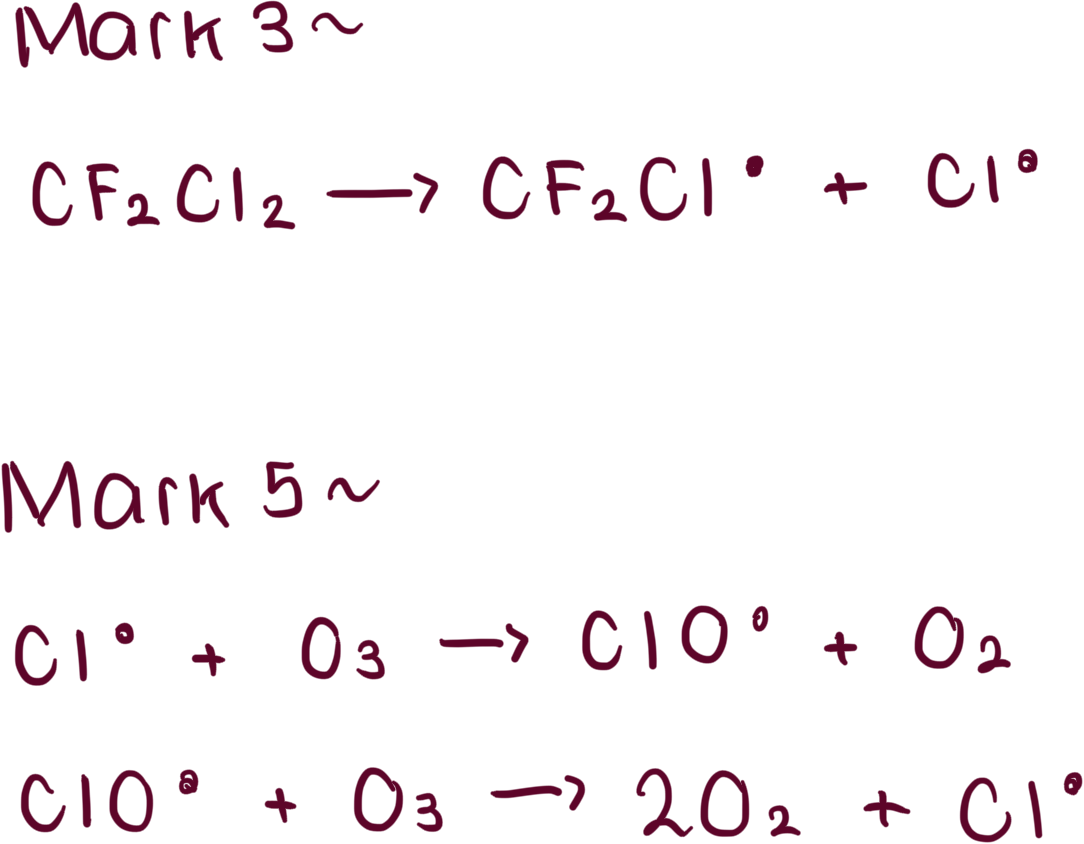chapter 5 - haloalkanes environmental effects
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
what is a CFC?
a chlorofluorocarbon molecule
they only contain carbon, chlorine and fluorine atoms
what is ozone?
has the formula O3
it is less stable than O2
ozone is formed from oxygen and UV light
it is present in low concentrations
the highest concentrations are in the ozone layer of the earth’s atmosphere
what is photolysis?
means broken down by light
CFCs undergo photolysis in the atmosphere
what does photolysis produce?
chlorine radicals
chlorine radicals react with ozone
what type of mechanism is the destruction of ozone?
free radical substitution mechanism
initiation, propagation and termination steps of the destruction of ozone?

overall mechanism reaction for destruction of ozone?

what are CFCs used in?
aerosols
as coolants in fridges
solvents in the industry
however, scientists collected evidence in the 1970s that they were damaging the ozone layer
this led to bans, and the development of safer alternatives like hydrofluorocarbons (HFCs) and hydrocarbons
what forms the holes in the ozone layer?
CFCs in the upper atmosphere absorb UV radiation and split to form chlorine free radicals
these free radicals catalyse the destruction of ozone, destroying ozone molecules
then they are regenerated to destroy more ozone
how many ozone molecules can one atom of chlorine destroy before it forms a stable compound?
10,000
why are holes in the ozone layer bad?
because they allow more harmful radiation to reach the earth
what else can destroy ozone?
NO (nitrogen monoxide) free radicals from nitrogen oxides
what are nitrogen oxides produced from?
car and aircraft engines
thunderstorms
question: explain how CFCs cause a depletion in the ozone layer and include relevant equations in your answer (5)
M1- chlorine free radicals are formed in the upper atmosphere
M2- when energy from ultra-violet radiation causes the C-Cl bonds in CFCs to break
M4- the chlorine free radical atoms catalyse the decomposition of ozone due to these reactions because they are regenerated