Module 22: Acids, Bases, and Salts
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Acid
a substance that produces hydrogen ions in a water solution
hydronium ions
positively charged and have the formula H3O+
indicator
A compound that changes color in the presence of an acid or a base

Common Acids
hydrochloric acid, nitric acid, sulfuric acid, acetic acid
Bases
Compounds that reduce the concentration of hydrogen ions in a solution.
Hydroxide ions
negative ions of OH--; can be produced when bases are dissolved in solution
properties of bases
taste bitter , slippery to the touch, can be poisonous & corrosive, release OH- ions in water
property of acids
tasting sour, dissolving in metal, and turning blue litmus paper red
Common bases
Household cleaners, baking soda, plaster, soft soaps, laundry detergent, bleach
strong acid
an acid that ionizes completely in water
weak acid
an acid that is only slightly ionized in water
Strong base
A base that dissociates completely into ions in solution.
weak base
A base that does not dissociate completely into ions in solution.
Strength vs concentration
Strength is a measure of the degree of dissociation of an acid or base in solution, independent of its concentration.
Concentration refers to the amount of solute per quantity of solution.
Strong vs weak describes
Strength
Diluted vs. Concentrated describes
Concentration
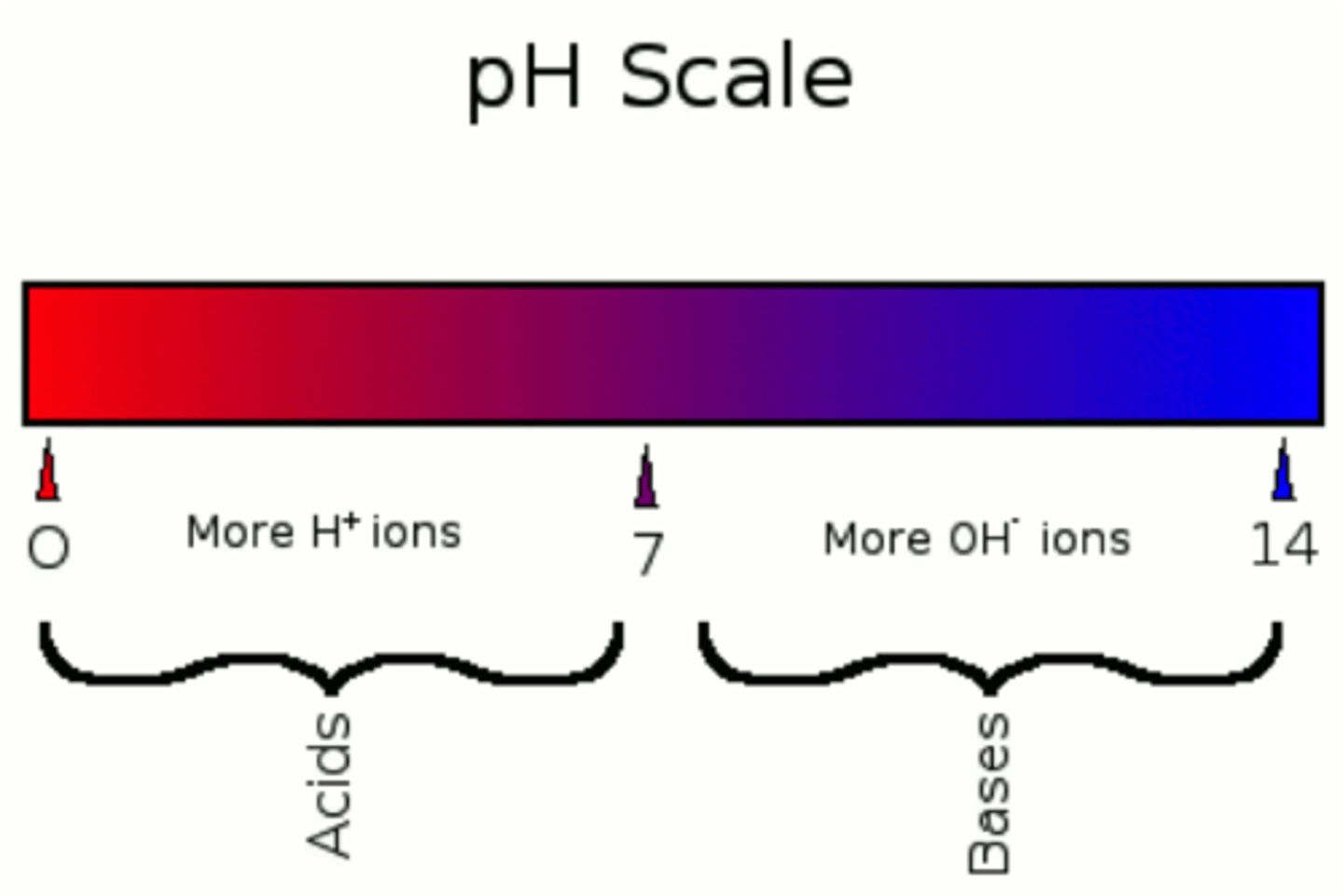
pH of a solution
A measure of the concentration of hydrogen ions it contains
Buffers
solution containing ions that react with acids or bases to minimize their effects on pH
Neutralization
a chemical reaction in which and acid and a base form a salt in water
salt
An ionic compound made from the neutralization of an acid with a base.
Titration
A solution of known concentration is used to determine the concentration of another solution.

soap
salt of a fatty acid