P2: Quantum Mechanics
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
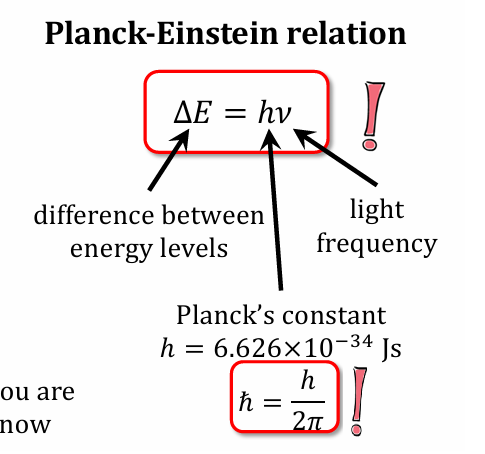
Give the formula related to change in E
and h dash
and a approximation to plank’s constant
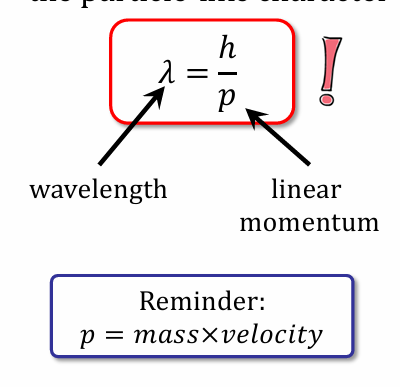
Give de Broglie relation and momentum formula

c=v x lambda, v (Hz/s-1) frequency

What is i and i²
what is a complex number z and its conjugate
i= square root (-1)
i²= (-1)
z = x+iy
its conjugate z* = x - iy
What is square modulus
and absolute value (modulus)
I𝑧I² =𝑧∗𝑧=𝑧𝑧∗ = (𝑥 +𝑖𝑦 )(x −𝑖𝑦)= 𝑥² +𝑦²
I𝑧I = square root(𝑧∗𝑧) = square root(𝑥² +𝑦²)
The equation 𝑥 +4 = 0
Has two solutions 𝑥 = 2𝑖 and 𝑥 = −2𝑖
Born Interpretation of the wavefunction?

What is Probability of finding a particle in region 0 ≤𝑥≤ 1 in a 1D box

What is the Probability of finding a particle in volume ?

What is the Probability of finding a particle anywhere in space?


Name all parts in the pic and
Let’s consider operator 𝐴 = dx/dy , and a function 𝑓 (𝑥) =12e ^-3x
f(x) is eigenfunction of 𝐴 with eigenvalue -3
𝑑𝑓/𝑑𝑥 =12× (−3)𝑒^−3× =(-3)f
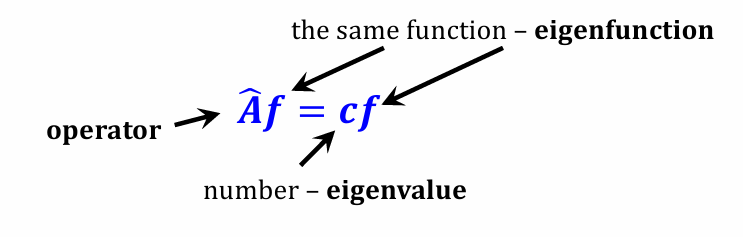
What is The time-independent Schrödinger equation is an eigenproblem of the energy operator?
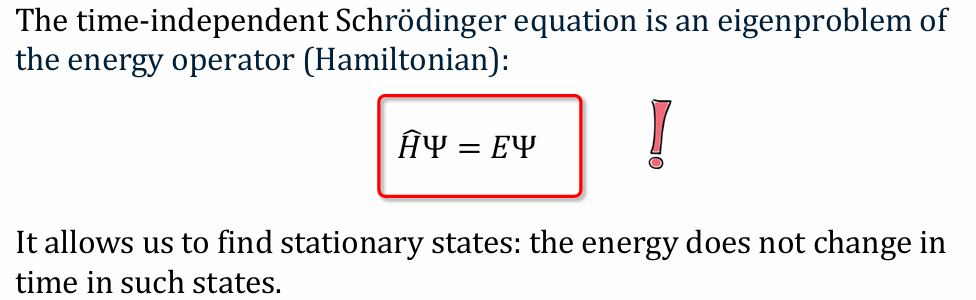
\what is the formula used for Calculating the quantum average?
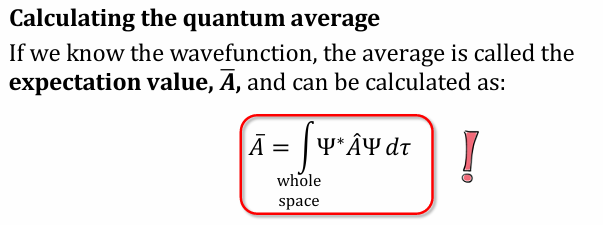


Write down the formula for the Uncertainty principle

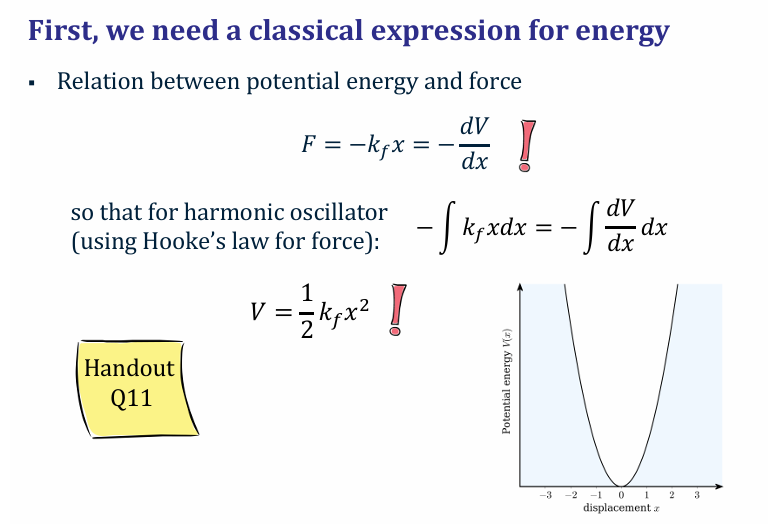
Give the Relation between potential energy and force in a harmonic oscillator
and its relation
V is potential E in J
kf force constant Nm-1
x in m, extension of the spring
give an approximation of with units:
Planck’s constant
speed of light in ms-1 and cm-1
what we use to convert miu → kg
h≈ 6.63×10^−34Js
c≈ 3.00×10^8 cm s−1
c≈ 3.00×10^10 cm s−1
1u≈ 1.66×10^−27kg
Write J in 4 different terms and N in kg
Kg m2 s-2
Nm-1
CV-1, V is (volts)
Ws-1
1N= 1Kgm-1s-2
C and V in terms of s
and what is the me ( e- mass)
and kinetic E formula
C ≡ A s
V ≡ kg m2 s −3 A −1
me≈9.11×10^−31 kg
Ek= 1/2(mv²)
How to convert wavenumber to frequency
and how c relates to wavelength
v=wavenumber x c
v=s-1 or Hz
c=cms-1
wavenumber = cm-1
c= v x lambda
how to turn v from cm-1 to J
how to turn energy per photon → energy per mole
what is 1pm
what is 1 Angstrom
E=hv
multiply by avogardo NA= 6.022×10^23mol−1
1pm= 10^-12m
1 angstrom = 10^-10m