Gas Properties & Laws
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
What are the properties of gases?
gases do not have a definite shape or volume:
expand to fill any space made available to them
do not have clear boundaries
colorless and odorless (difficult to detect)
low densities
can be mixed together to form a solution (ex: air)
volume changes dramatically with change in pressure or temperature
What does the Kinetic Molecular Theory (KMT) explain the behavior of?
KMT explains the behavior of ideal gases
What are the basic assumptions of the Kinetic Molecular Theory (KMT)?
gases consist of extremely small particles (molecules) with negligible volume
molecules of a gas are in rapid, random, straight-line motion
gas molecules collide with each other elastically (no energy loss)
there are no attractive forces between molecules
avergae kinetic energy of gas particles is directly related to temperature
Under what conditions do real gases behave like ideal gases? Why?
real gases behave most like ideal gases at high temperatures and low pressure
high temperature: increases kinetic energy, overcoming attractive forces
low pressure: reduces intermolecular interactions
What is the Ideal Gas Law?
PV = nRT
P = pressure (kPa, atm)
V = volume (L)
n = number of moles (mol)
R = universal gas constant
8.314 (for kPA)
0.008206 (for atm)
T = temperature (K)
What is Boyle’s Law?
P ∝ 1/V
pressure and volume are inversely related
What is Charles’ Law?
V ∝ T
volume and temperature are directly related
What is Gay-Lussac’s Law?
P ∝ T
pressure and volume are directly related
What is Avogadro’s Law?
n ∝ V
the volume of a gas is proportional to its number of moles
What is the formula for the density of a gas?
d = Pmm / RT
What is the Combined Gas Law? What is it used for?
converts the volume, pressure, and temperature of a given amount of gas from one set of conditions (1) to another set of conditions (2)
n (# moles) and R are constant
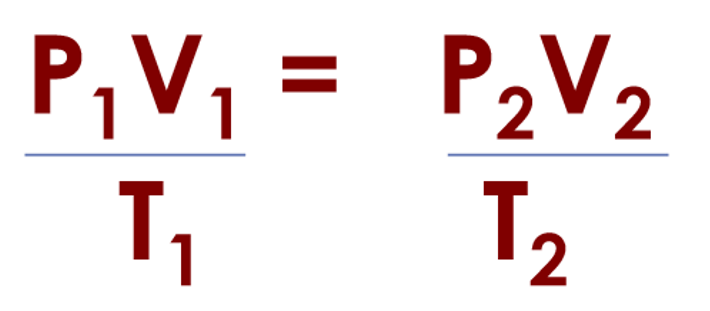
What is Dalton’s Law of Partial Pressure?
the total pressure of a gaseous mixture = the sum of the partial pressures (pressures of the individual gases)
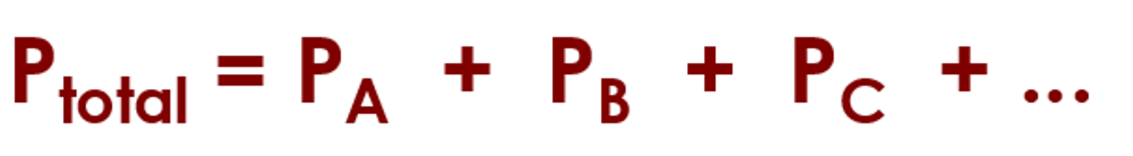
How to use Dalton’s Law to find the pressure of a gas in a mixture containing water vapor (collected over water)?
pressure of dry gas alone = total pressure - pressure of water vapor (found using the reference chart depending on temperature)

What does the Maxwell-Boltzmann distribution curve say about the temperature and kinetic energy of gases?
increased temperature → increase the average speed of molecules (T ∝ average KE)
bell curve because not all molecules are traveling at the same speed
note: the number of molecules (n) stays the same
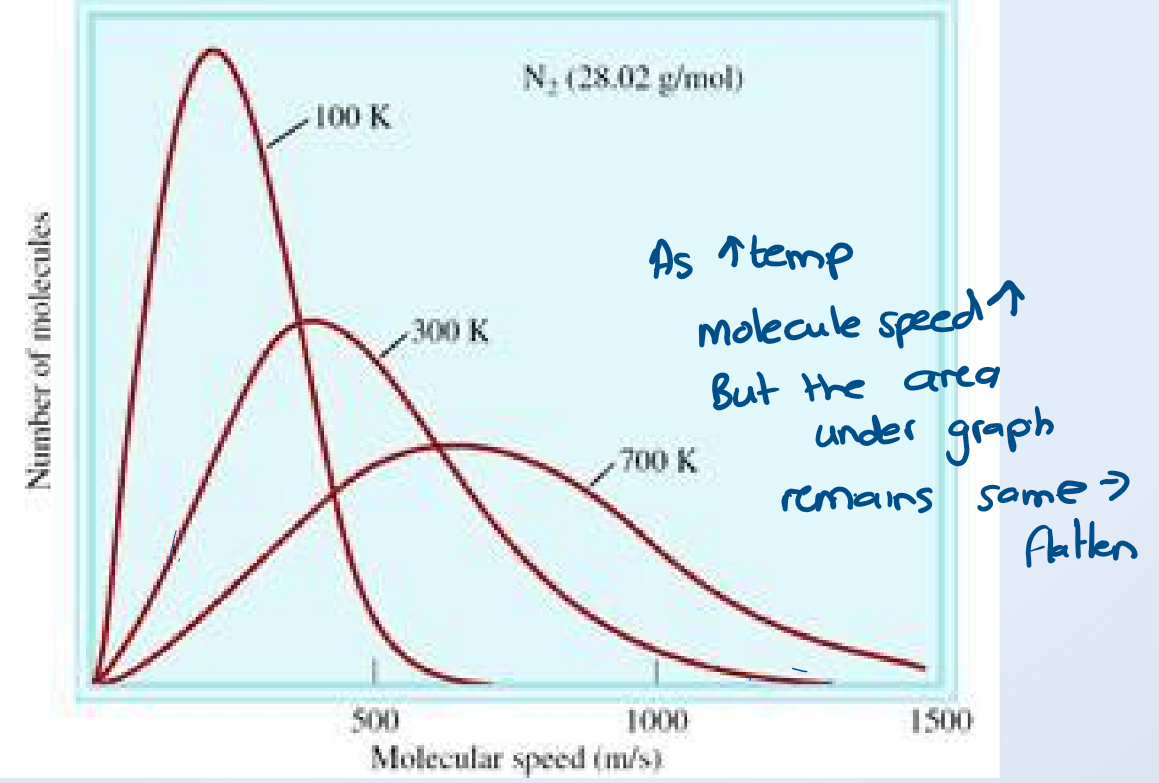
Consider three different gases with at the same temperature. Will the average kinetic energy of these gases be the same?
Yes, temperature is a measure of the average kinetic energy of a gas. Thus, saying that the gases have the same temperature is the same as saying that the gases have the same average kinetic energy.
However, speeds are dependent on masses
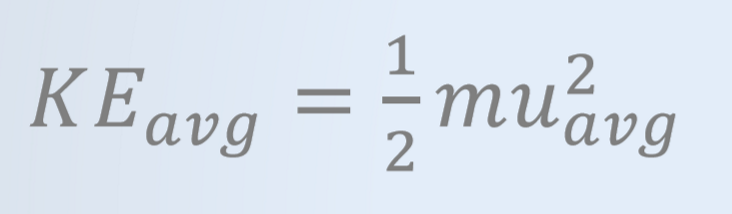
Consider three different gases with different molar masses at the same temperature. Will the speed of these gases be the same?
No, the average speed of the gases are not the same.
At the same temperature (the same average kinetic energy), gases with greater molar masses will have a lower speed (m ∝ 1/u²)
How is the average molecular speed of gases related to their molar mass?
Average molecular speed is greater when the mass of the molecular is smaller (m ∝ 1/u²)
What is effusion?
The escape of gas moleucles through a tiny hole into an evacuated (empty) space.
What is diffusion?
The spread of one substance throughout a space.
What is Graham’s Law?
a way to quantify the rate of effusion and diffusion of gases
rate of diffusion or effusion is inversely related to the square root of its molar mass
makes sense because m ∝ 1/u²

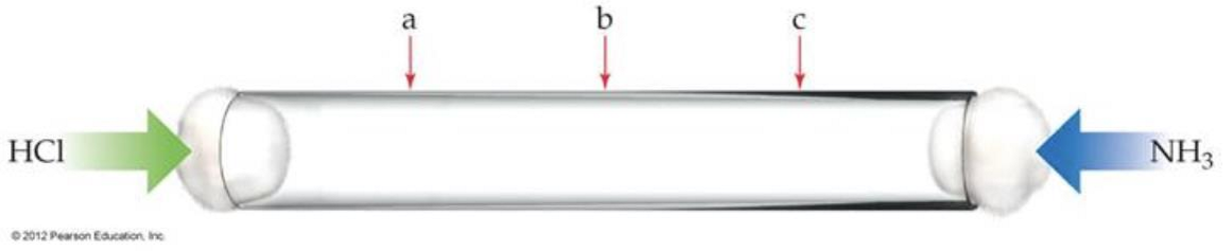
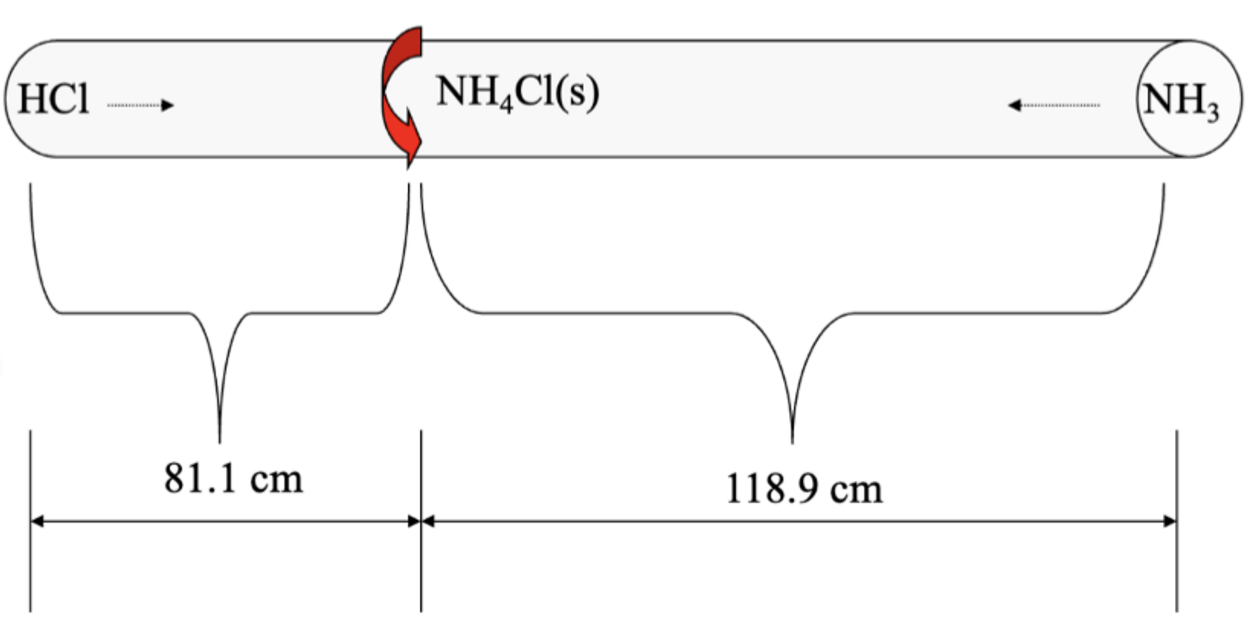
Consider two different gases released at opposite ends into a tube. They will combine to form a solid. Where will the solid form? (mm of HCl > mm of NH3)
the solid will form closer to the HCl
this is because a lighter gas travels faster than a heavier gas