Higher Physics Particles and Waves definitions
0.0(0)
0.0(0)
New
Card Sorting
1/96
Earn XP
Description and Tags
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
97 Terms
1
New cards
Voltage
The energy given to each coulomb of charge that passes through a power supply
2
New cards
Rutherford scattering experiment results
* Most of the alpha particles passed straight through the foil with little or no deflection. Given the foil was at least 100 atoms thick this suggests the atoms must be mostly empty space
* A few particles were deflected through large angles. In order to produce these large deflections, the positively charged alpha particles must be encountering something of very large mass and a positive charge
* A few particles were deflected through large angles. In order to produce these large deflections, the positively charged alpha particles must be encountering something of very large mass and a positive charge
3
New cards
Antimatter
Consists of particles that are identical to their counterparts in every way apart from charge
4
New cards
What revealed the existence of antimatter
High-energy collisions
5
New cards
Annihilation
When a matter particle meets an anti-matter particle they annihilate, giving off energy
6
New cards
The Standard Model
A model of fundamental particles and interactions
7
New cards
What is the Standard Model a theory of
The Standard Model is our best theory to explain what the Universe is made of. It consists of 17 particles: 12 fermions (6 quarks and 6 leptons) and 5 bosons
8
New cards
Fundamental Particle
A subatomic particle which is not composed of other particles
9
New cards
Fermions
Matter particles. This group contains both quarks and leptons.
10
New cards
6 type of quark
* up (u)
* down (d)
* charm (c)
* strange (s)
* top (t)
* bottom (b)
* down (d)
* charm (c)
* strange (s)
* top (t)
* bottom (b)
11
New cards
What are neutrons and protons made up of
quarks
12
New cards
Hadrons
Composite particles which consist of quarks
13
New cards
2 types of hadron
Baryons and mesons
14
New cards
Baryons
Made up of 3 quarks
15
New cards
Mesons
Made up of quark-antiquark pairs
16
New cards
6 Leptons
* electron
* muon
* tau
* electron neutrino
* muon neutrino
* tau neutrino
* muon
* tau
* electron neutrino
* muon neutrino
* tau neutrino
17
New cards
First evidence for the neutrino
Beta decay
18
New cards
How did beta decay show the evidence of the neutrino
When looking at the reaction it was seen that energy and momentum was not conserved, so this must have been carried away by an unseen particle
19
New cards
4 fundamental forces present when particles interact
* Strong nuclear force
* Weak nuclear force
* Electromagnetic force
* Gravitational force (weakest)
* Weak nuclear force
* Electromagnetic force
* Gravitational force (weakest)
20
New cards
Strong nuclear: Exchange particle & example effect
* Gluon
* Holding protons in nucleus
* Holding protons in nucleus
21
New cards
Weak nuclear: Exchange particle & example effect
* W and Z bosons
* Beta decay
* Beta decay
22
New cards
Electromagnetic force: Exchange particle & example effect
* Photon
* Holding electrons in atoms
* Holding electrons in atoms
23
New cards
Gravitational force: Exchange particle & example effect
* Graviton
* Holding matter in planets, stars and galaxies
* Holding matter in planets, stars and galaxies
24
New cards
Bosons
particles associated with the 4 fundamental forces
25
New cards
Electric Field
* A place where there is a force on a charge
* In an electric field a charged particle will experience a force
* In an electric field a charged particle will experience a force
26
New cards
Electric charge and electric field
Electric charges are surrounded by an electric field
27
New cards
Movement of Charge in an electric field key concept
* Work must be done to move the charge against the direction of force
* The work done in moving the charge against the field = the change in electrical potential energy
* The work done in moving the charge against the field = the change in electrical potential energy
28
New cards
Magnetic field wire
A wire with a current flowing through it creates a magnetic field
29
New cards
Stationary charge
A stationary charge creates an electric field
30
New cards
Moving charge
A moving charge creates both an electric field and magnetic field
31
New cards
Acceleration by electric fields
As the particles speed around the beam pipes they enter special accelerating regions where there is a rapidly changing electric field
32
New cards
Deflection by magnetic fields
The protons in the beam pipes would go in a straight line if they were not constantly going past powerful, fixed magnets which cause them to travel in a circle
33
New cards
High energy collisions (particle accelerators)
When they are travelling fast enough they are made to collide within a detector
34
New cards
Fission
A heavy nucleus disintegrates, forming two nuclei of smaller mass number and several free neutrons and energy
35
New cards
Chain reaction
The neutrons released in a fissions reaction go on to cause further fission reactions
36
New cards
2 types of fissions
* Spontaneous fission
* Induced fission
* Induced fission
37
New cards
Induced fission
Fission can be induced/persuaded to happen by neutron bombardment
38
New cards
Difficulties with fusion
* Coolant issues
* Containment issues
* Containment issues
39
New cards
Coolant issues fusion
* A nuclear fusion reactor generates a lot of heat energy and a safe method is needed to transfer that heat into electricity
* A coolant (usually water) is used to transfer the heat energy
* A coolant (usually water) is used to transfer the heat energy
40
New cards
Containment issues fusion
* If plasma comes into contact with the walls of the reactor, it will cool down
* To prevent this from happening, a powerful magnetic field is used to contain the moving charges within the reactor
* To prevent this from happening, a powerful magnetic field is used to contain the moving charges within the reactor
41
New cards
Irradiance
Irradiance is the power per unit area incident on a surface
42
New cards
Inverse Square Law
Irradiance on a surface will reduce as the distance is increased
43
New cards
Inverse Square Law Experiment
1. Measure the distance (d) between the lamp and the light detector using a metre stick
2. Record the irradiance (I) of the lamp using the light sensor
3. Increase the distance between the lamp and the source and repeat for a further 4 distances
4. Plot a graph with I on the y-axis and 1/d^2 on the x-axis. This will show that irradiance is directly proportional to I/d2
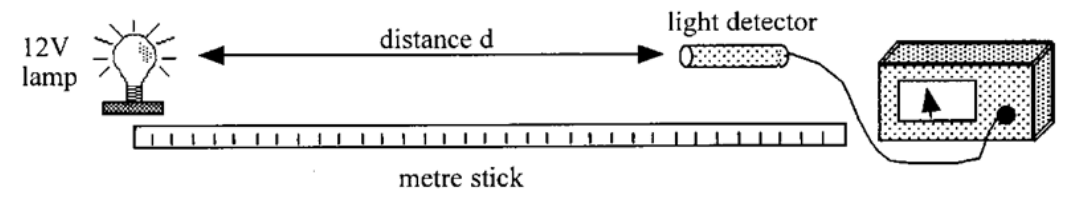
44
New cards
Main variable to ensure constant in inverse square law experiment
Background light level
45
New cards
Wave-particle duality
Light can act both like a wave and like a particle without contradiction. This is known as wave-particle duality
46
New cards
Photoelectric effect
The photoelectric effect is evidence for the particle model of light
47
New cards
Photoemission
Photons of sufficient energy can eject electrons from the surface of materials
48
New cards
When will the photoelectric effect occur
* If the radiation has a high enough frequency
* If the surface is suitable - UV light will not eject electrons from iron, copper or lead but will from sodium and potassium
* If the surface is suitable - UV light will not eject electrons from iron, copper or lead but will from sodium and potassium
49
New cards
As irradiance increases how is electron emission affected
The greater the irradiance the more electrons emitted
50
New cards
Current relationship graph with irradiance
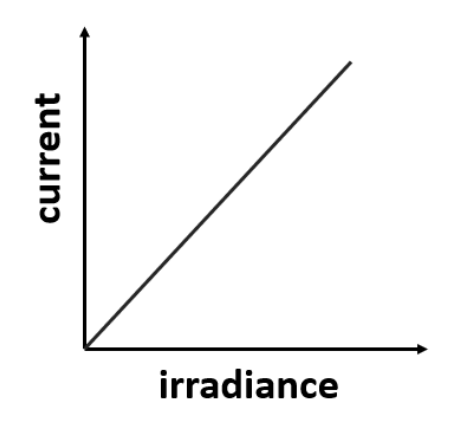
51
New cards
Threshold frequency
The minimum frequency of a photon required for photoemission
52
New cards
How is current affected as threshold frequency increases
An increase in frequency will cause an increase in the speed of the electrons being emitted, in turn leading to an increase in current
53
New cards
current threshold frequency graph
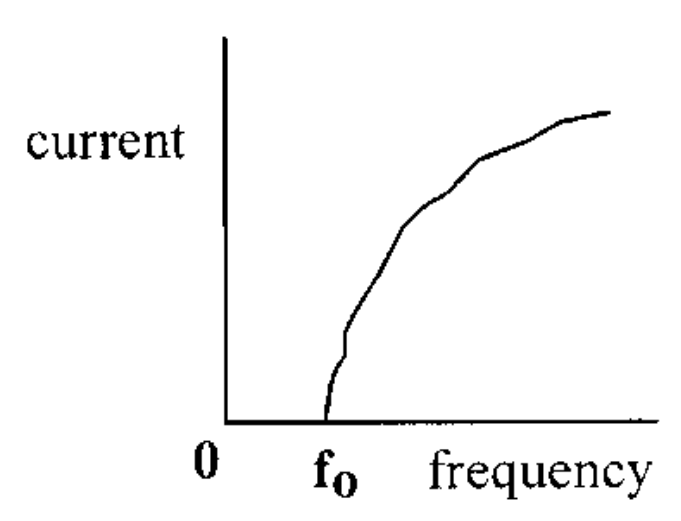
54
New cards
Work function
The work function of a material is the minimum energy of a photon required to cause photoemission
55
New cards
Equation for work function
Work function = hfo
56
New cards
Period
The time it takes for one wave to pass a point (s)
57
New cards
Frequency
The number of waves (N) which pass a point in one second (Hz)
58
New cards
Diffraction
The spreading out of waves around obstacles or through a gap
59
New cards
Long wavelength compared to short wavelength diffraction
Long wavelength waves diffract **more** than short wavelength waves
60
New cards
Energy of wave dependent on amplitude
The larger the amplitude the more energy the wave has
61
New cards
Constructive interference
Two sets of waves meet exactly in phase
62
New cards
Destructive interference
Two sets of waves meet completely out of phase
63
New cards
Coherent Sources
Two waves are coherent if they have a constant phase relationship. They will have the same frequency and velocity
64
New cards
Interference of light
Interference can only be explained in terms of wave behaviour and as a result, interference is taken as proof of wave motion
65
New cards
Two sources of coherent light are needed to produce what?
Two sources of coherent light are needed to produce an interference pattern
66
New cards
Zero order (central) maxima
* The waves have zero path difference
* The point they meet is equidistant from each source
* The point they meet is equidistant from each source
67
New cards
Maxima
* Waves arrive in phase
* The waves at the first maxima have a path difference of 1λ
* The waves at the 2nd maxima have a path difference of 2λ
* The waves at the first maxima have a path difference of 1λ
* The waves at the 2nd maxima have a path difference of 2λ
68
New cards
Minima
* Waves arrive out of phase
* The waves at the first minima have a path difference of 1/2λ
* The waves at the 2nd minima have a path difference of 1 1/2λ
* The waves at the first minima have a path difference of 1/2λ
* The waves at the 2nd minima have a path difference of 1 1/2λ
69
New cards
Maximum path difference equation
S2P - S1P = mλ
70
New cards
Minimum path difference equation
S2P - S1P = (m + 1/2)λ
71
New cards
Increasing and decreasing separation of maxima
To increase θ, the separation of the maxima, you can:
* increase the wavelength, i.e. move from blue towards red light
* decrease the slit separation, i.e. have more lines per mm
* increase the wavelength, i.e. move from blue towards red light
* decrease the slit separation, i.e. have more lines per mm
72
New cards
Bohr Model of the Atom
* Electrons exist only in allowed orbits and they do not radiate energy if they stay in this orbit, i.e. Bohr’s model proposed that the classical electromagnetic theory was incorrect
* Electrons tend to occupy the lowest available energy level, i.e. the level closest to the nucleus
* Electrons in different orbits have different energies
* Electrons can only jump between allowed orbits
* Electrons tend to occupy the lowest available energy level, i.e. the level closest to the nucleus
* Electrons in different orbits have different energies
* Electrons can only jump between allowed orbits
73
New cards
Bohr Model: How are electrons able to move between levels
* If an electron absorbs a photon of exactly the right energy, it moves up to a higher energy level
* If an electron drops down from a high to a low energy state it emits a photon which carries away the energy
* If an electron drops down from a high to a low energy state it emits a photon which carries away the energy
74
New cards
Ground state, E0
The orbit closest to the nucleus
75
New cards
Excited state (E1 - E5 above)
Orbits above the ground state, further away from the nucleus
76
New cards
Ionisation level
The level furthest away from the nucleus - an electron which has gained just enough energy to leave the atom has Ek = 0J
77
New cards
Zero potential energy
Taken to be at infinity (from the nucleus)
78
New cards
Types of spectra
* Continuous Spectra
* Line Emission Spectra
* Line Absorption Spectra
* Line Emission Spectra
* Line Absorption Spectra
79
New cards
Continuous Spectra

80
New cards
Line Emission Spectra

81
New cards
Line Absorption Spectra

82
New cards
Planck equation relationship
The frequency of the emitted radiation is related to the change in energy ΔE of the electron using the Planck equation
83
New cards
Fraunhofer lines
The absorption lines (Fraunhofer lines) in the spectrum of sunlight provide evidence for the composition of the Sun’s outer atmosphere
84
New cards
Refraction
Refraction is the change in speed and wavelength when a wave moves from one medium into another
85
New cards
Normal (refraction)
An imaginary line at 90\* to the surface, drawn as a dotted line
86
New cards
Incident ray
The ray going towards the surface
87
New cards
Angle of incidence (i)
The angle between the incident ray and the normal
88
New cards
Refracted ray
The ray leaving the surface
89
New cards
Angle of refraction (r)
The angle between the refracted ray and the normal
90
New cards
speed of light as it moves from air to glass
The speed of light decreases as it moves from air to glass
91
New cards
wavelength of light as it moves from air to glass
The wavelength of light decreases as it moves from air to glass
92
New cards
frequency of light as it moves from air to glass
The frequency of light remains the same
93
New cards
Experiment to determine the refractive index of a material
Aim: To determine the refractive index of a medium
\
1. Measure angle of incidence θ1 in air and angle of refraction θ2 in the medium using a protractor
2. Gradually increase θ1, measuring pairs of values θ1 and θ2 each time
3. Calculate sinθ1 and sinθ2 for each pair of values
4. Plot a graph with sinθ2 on the x-axis and sinθ1 on the y-axis, this will produce a straight line through zero and the gradient is equal to the refractive index n
\
1. Measure angle of incidence θ1 in air and angle of refraction θ2 in the medium using a protractor
2. Gradually increase θ1, measuring pairs of values θ1 and θ2 each time
3. Calculate sinθ1 and sinθ2 for each pair of values
4. Plot a graph with sinθ2 on the x-axis and sinθ1 on the y-axis, this will produce a straight line through zero and the gradient is equal to the refractive index n
94
New cards
Refractive index
The (absolute) refractive index of a medium as the ratio of the speed of light in a vacuum to the speed of light in the medium
95
New cards
Refractive index relational to the frequency of light
As the frequency of light increases, the refractive index increases
96
New cards
Critical Angle
The critical angle is the angle of incidence which produces an angle of refraction of 90\*
97
New cards
Total internal reflection
Total internal reflection occurs when the angle of incidence is greater than the critical angle