Biological molecules ocr A level biology
1/56
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
Biological molecules
Carbohydrates
Lipids
Proteins
Nucleic acids
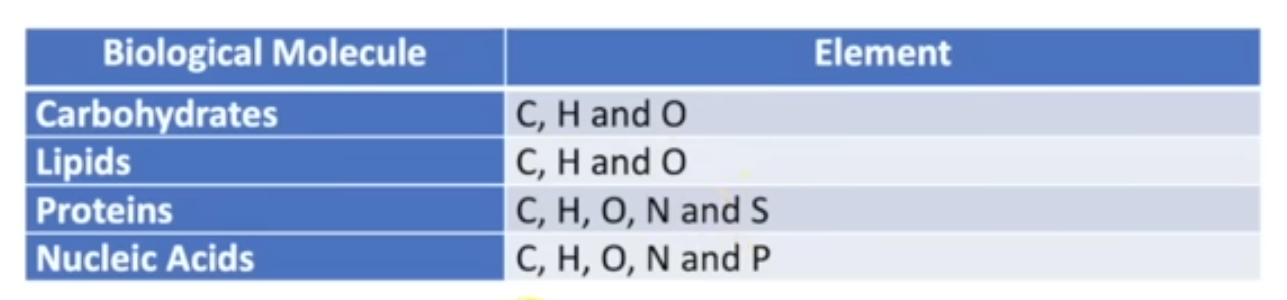
Carbohydrates - C,H,O
Lipids - C,H,O
Proteins -C,H,O,N,S
Nucleic acids - C,H,O,N,P,S
(calcium ions)
Cations
(Ca2+)
involved in muscle contraction and nerve impulse transmission
sodium ions
Cation
Na+
Involved in co - transport
reabsorption of water in the kidney
nerve impulse transmission
Potassium ions
cation
(K+)
involved in stomatal opening
nerve impulse transmission
Hydrogen ions
cation
(H+)
very important for catalyzing enzyme-controlled reactions
ammonium ions
Cation
(NH4 +)
Useful in nitrogen cycle, bacteria converts ammonium ions to nitrate ions
nitrate
anion
(NO3 –)
Mineral ions absorbed by plants to provide nitrogen for amino acids
phosphate
anion
PO43-
Formation of phospholipids for cell membranes
creating nucleic acid
ATP for making bones
Chloride
Anion
(Cl –)
Provide a negative charge to balance positive sodium ions and positive ions in cells
hydrogencarbonate
anion
Maintains pH of blood
HCO3-
Water charge
Oxygen and hydrogen chemically bonded with covalent bond
Oxygen is slightly negative Hydrogen is slightly positive
Water is a polar molecule
Opposite charges form hydrogen bonds
Specific Heat capacity
Water has a very high specific heat capacity ( a lot of energy is required to change temperature)
Heat energy go into breaking hydrogen bonds not kinetic energy of molecules so water is a buffer to instant temp change - allowing organism to live in water
Ice is less dense than water and insulates water below so it doesn’t freeze - organism can live in water
latent heat of vaporization
Very high latent heat of vaporization ( a lot of heat energy to evaporate water)
Organisms can cool themselves without losing a lot of water
Sweating - water evaporates allowing organism to cool down
Water as a solvent
Water is an excellent solvent - contain dissolved oxygen used for organisms in respiration
Can be used to transport CO2, glucose, amino acids in blood
Also used in xylem e.g. magnesium
Cohesion - surface tension allows surface of water to a habitat for insects e.g. pondskaters
Water metabolic reactions
Used in hydrolysis reactions
Used Photosynthesis
Produced in condensation reactions
Produced in respiration
Glucose
Hexose sugar
Monosccharide - single sugar molecule
A store of chemical energy - very soluble in water -Have many hydroxyl groups which are polar can form hydrogen bonds with water molecules
Hydrophilic
Why do plant cells store glucose as starch?
As glucose is so soluble
If a cell contains high amounts of glucose - water will move into cell by osmosis
Starch
Made of Amylose and Amylopectin
Store of glucose - very compact
Insoluble in water - does not make water enter through osmosis
Too large too diffuse out cell membrane
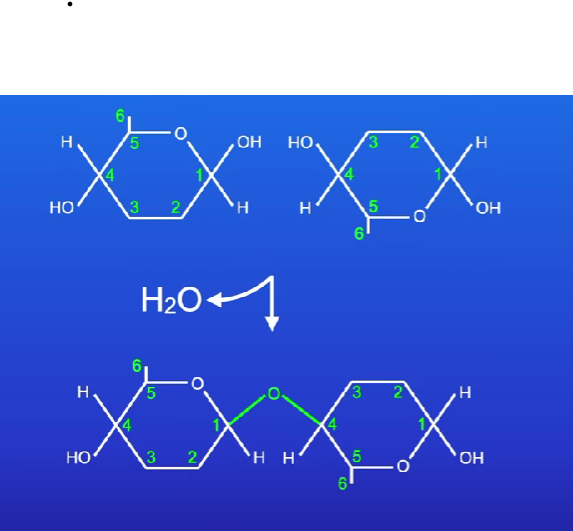
When glucose is needed water breaks glycosdic bonds in (hydolysis reactions)
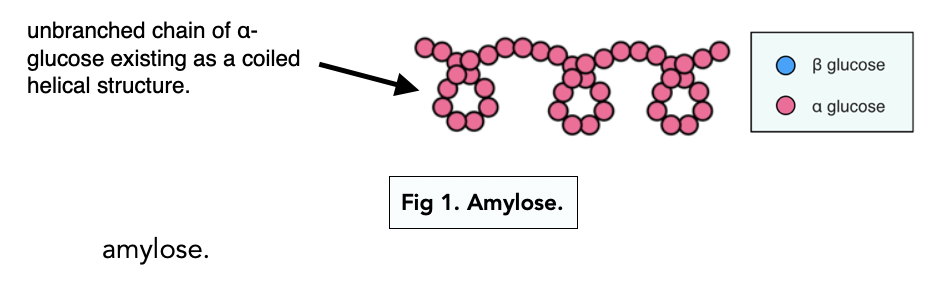
Amylose
Polymer of α glucose molecules
Chain of alpha glucoses with 1,4 glycosdic bonds - produce water
Twists into compact helix held in place by hydrogen bonds between glucose molecules
Tightly packed storage molecule
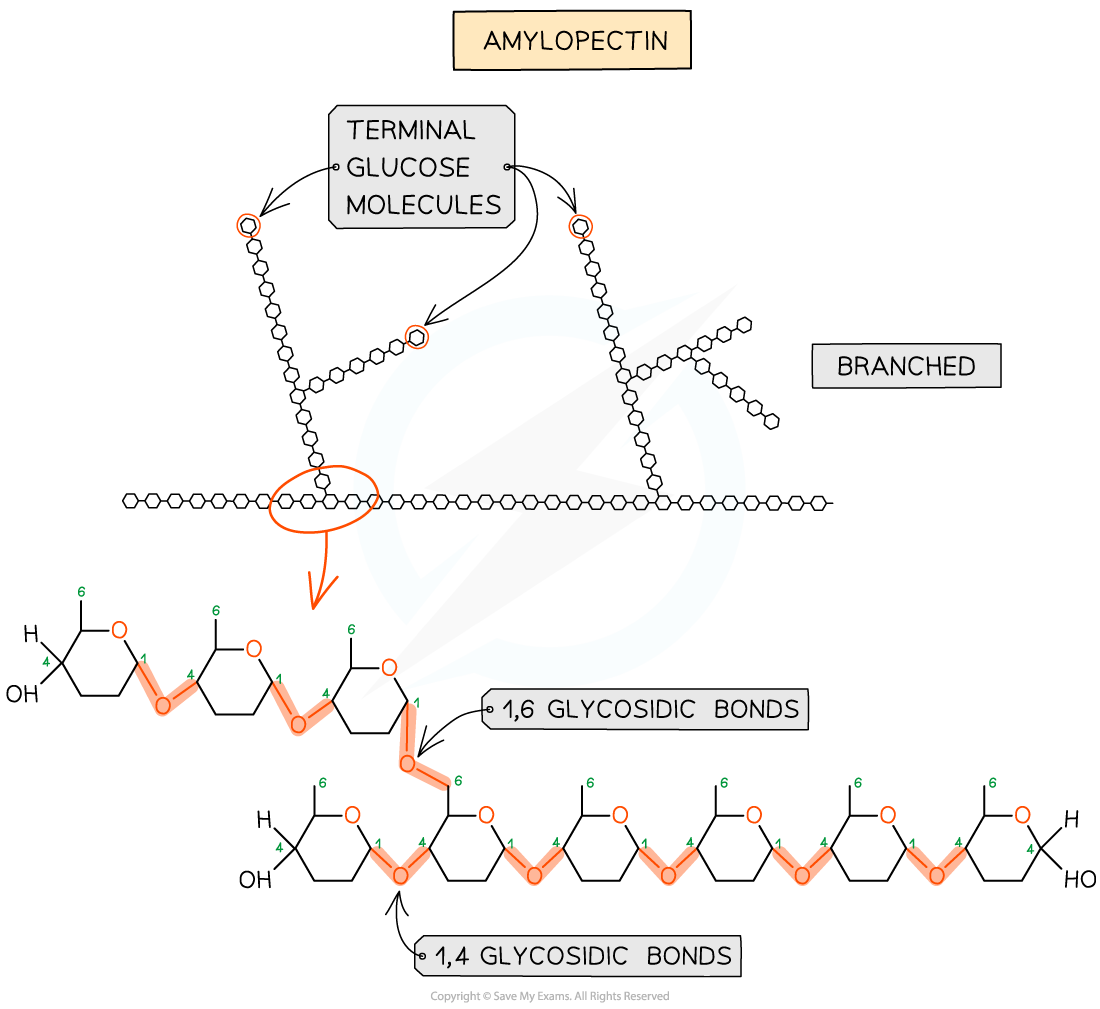
Amylopectin
Polymer of α glucose joined by 1,4, glycosidic bonds
Amylopectin is branched with another 1,4 chain between carbon 1’ of one a glucose molecules and carbon 6’ of another (every 25 - 30 molecules)
1,6 glycosdic bond
Enzymes break glycosidic bonds at ends of molecules - very branched - many ends - enzymes can break down starch rapidly into glucose
Glycogen
Animals store glucose as glycogen found in liver, muscle cells
Polymer of α glucose joined by 1,4, glycosidic bonds
Branched with 1,6, glycosidic bonds
More branched than amylopectin - very compact - many free ends - enzymes can convert glycogen to glucose very rapidly
Cellulose
Polymer of β glucose
Unbranched straight chain - cellulose molecules can get close together with hydrogen bonds chains form microfibrils
microfibrils —→ macrofribils ——> cellulose fibres ——→ cellwall
Huge number of hydrogen bonds makes cellulose strong
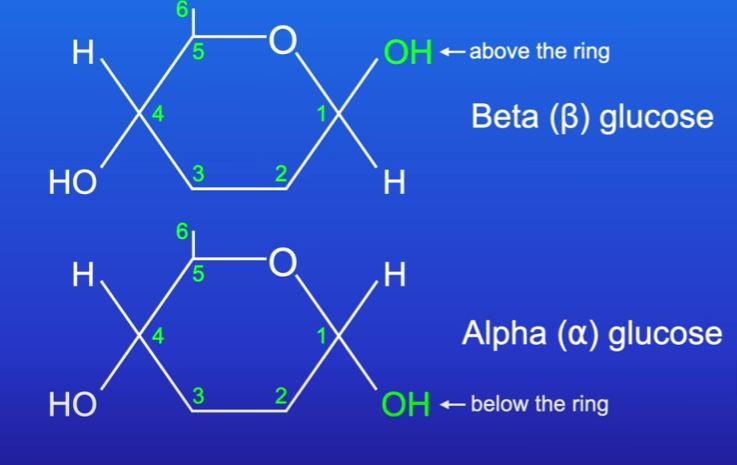
Hydroxyl of carbon 1’ points above ring
Every second β glucose molecule is flipped in cellulose as hydroxyl groups must be next to each other to form glycosidic bonds
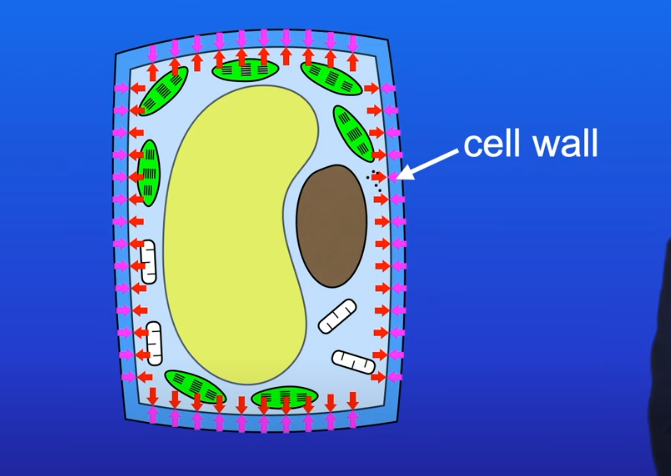
How does cellulose structure help its function
Cellulose cell wall is permeable to moleculues
As water moves in via osmosis it pushes cellulose cell wall
strength resists outward pressure
prevents plant cell from bursting - makes plant turgid creating upright structure
Why do animals need glucose
High rate of respiration
Energy need can change rapidly -move quickly to escape predator - glycogen can be rapidly transferred into glucose to be used in respiration
Glycogen is insoluble can not draw water in cell via osmosis
Large molecule cannot diffuse out of cell
Glucose isomers
Alpha glucose - Carobn 1’ hydroxyl group points below plane of ring
Beta glucose Carbon 1’ hydroxyl group points above plane of ring
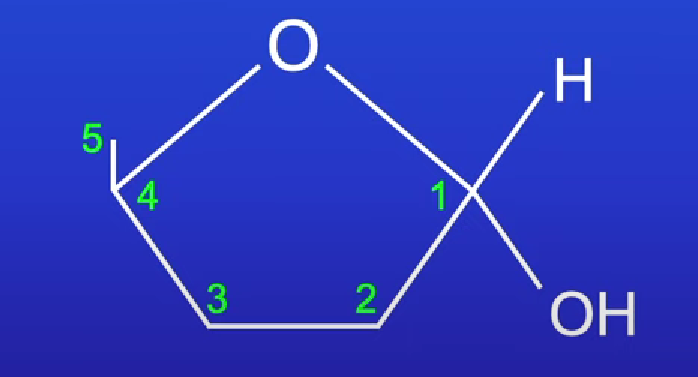
Ribose
Pentose sugar
Monosaccharides
Soluble in water - many hydroxyl groups - can form hydrogen bonds with water molecules
These molecules are hydrophillic
Can join to form disaccharides and polysaccharides
more monosaccharides - glucose, galactose, fructose
Disaccharide
Two monosaccharides react to form disaccharide
Glucose + glucose ——> maltose
Glucose + fructose ——> Sucrose
Glucose + galactose——> lactose
When a disaccharide is made a molecule of water is also produced from one hydrogen atom from one monsaccharide and a hydroxyl of another
Condensation reaction - water molecule is formed
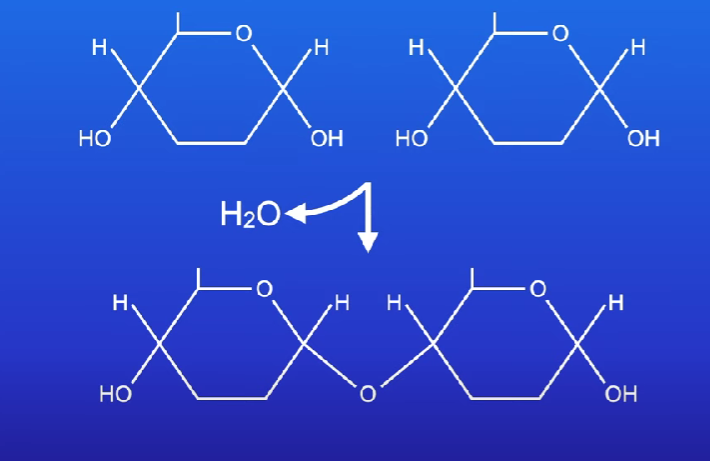
Glycosdic bond
Bond formed by two monosaccharides (condensation reaction)
Between carbon 1’ and carbon 4’
Oxygen link
1,4 glycosidic bond
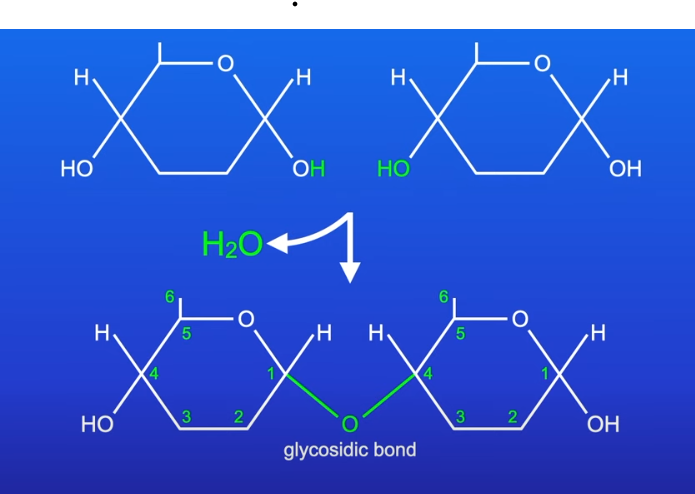
Condensation reaction
Disaccharides and polysaccharides are formed when two hydroxyl groups (on different saccharides) interact to form a strong covalent bond called the glycosidic bond
(the oxygen link that holds the two molecules together)
Every glycosidic bond results in one water molecule being removed and formed as a product
Thus glycosidic bonds are formed by condensation
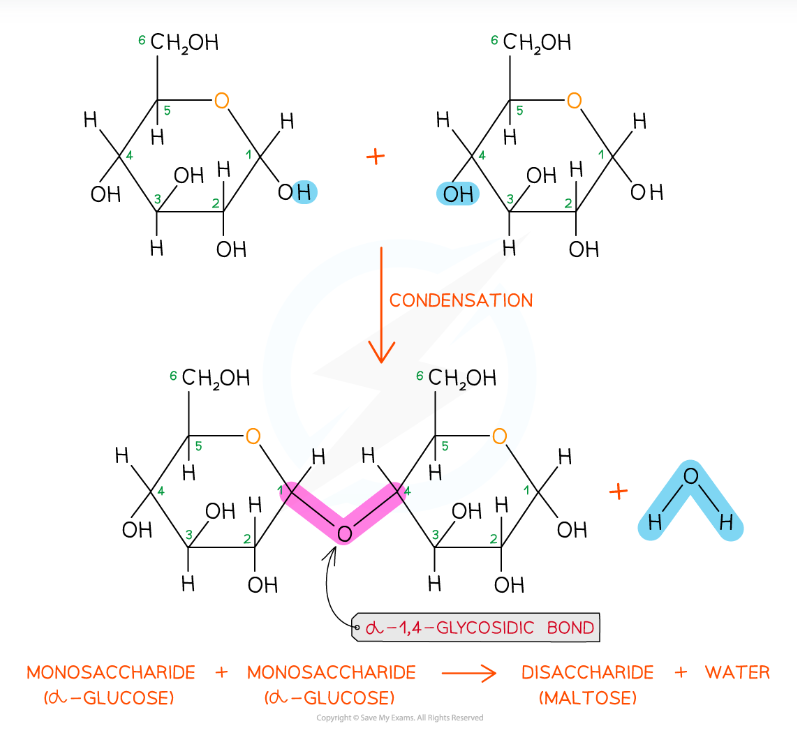
Hydrolysis reaction
Adding water will break glysosidic bond and revert disaccharide back to monsaccharide
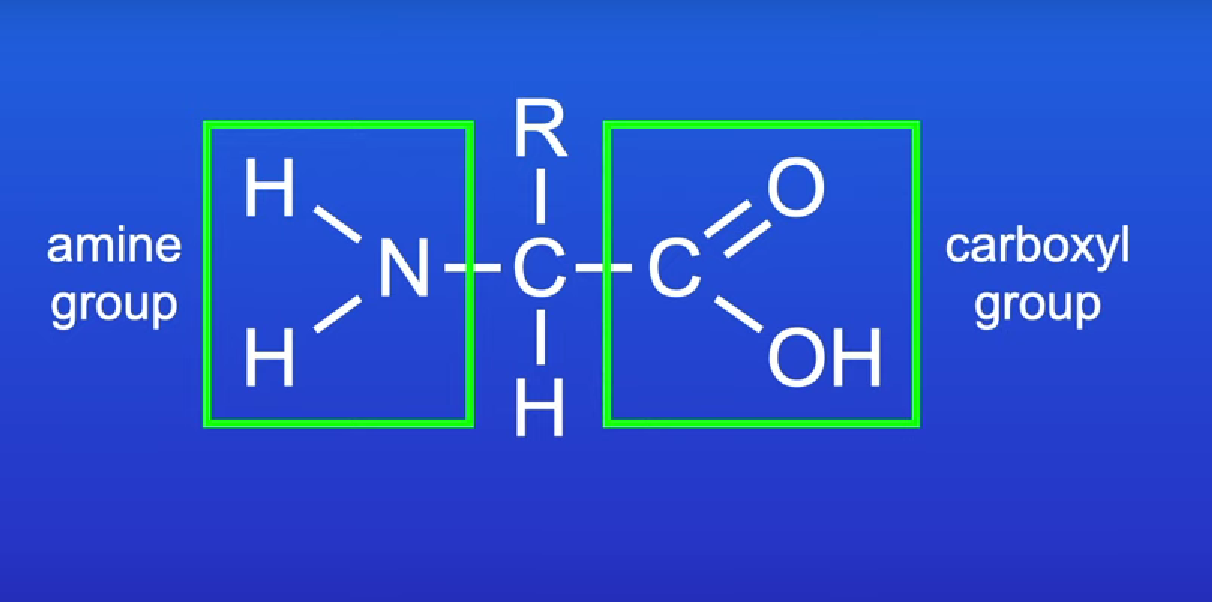
Amino acid groups
amine, carboxyl and R group
R group changes in different amino acids
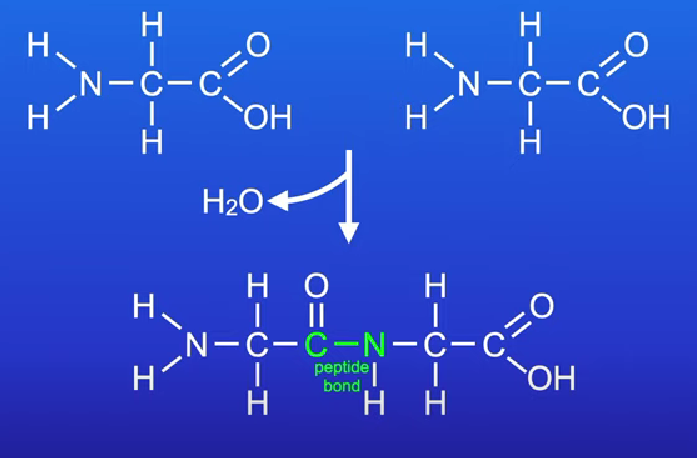
Peptide bond
Amino acids form a peptide bond (takes place in ribosomes (condensation reaction) makes water
peptide bond can be broken by adding water (hydrolsysis reaction) e.g. protease enzyme in digestive system
many peptide bond between amino acids polypeptide
Differemce between polypetide and protein?
Polypeptide needs to fold into complex 3d shape to become a protein
Can carry out its function
Primary structure
Primary structure - specific order of amino acids sequences in a polypeptide - helps to determine 3D shape - for function
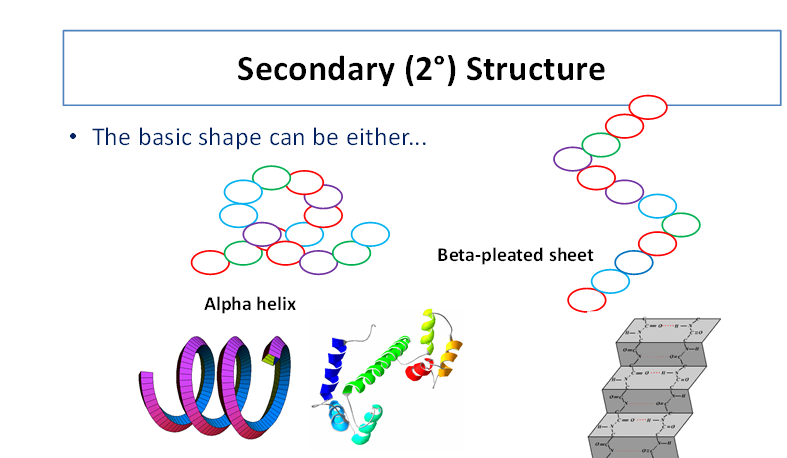
Secondary structure
C=O(oxygen small negative charged) groups and N-H (hydrogen small positive charge) groups
These charges attract each other - hydrogen bonds form - make polypeptide chain twist and fold - secondary structure e.g. alpha helix
Another type is beta pleated sheet - polypeptide chain folds into - flatter sheet like structure - hydrogen bonds between amino acids hold shape
Some amino acids found in alpha helices others in beta pleated sheets
Tertiary structure
Region of secondary structure fold into precise 3D shape - tertiary struucture
Weak Hydrophobic an Hydrophillic interactions - between R groups
Weak Hydrogen bonds
Strong covalent Disulfide bonds - between two R groups (only if contains sulfur)
ionic bonds between positively charged amine and negative R group
In picture - 5 alpha helices very specific pattern around a beta pleated sheet. e.g. active site of enzyme relies on protein forming of specific tertiary structure

Quarternary strucutre
Made from more than one polypeptide chain specific 3d structure
e.g. Haemoglobin with 4 chains
Haemoglobin has prosthetic haem group attached to each polypeptide chain
Prosthetic group - no amino acids but iron
Is a conjugated protein - non protein group added
Fibrous proteins
Polypeptides form long twisted strands linked together - highly repetitive - strong
Stable (unreactive)
insoluble in water
Form H bonds with adjacent chains
Very suitable for structural/ stability roles e.g. keratin - hair and nail
Outline 3 named fibrous proteins
Collagen - makes skin, cartilage, ligaments - quarternary structure has 3 polypeptide chains wound
Chains held by hydrogen and covalent bonds
Keratin - makes hair, skin and nails - insoluble so these strucutres are not broken by water
Elastin - makes elastic fibres of alveoli and arteries - stretch and g
Globular proteins
Polypeptide chains form a spherical shape
Unstable e.g. enzymes denatured
Soluble in water - hydrophillic R groups
Involved in metabolic functions e.g. enzymes, antibodies. hormones, haemoglobin
Outlined 3 named globular proteins
Haemoglobin - 4 polypeptide chains 2 alpha/ 2 beta each bind to O2
Enzymes - Pepsin in stomach digests proteins using specific active site
Insulin - produced by beta cells reduces blood glucoose conc - specific to muscle and liver cells
Lipids
Made of fatty acids and glycerol
Non - polar
Insoluble in water
Hydrophobic dissolve in ethanol
Do not form polymers
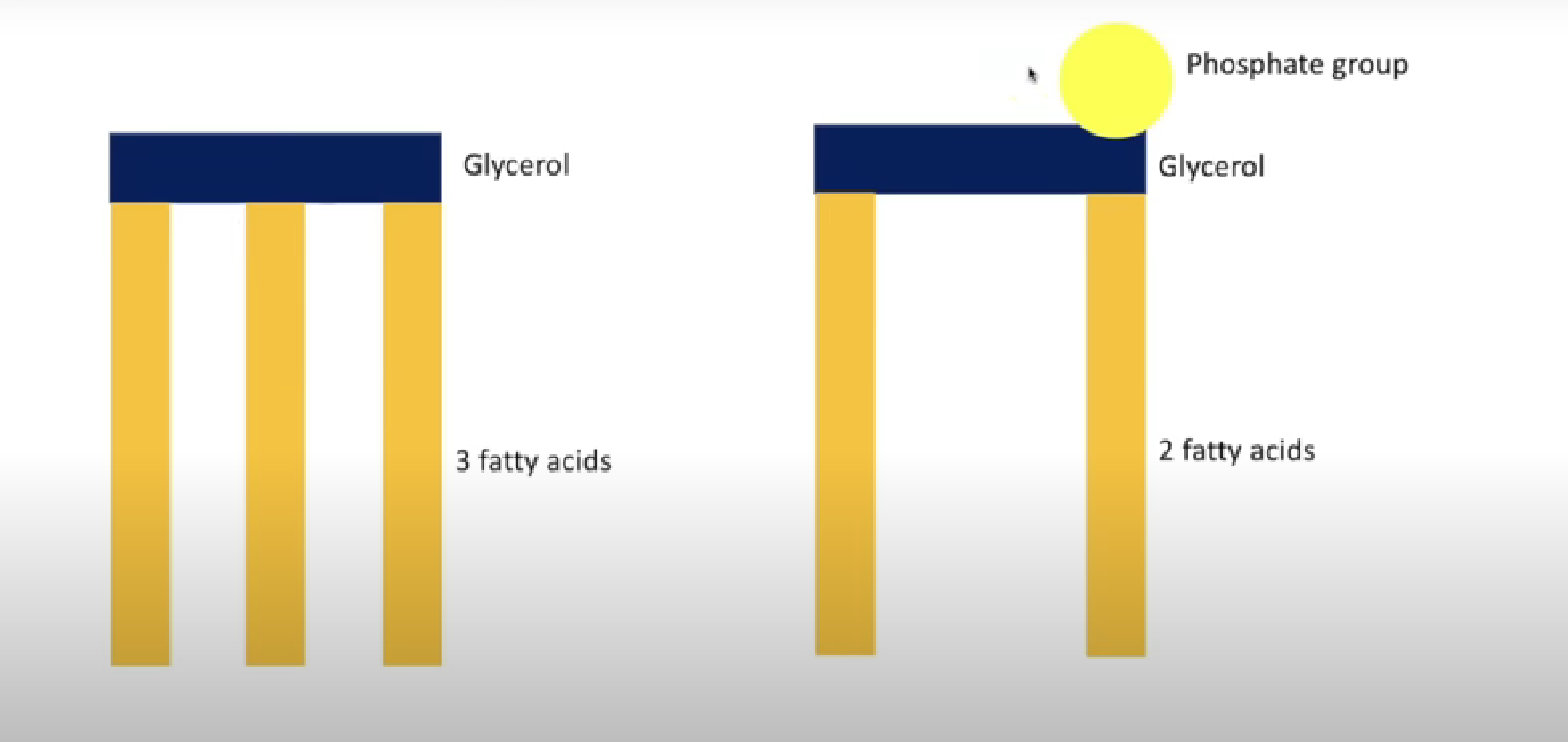
Triglyceride
Triglycerides are formed via condensation reactions
one molecule of glycerol + three molecules of fatty acids ———> tryglycerides
3 Ester bond formed
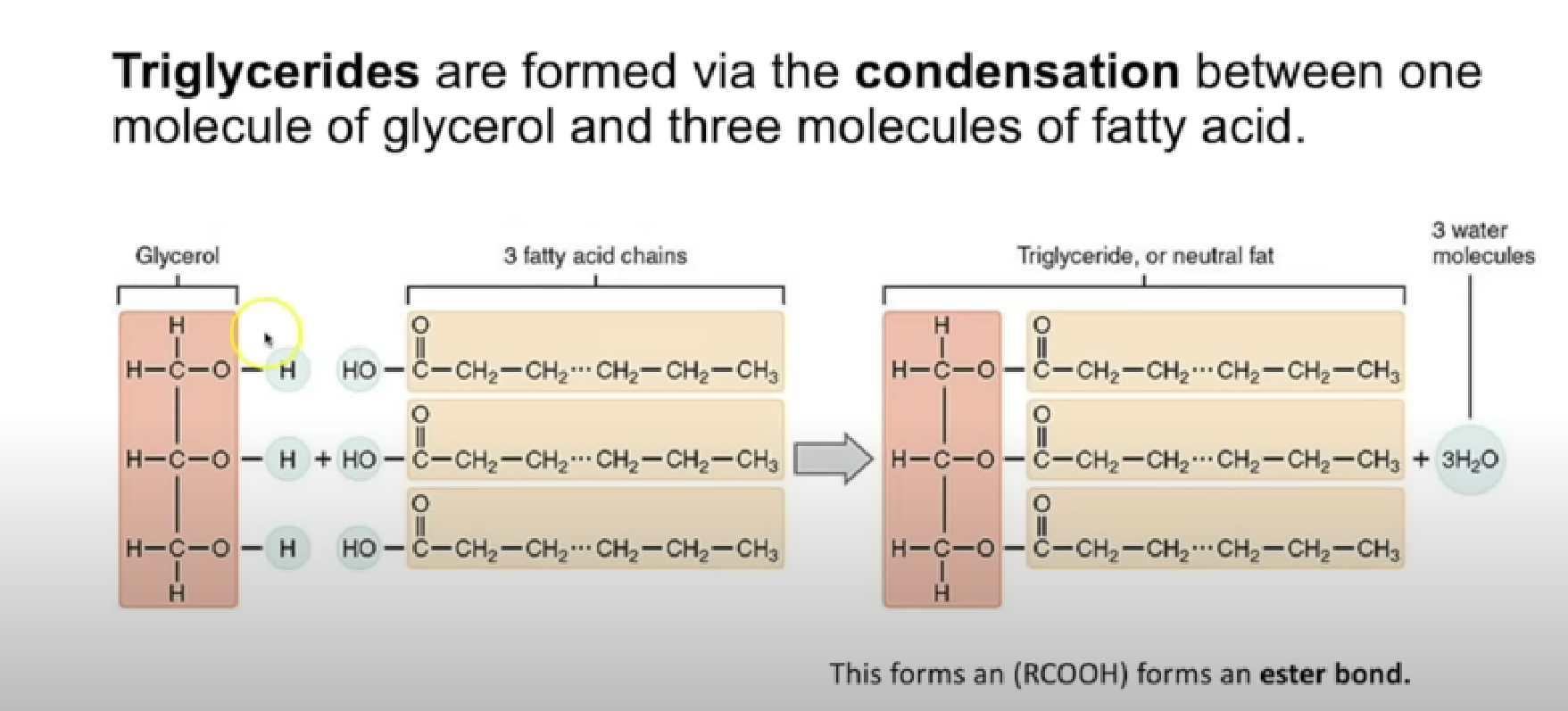
Properties of tryglycerides
Can transfer energy - large ratio of energy - storing carbon - hydrogen bonds compared to number of carbon atoms. A lot of Energy can be transferred when be broken down
High ratio of hydrogen to oxygen atoms they can act as a metabolic water source. This is because triglycerides can release water if oxidised. Essential for desert animals e.g. camels
Hydrophobic - Insoluble in water, do not affect osmosis
Low in mass can be stored without increasing mass - decreasing movement like muscle does
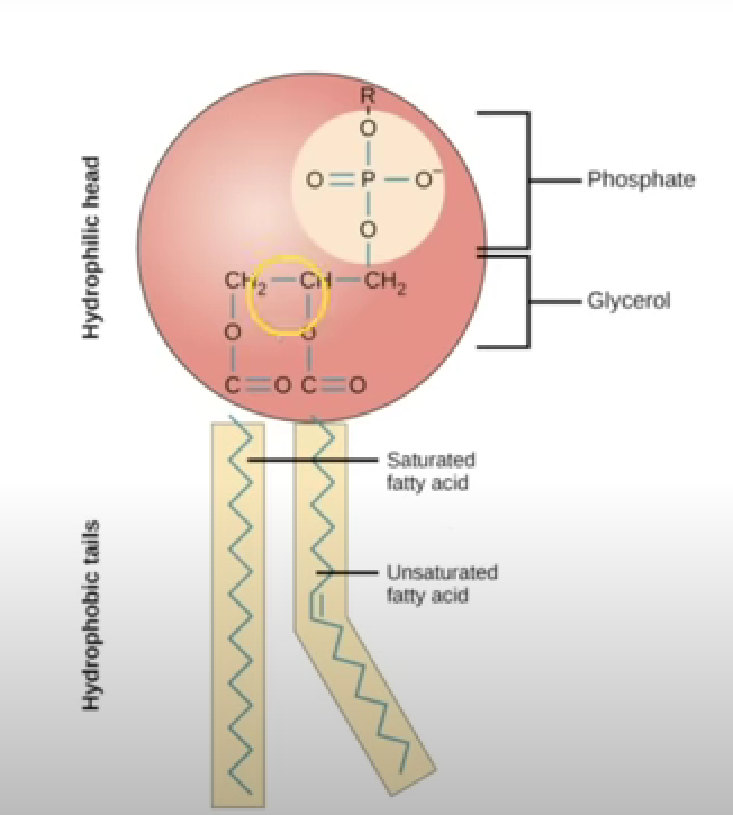
Phospholipid
Made of glycerol molecule, two fatty acids, phosphate group
Two fatty acids also bind to glycerol via condensation reactions forming two ester bonds
Properties of phospholipids
Hydrophilic head attracts water - as phosphate is charged repelling other fats
“tail” fatty acid chain is not charged repels water but mixes with fats
Forms phopsholipid bylayer which makes up structure for plasma cell membrane. hydrophillic head attracted to water hydrophobic tails repel from water
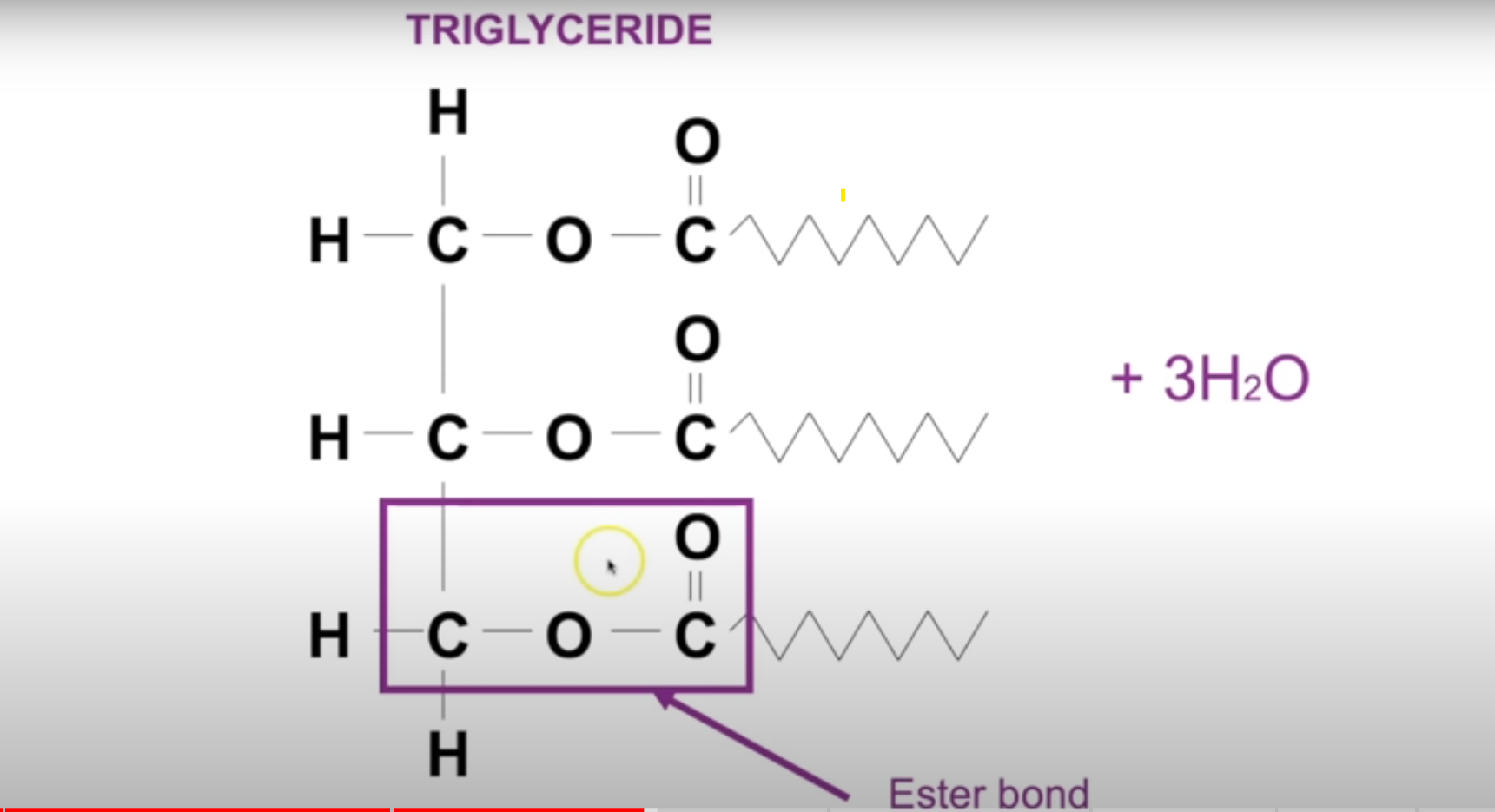
Ester bond
Bond formed between fatty acids and glycerol to form tryglyceride
Fatty acids
Saturated fatty acid - hydrocarbon chain has only single bonds between carbons
Unsaturated fatty acid - hydrocarbon chain has a double bond between carbon atoms
Chloesterol
Cholesterol is a sterol
4 carbon rings and a hydroxyl group
Both hydrophobic and hydrophillic regions
Imbedded in cell membranes to impact fluidity
Reduce fluidity high temperatures
Increase fluidity at low temperatures
Help control movement across cell membrane
Test for starch
iodine solution turns from orange to blue/black
Test for reducing sugars
Add benedict’s solution and heat 5mins at 80oC
Positive test blue - green - orange - brick red
can also use reagent test strips
Test for non reducing sugars
Following negative benedict’s test
Add acid and boil - hydrolysis
Cool solution then add alkali to neutralise
Add benedict’s solution and heat heat for 5mins 80oC
Only orange and brick red as when hydrolysed sucrose create fructose and glucose is made doubling sugar content
Test for protein
Add biurets solution
blue → purple
Test for lipids
Dissolve sample in ethanol
Add distilled water
Positive test white emulsion forms
Colorimeter method
Set filter opposite on the colour spectrum
Calibrate with distilled water to zero
Insert sample with different concentrations of glucose
Measure percentage transmission of light
Create callibration curve
As glucose conc increases benedict’s solution proportion decreases making solution less tinted and allows less light to be absorbed
Thin Layer Chromatography
Molecules move based on solubility in solvent
Calculate Rf value
Distance moved by solute/ Distance moved by solvent