Targets for cancer therapy Small molecule inhibitors and biologics.
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Why is there a need for targeted therapy
•To provide effective therapy
•cure or induce remission, maintain remission, slow disease progression
•To provide a selective therapy because many conventional systemic therapies are associated with a variety of serious adverse effects as they are unselective in targeting cells
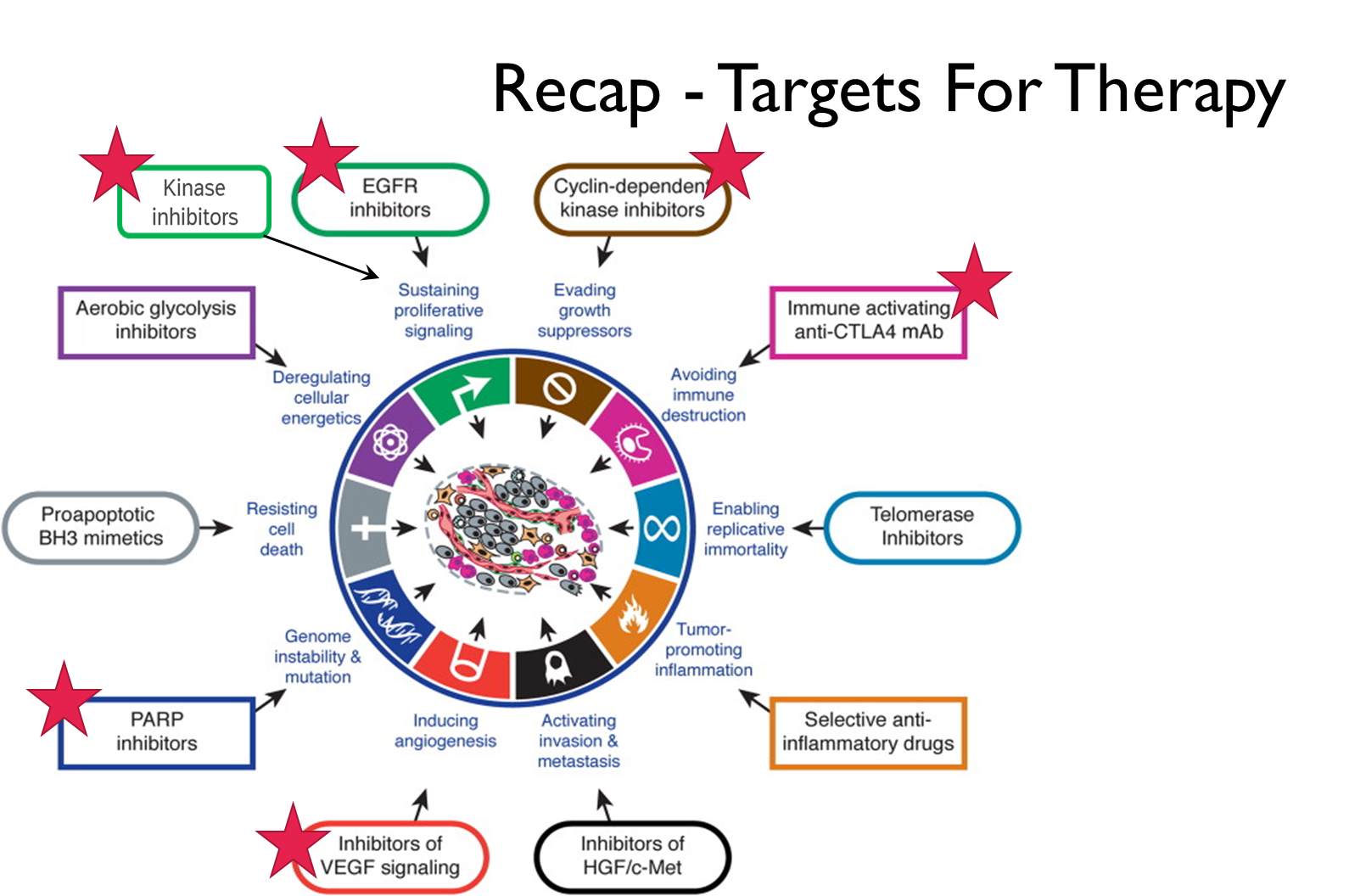
Recap - Targets For Therapy
Use of Biologics In Cancer Therapy
Biologics are used in cancer therapy to target and prevent proliferation, to inhibit angiogenesis and to act as vehicle for chemotherapy/radiotherapy. Can also act on the immune system to stimulate immune response or to prevent the inhibition of the immune response.
1.Block the action of the cell surface protein by preventing proteins binding to them (ligand binding to receptor / dimerization of receptor.) This is how biologics with specificity to EGFR work.
2.Bind to proteins to prevent them binding to cell surface proteins (i.e. act as neutralising antibodies) and therefore prevent it from binding to its receptor. This is the mechanism of the biologic that targets the vascular endothelial growth factor
3.Delivering targeted radiotherapy (radioimmunotherapy) or chemotherapy as conjugated antibodies, minimising effect on healthy cells.
4.Immunotherapy → act as marker for a cell that should be removed.
•Cytotoxic response (ADCC)
•Complement activation (CDCC)
•Blocking inhibition of the immune response
•Linking cancer and cytotoxic T cells (bi-specific antibodies)
Receptor Tyrosine Kinase & Kinase
Inhibitors
Biologics prevent ligand binding to the extracellular domain of the receptor
Small molecule inhibitors bind to ATP binding site of the receptor tyrosine kinase domain or those of intracellular kinases
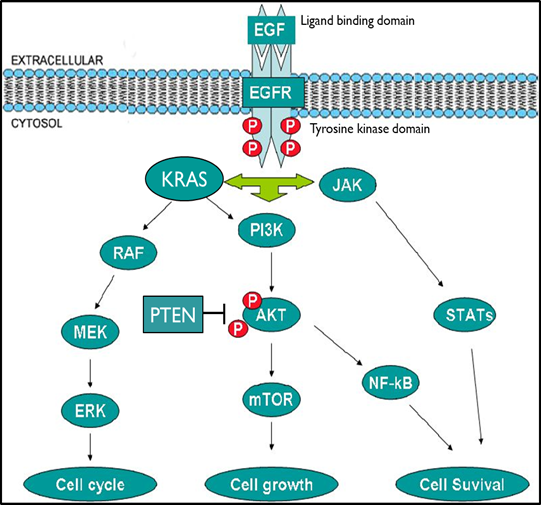
Good targets for therapy as they often contribute significantly to cancers development. Better targets if they are overexpressed within the disease process or have limited disease expression pattern.
Biologics are used to interfere with the ligand binding to the extracellular domain of receptors tyrosine kinases. Small molecule inhibitors are designed to target the kinase domain of the receptors or intracellular kinases.
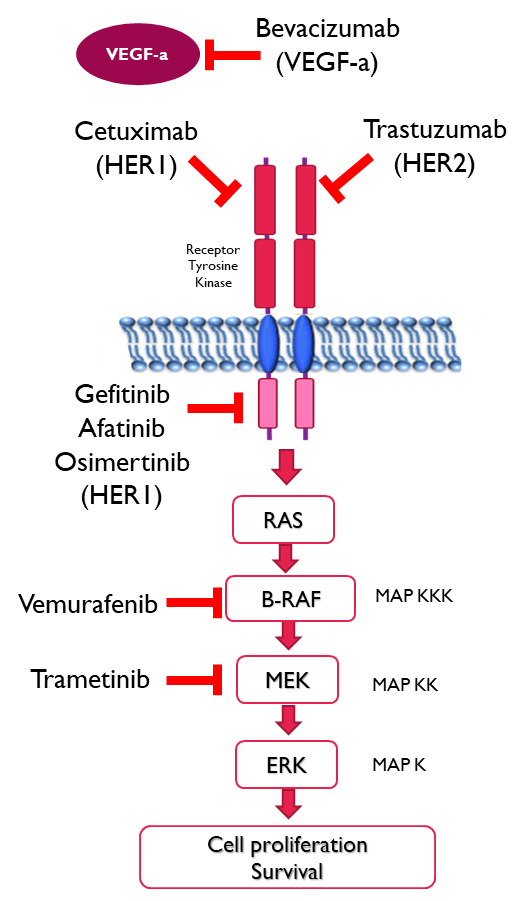
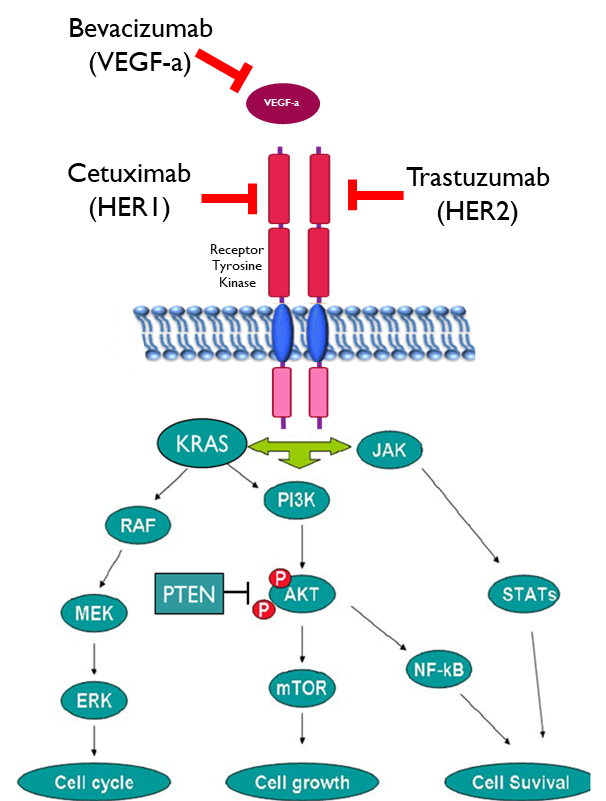
Biologics Targeting
Receptor Tyrosine Kinases
•Target the receptor
•Used when there is overexpression of the receptor
•Cetuximab is a chimeric (mouse/human) Mab that binds to and inhibits EGFR (HER1)
•Trastuzumab is a humanised Mab targeting HER2
•Target the growth factor
•Bevacizumab binds to vascular endothelial growth factor (VEGF-a) preventing it from binding to its receptor
•Both approached block the signalling pathway
•N.B. Only useful in tumours that do not have downstream mutations
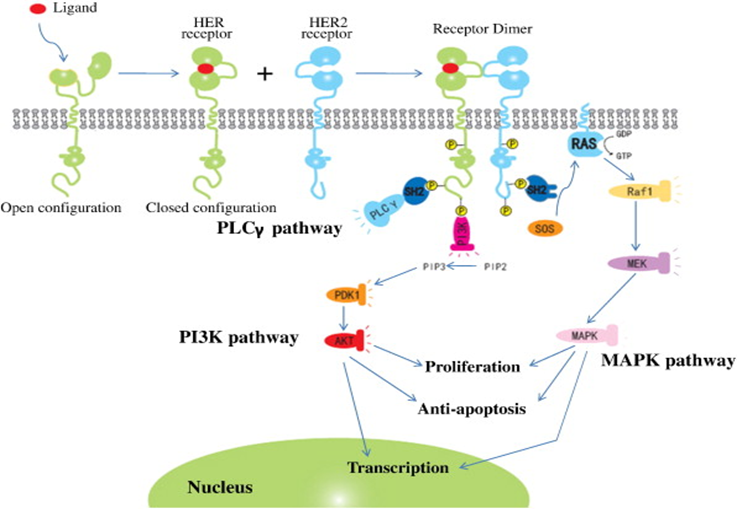
Trastuzumab
HER2 is a member of the EGFR family and can forms a dimer with other EGFRs. Overexpression of this protein is found in a percentage of breast cancer and also in some gastric cancers.
•Forms an active conformation without ligand binding
•Forms homodimers in the absence of ligand
Trastuzumab mechanism of action
1.Inhibition of tumour cell proliferation – cytostatic activity
•Binding of Trastuzumab to HER2 prevents it dimerising and signalling
•Stimulates HER2 receptor downregulation by promoting its endocytosis and proteolysis limiting the signal for further cell growth
2.Facilitation of immune function (cytotoxic)
•Trastuzumab binding promotes antibody-dependent cellular cytotoxicity (ADCC) by activating natural killer cells and macrophages
3.Prevention of constitutive activation
•Prevents shedding of the extracellular portion of the receptor by inhibiting the action of metalloproteinases which cleave it
Overall these effects prevent HER2 signalling and limit further cell division
Possible Mechanisms Of Resistance To
Receptor Tyrosine Kinase Targeting Antibodies
1.Mutations in downstream signalling proteins bypass the receptor inhibition, in other words the pathway works regardless of the presence of the receptor.
2.Masking of the MAb binding epitope of the receptor by membrane-associated glycoprotein preventing the biologic binding to it → basically cancer cells can mask the binding site by covering it with glycoproteins
3.Impairment of MAb-induced receptor endocytic downregulation → normally when the antibody makes a complex with the receptor it is pulled into the cell and is destroyed preventing further signalling of the receptor. Sometimes this fails to happen so the signalling will continue.
4.Overexpression of receptor ligands
5. Neutralising antibodies to the MAb (anti drug antibodies)
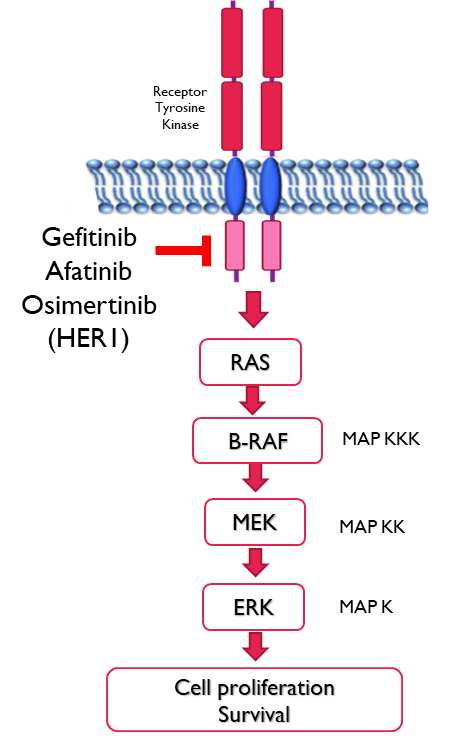
HER1 Receptor Tyrosine kinase inhibitors
•Gefitinib - 1st generation reversible binding to wildtype and mutated receptors inhibiting phosphorylation
•
•Afatinib - 2nd generation irreversible binding to catalytic domain in wildtype and mutated receptors
Since both of these bind to mutated and wild type they have considerable side effects such inflammation of the mouth, diarrhoea, and rashes that often require dose reductions. Furthermore resistance can also develop. Most common cause of resistance is the change of single amino acid within the receptor, preventing the inhibitors from binding.
•Osimertinib - 3rd generation irreversible binding to receptors with the resistance mutation T790M → reduced side effects as it doesn’t bind to wildtype receptors.
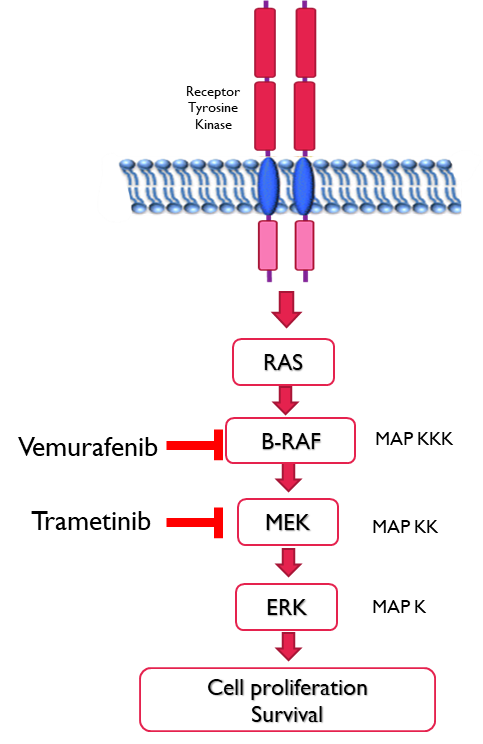
Kinase Inhibitors
•Vemurafenib
•B-RAF kinase inhibitor with selectivity for the ATP binding site with the V600E mutation (which is constitutively active) inhibiting the kinase activity
•CYP1A2 inhibitor; CYP3A4 inducer therefore care needs to be taken with other medications taken at the same time.
•
•Trametinib
•Reversible MEK1 and MEK2 kinase inhibitor
•Binds to S218 in the activation loop of the enzyme preventing phosphorylation by RAF at this residue
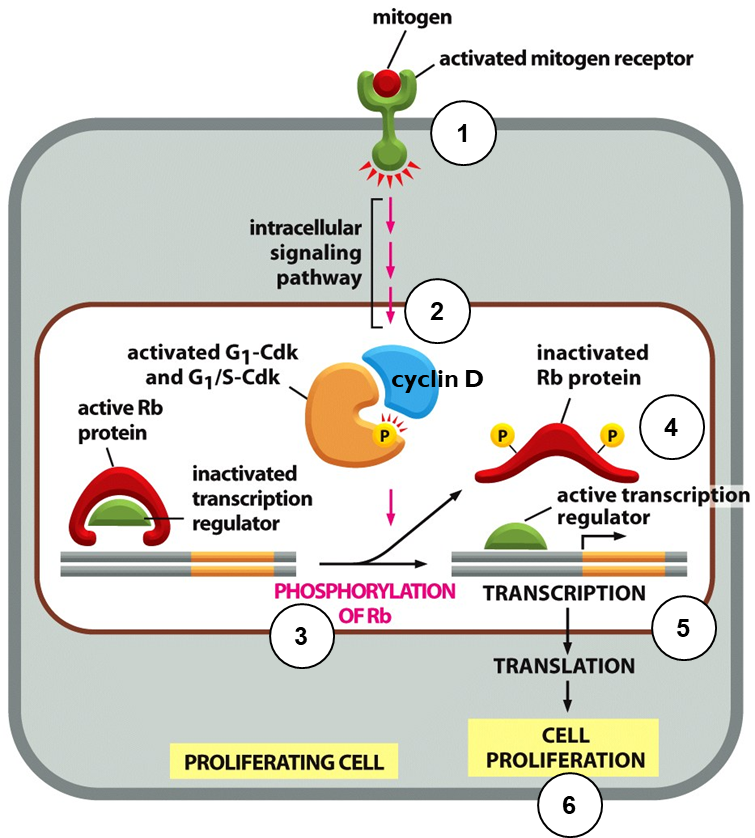
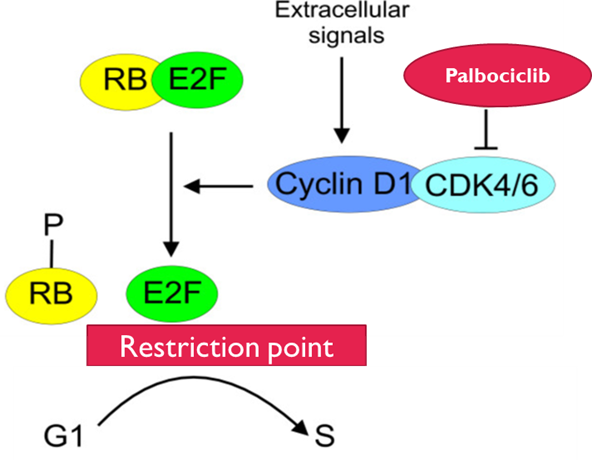
Recap: The G1/S Transition
Entry Into Cell Cycle
Retinoblastoma protein inhibits the E2F family of transcription factors when hypo-phosphorylated
1.Mitogens bind to cell surface receptors and activate downstream signalling pathways
2.Expression of cyclin D and activation of CDK4 and CDK6
3.CDK4/6 phosphorylates Rb
4.Phosphorylated Rb cannot bind to E2F transcription factor, and E2F is activated
5.G1/S genes are transcribed and translated
6.Cell passes through G1/S checkpoint – the restriction point
CDK4 / 6 Inhibitors
•RB is the only target of CDK4/cyclin D1
•RB must be phosphorylated to enable the cell to pass the restriction point
•Cdk4 is ubiquitous, Cdk6 is tissue specific
•Cdk4 is inhibited by p16 / p21/ p27
•p21 is induced by p53
•Overexpression of Cyclin D1, loss of expression of p16 or p53 occur in some cancers
•Palbociclib
•Reversible inhibitor of CDK4 and CDK6
•Cell cycle arrest and a block to cell cycle progression
Cancer Immunotherapy
•Immunotherapy - Use of an individuals immune system to target their disease
•Rituximab (chimeric) and Ofatumumab (human)
•Directed against CD20 expressed on B cells
•Recruits macrophages, natural killer cells, complement proteins for cell destruction
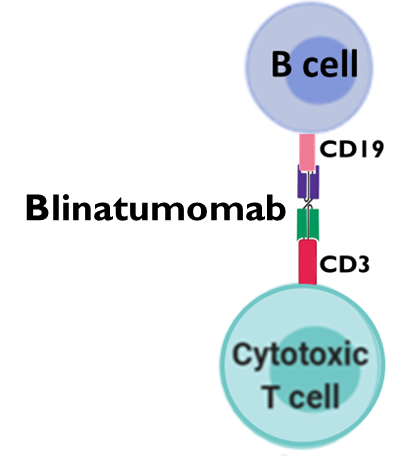
•Blinatumomab (human)
•A bi-specific antibody composed of 2 variable regions which recognise CD19 (B cell) and CD3 (CD8+ T cell )
•Tethers the cancerous B cells to cytotoxic T cells promoting their destruction
Cancer Immunotherapy
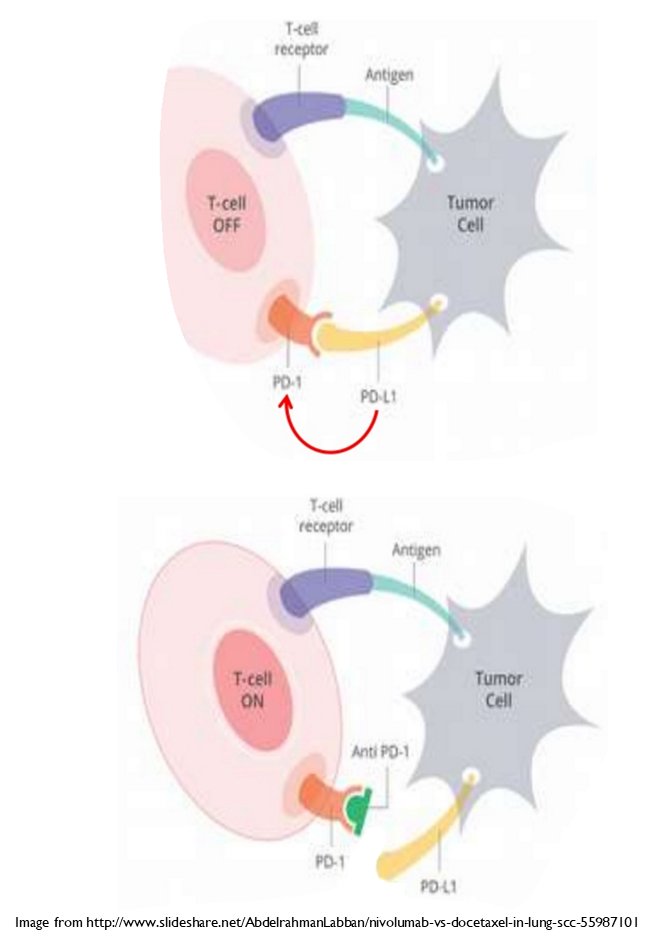
•Checkpoint inhibitors target proteins that normally downregulate immune cell function
•Ipilimumab (human) binds to CTLA-4.
•CTLA-4 is an inhibitory protein expressed on the surface of activated T cells. It binds to C80/86 to downregulate T cell function
•Nivolumab (human) binds to PD-1 preventing PD-1L binding to it
•Atezolizumab (humanised) binds to PD-1L
•PD-1 limits the activation of immune cells. When PD-1L binds to PD-1 it triggers prompt apoptosis of the T cell
•Checkpoint inhibitors act to increase the immune response.
Poly ADP Ribose Polymerase (PARP) Inhibitors
•Poly ADP ribose polymerase (PARP) is involved in the repair of single strand breaks in DNA
•DNA repair pathways are often upregulated in cancer cells (a mechanism of resistance)
•Poly ADP ribose polymerase (PARP) inhibitors e.g. Olaparib and Niraparib
•Inhibit repair of single strand breaks in DNA
•Traps PARP on the DNA causing double strand breaks and stalling of the replication folk during DNA replication
•Used:
•To potentiate the effect of chemotherapy used to induce DNA strand breaks
•As monotherapy in tumours with impaired homologous recombination e.g. BRCA mutation containing tumours.
•To extend time between conventional chemotherapy for relapsed cancer