Enthalpy change (3.1.4.1)
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
What are enthalpy and enthalpy change?
Enthalpy - measure of the heat energy of a substance
Enthalpy change (ΔH)- change in heat energy measured under conditions at constant pressure - usually in units of kJmol-1
What are endothermic and exothermic reactions?
Bond breaking is endothermic and bond making is exothermic. This is because to break bonds, energy has to be taken in from the surroundings and, to form bonds, energy is given out.
An endothermic reaction is one in which more energy is taken in to break the bonds than given out to make new bonds.
An exothermic reaction is one in which more energy is given out forming the new bonds than taken in to break the bonds.
In an endothermic reaction, energy is transferred from the surroundings to the system and the products thus have more energy than the reactants - positive enthalpy change:
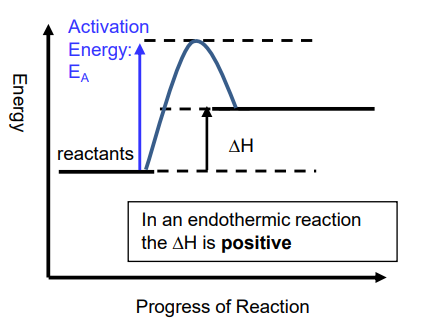
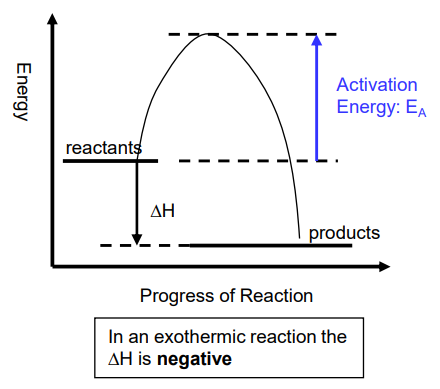
In an exothermic reaction, energy is transferred from the system to the surroundings and the products thus have less energy than the surroundings - negative enthalpy change:
What are standard conditions?
Standard conditions are:
Pressure - 100 kPa
Temperature - 298 K (25°C or room temperature)
Standard conditions are represented by a plimsoll - ⦵
What is standard enthalpy change of reaction?
Standard enthalpy change of reaction (ΔrH⦵) - The enthalpy change for a reaction with quantities shown in the chemical equation. The value should always be quoted along with the equation.
What is standard enthalpy change of formation?
Standard enthalpy change of formation (ΔfH⦵) - The enthalpy change when 1 mole of a substance is formed from its constituent elements with all reactants and products in standard states under standard conditions.
E.g. Mg(s) + Cl2(g) → MgCl2(s)
The enthalpy of formation of an element is 0 kJmol-1 as no bonds are broken or formed at its standard state
What is standard enthalpy change of combustion?
Standard enthalpy change of combustion (ΔcH⦵) - The enthalpy change when 1 mole of a substance is completely burned in oxygen with all reactants and products in standard states under standard conditions.
E.g. CH4(g) + 2O2(g) → CO2(g) + 2H2O(l)
If incomplete combustion occurs, it will be less exothermic than complete combustion
What is standard enthalpy change of neutralisation?
Standard enthalpy change of neutralisation (ΔneutH⦵) - The enthalpy change when 1 mole of water is formed between an acid and alkali under standard conditions.