chapter 1 -ionisation energy
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
what is a period?
a row in the periodic table
elements in the same period have their valence electrons in the same shell
what is a group?
a column in the periodic table
elements in the same group have their electrons in similar orbitals
they often have similar properties
what is the s block?
the 2 leftmost columns make up the s block
this is called the s block because elements in it have all their valence electrons in s orbitals
helium is also an s block element

what is the p block?
the 6 rightmost columns make up the p block
this is called the p block because elements in this block have a partially filled p sub-shell

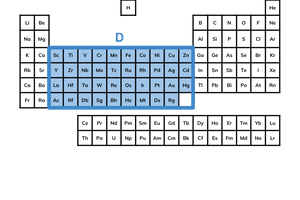
what is the d block?
the d block lies between the s block and p block
elements in the d block have a partially filled sub-shell
elements in this block are often called transition metals
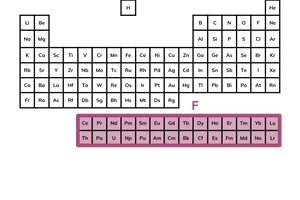
what is the f block?
this block is detached along the bottom of the periodic table
elements in the f block have a partially filled f sub-shell
these elements are referred to as the lanthanides and the actinides (top row and bottom row respectively)
what is ionisation energy?
a specific amount of energy needed to remove an electron from an atom or ion
what is the first ionisation energy?
energy required to remove an electron from every atom in a mole of atomic gas, to produce a mole of uni-positive gaseous ions
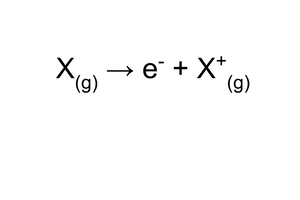
second ionisation energy?
energy required to remove an electron from every ion in a mole of uni-positive gaseous ions, to produce a mole of di-positive gaseous ions
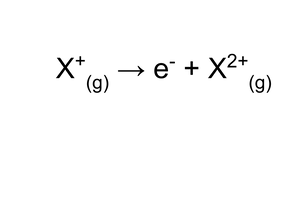
third ionisation energy?
energy required to remove an electron from every ion in a mole of di-positive gaseous ions, to produce a mole of tri-positive gaseous ions
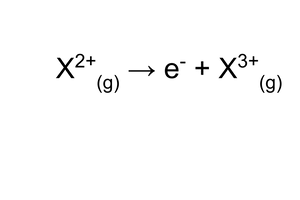
nth ionisation energy?
overall we can form an equation for each successive ionisation energy
where the value for n, is the value of the charge on the ion in the products
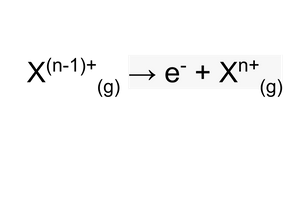
factors affecting ionisation energies?
nuclear charge
distance from the nucleus
shielding
nuclear charge?
the greater the number of protons in the nucleus, the greater the attraction of the electron to the nucleus
a greater attraction of electron means more energy is needed to remove the electron
this means the ionisation energy is greater
distance from the nucleus?
electrostatic attraction decreases sharply with distance
this means that less energy is needed to remove the electrons which are further away
so, as distance increases, ionisation energy decreases
shielding?
the greater the number of electrons between the nucleus and the outer electrons, the lower the effective nuclear charge
this is because the positive charge felt by the electron is reduced by the electrons in between
therefore, the greater number of electrons, the lower the ionisation energy
why does first ionisation increase across a period?
because the atomic radius decreases as the proton number increases across a period, so electrons are more attracted to the nucleus
the electron is closer to the nucleus, and so experiences a greater attraction
why does first ionisation energy decrease down a group?
because the atomic radius increases because the electron is further from the nucleus, and so experiences a lower attraction
there are also more electrons between the nucleus and outer electrons, due to greater shielding