OChem 2 - Exam 3
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Grignard/organolithium
Ketones & Aldehydes - require 1 equivalent
Esters & Acid Chlorides - require 2 equivalents
Acid Protons (OH) - require 1 equivalent
Carboxylic Acids (Grignard) - require 1 equivalent and will only deprotonate
Carboxylic Acids (Organolithium) - requires 2 equivalents
Aldehyde
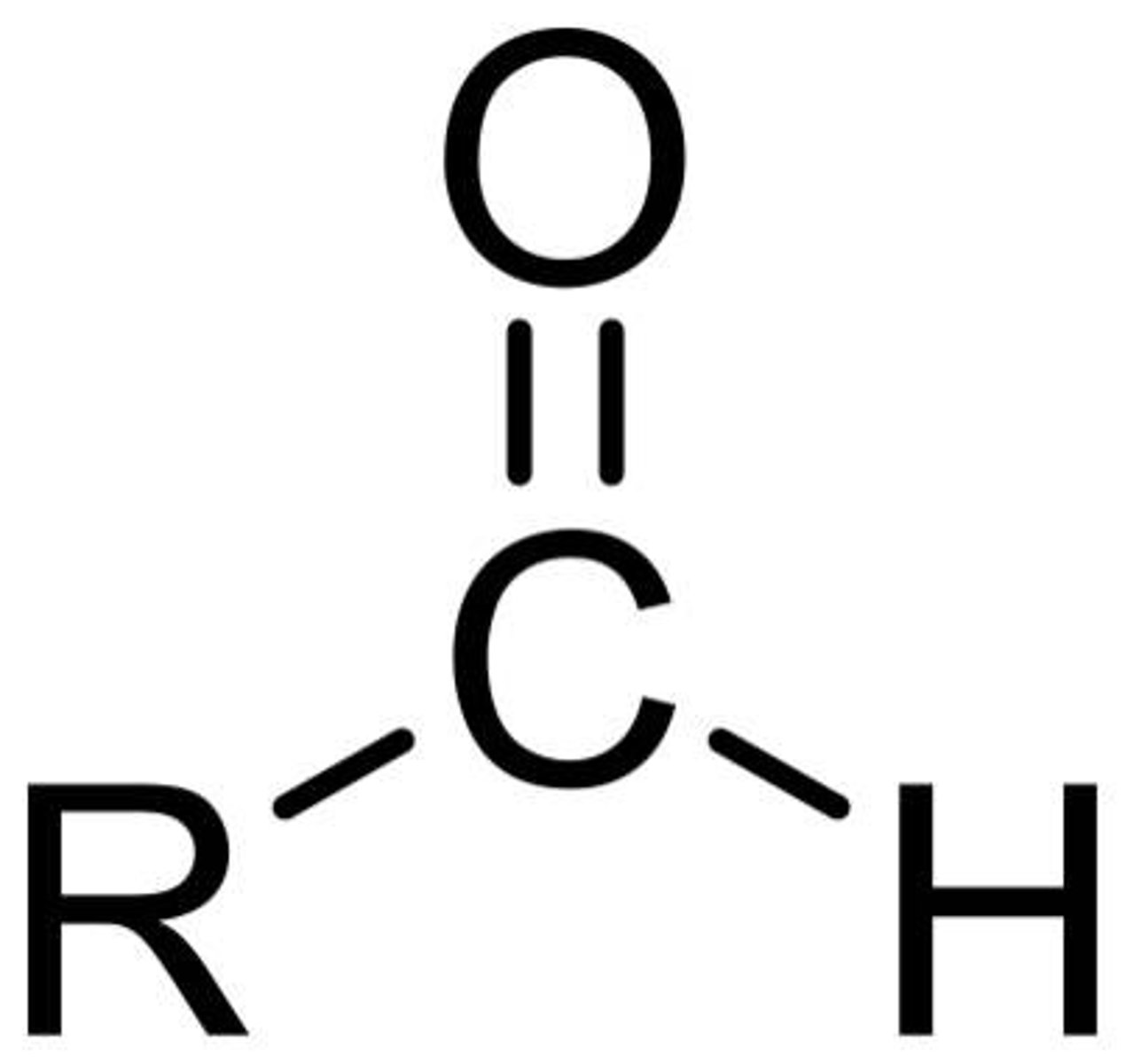
Ester
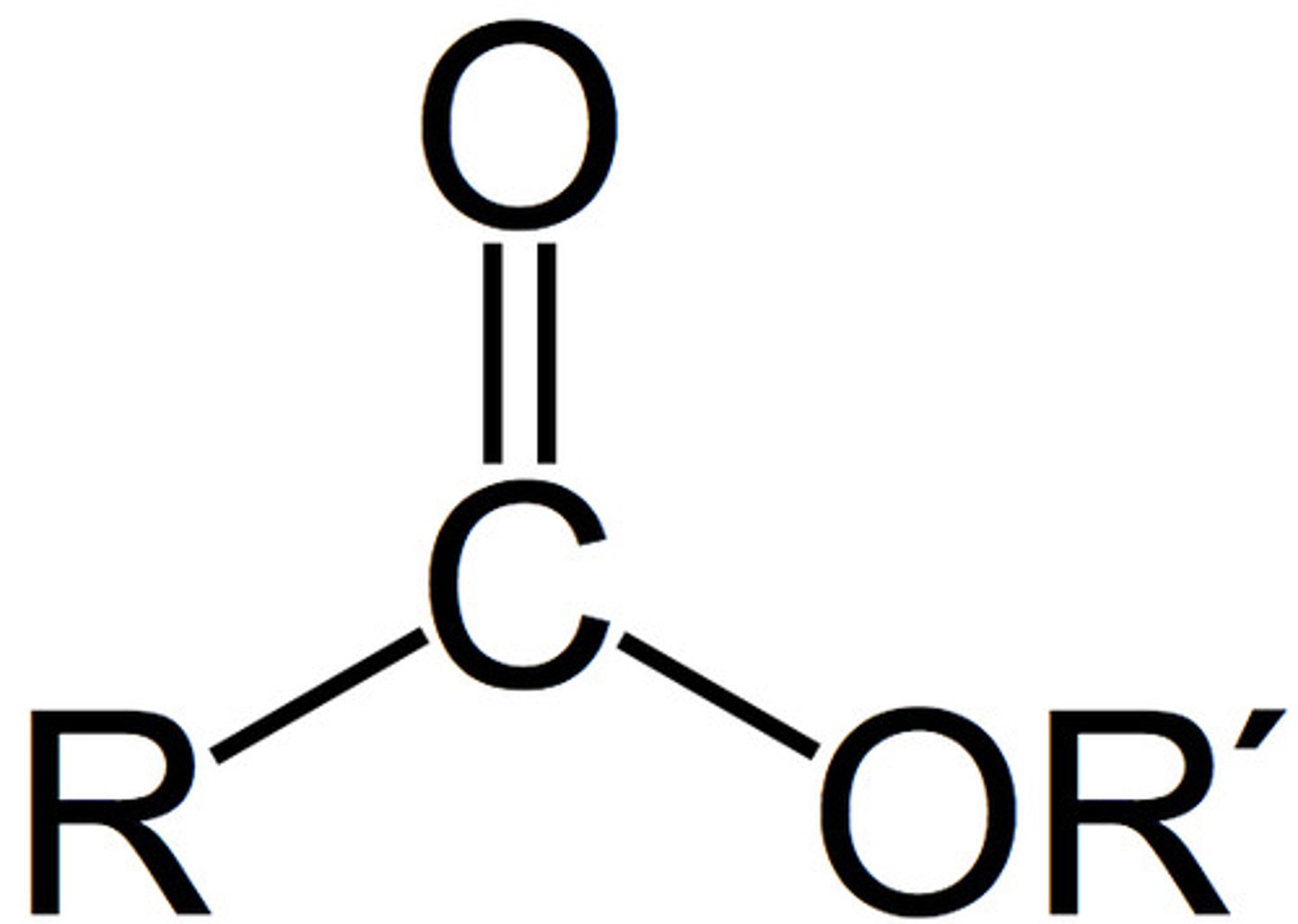
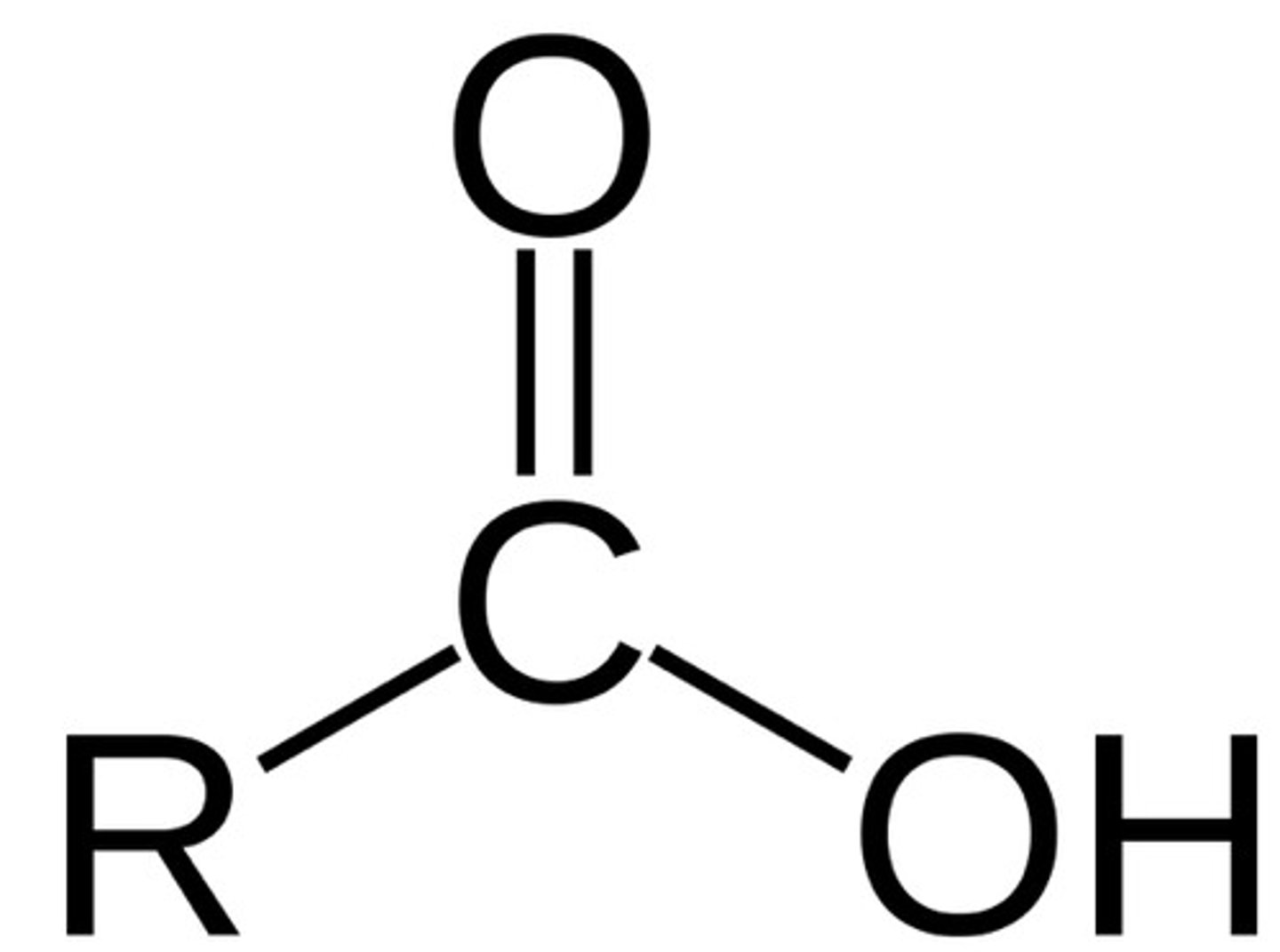
Carboxylic Acid
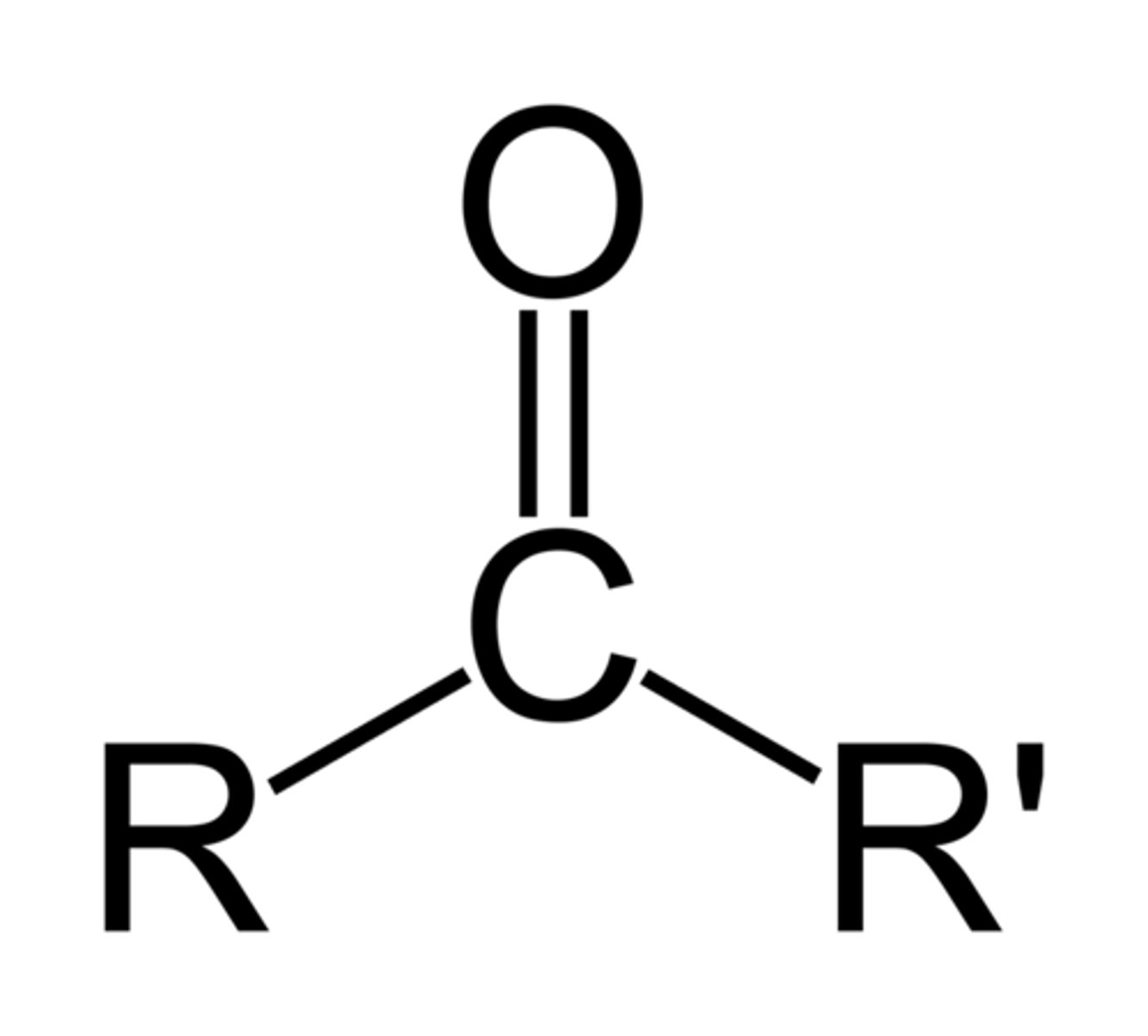
Ketone
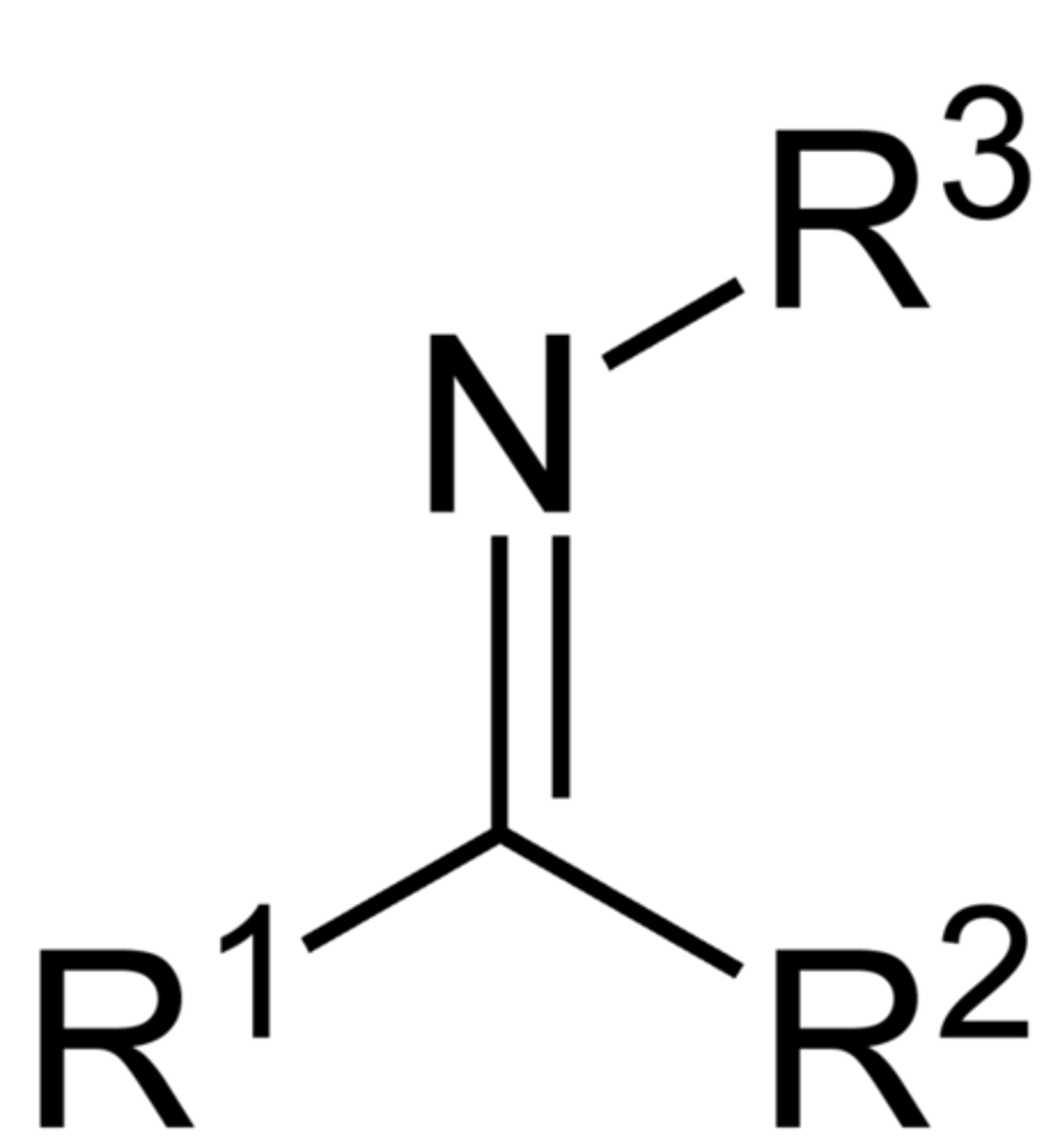
Imine
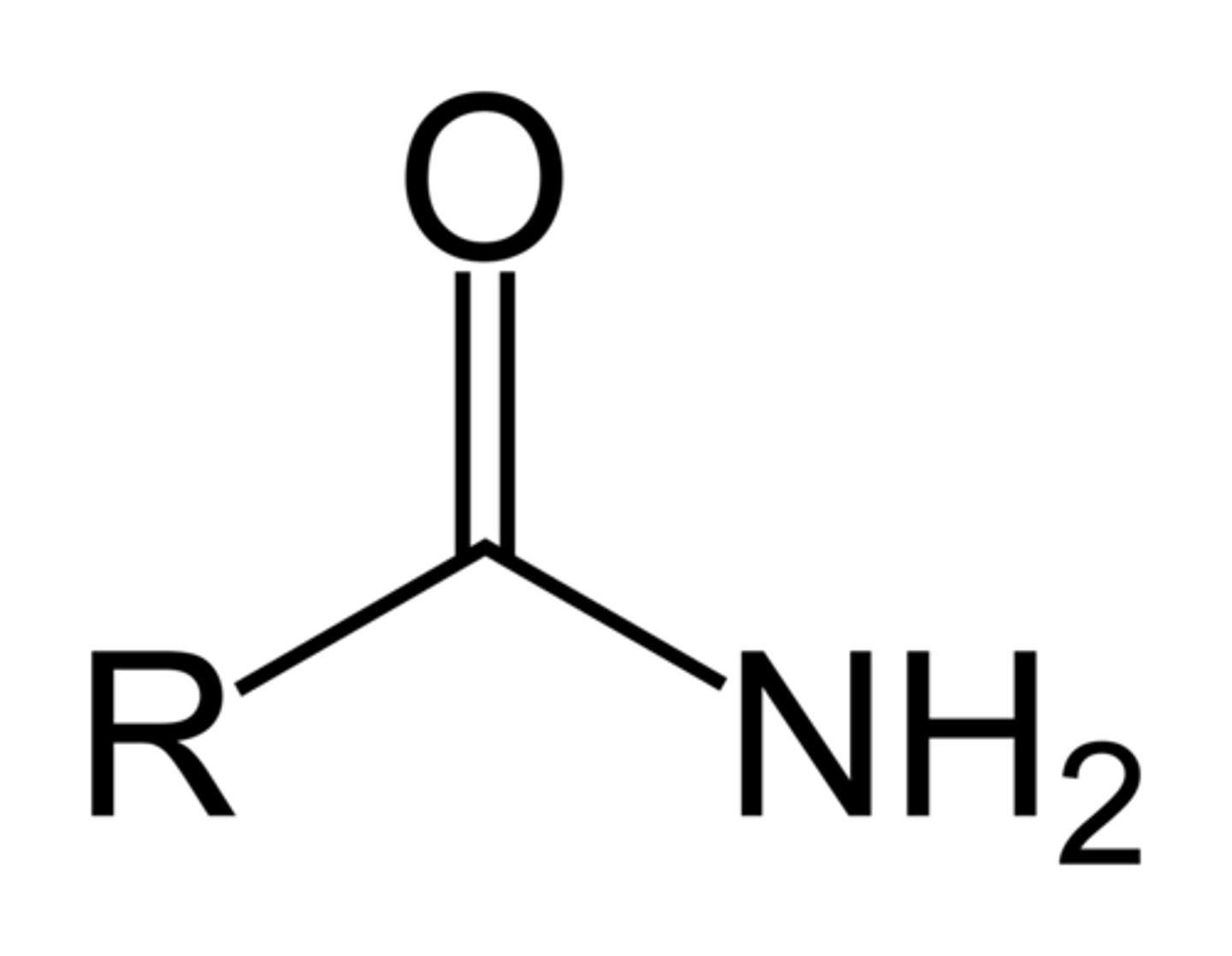
Amide
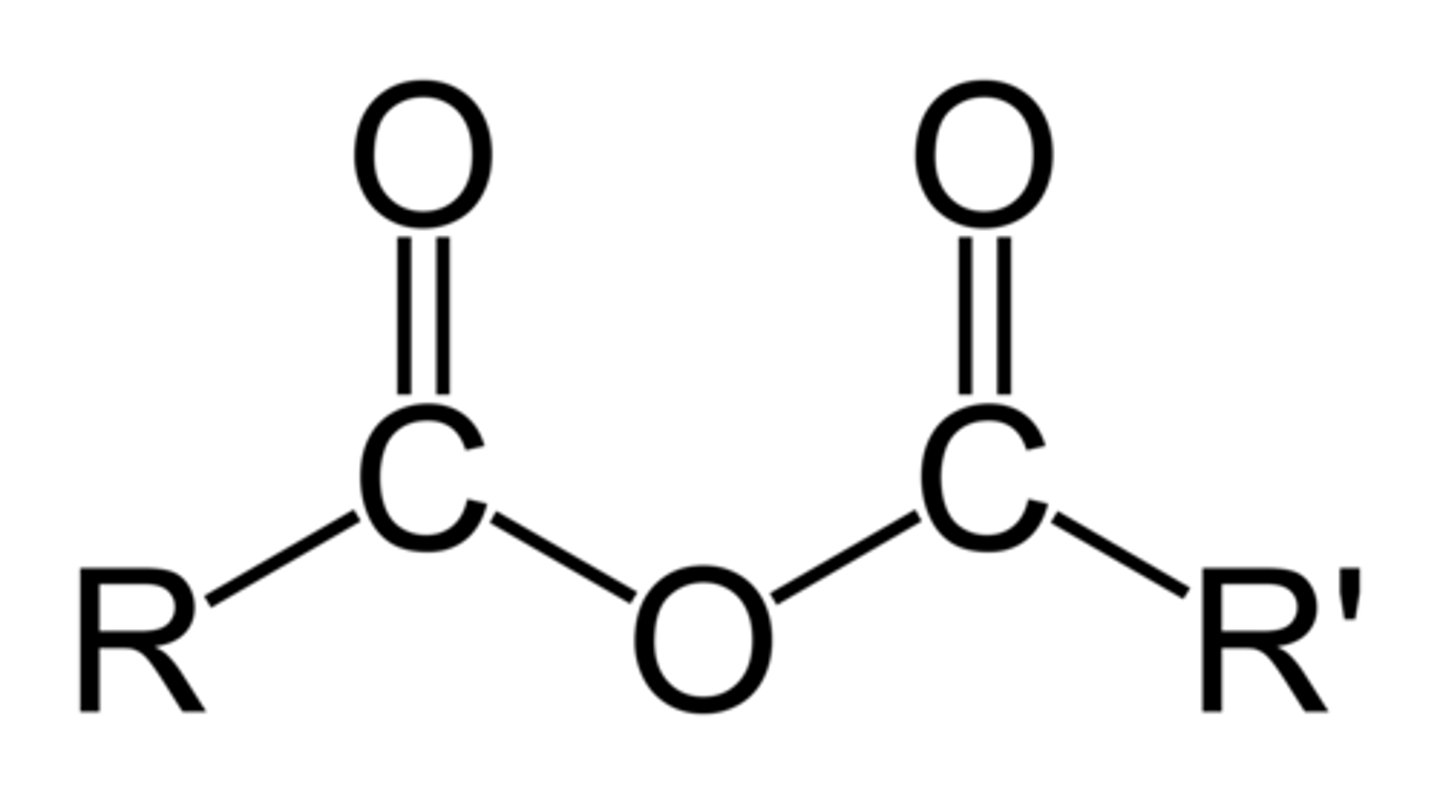
Anhydride
Substitution of Carboxylic Acid with Grignard
Will not undergo substitution, only deprotonation
NaBH4 cannot
Cannot reduce carboxylic acids, only deprotonate
Carboxylic Acid to Acid Chloride
SOCl2
Acid Chloride to Carboxylic Acid
H20, pyridine
Carboxylic acid to first degree alcohol
1.) Excess LiAlH4 2.) H3O+
Acid Chloride to Anhydride
Carboxylic acid and H+
Carboxylic Acid to Anhydride
Acid Chloride, pyridine
Anhydride to Ester
R-OH
Ester to Carboxylic acid
H+, H20
Carboxylic Acid to Ester
R-OH, H+
Ester to Amide
excess R-NH2, Heat
Amide to Carboxylic Acid
[H+], H2O, Heat
Acid Chloride to Ester
R-OH, pydridine
Acid Chloride to Amide
Excess NH3 (2 eq)
Ester to Aldehyde
1.)DIBAL-H, cold -78c
2.)H3O+
Acid Chloride to Aldehyde
1.) LiAl(OtBu)3H
2.)H3O+
1st degree alcohol to Carboxylic Acid
Na2CrO7 (same as CrO3)
H2SO4 / H+, H2O
Ester to 1st degree alcohol
1.) Excess LiAlH4
2.) H3O+
Amide to Amine
1.) Excess LiAlH4
2.) H+, H2O
NaBH4
Reduces aldehydes to primary alcohols
Reduces ketones to secondary alcohols
Doesn’t react with anything less reactive than ketone (Carboxylic acids, amides, Esters)
TBSCl (used with Imidazole or NEt3)
Protective group for hydroxyl and amine groups
TBAF
Gets rid of TBSCl protective group
LiAlH4
Reduces aldehydes to 1st degree alcohols,
ketones to 2nd degree alcohols
esters and carboxylic acids to 1st degree alcohols
Reduces amides and nitriles to amines
Acid Chloride to Aldehyde
LiAlH(OtBu)3
H+ workup
Ester to Aldehyde
DIBAL (1eq)
-78 C
NEt3
strong bulky base
Carboxylic Acid to Amide
DCC
R-NH2
CrO3, pyridine
oxidize primary alcohols to aldehydes,
secondary alcohols to ketones.
When acid is present: primary alcohols and aldehydes are oxidized to carboxylic acids.
KMnO4
primary alcohols and aldehydes to carboxylic acids
secondary alcohols to ketones
Reactivity of Carbonyl Function Groups
Acid Chloride>Anhydride> Aldehyde>Ketone>Ester>Carboxylic Acid>Amide
Acid Chloride to Ketone
CuLiCH4 (2 eq)
Anhydride to amide
NH3 (excess)
Ester to 3rd Degree alcohol
R—MgBr
H+ workup
Ester to 1st degree alcohol
excess LiAlH4
Anhydride to Ester
R—OH
Ketone to 2nd Degree Alcohol
LiAlH4
or
NaBH4
Ketone to 3rd degree alcohol
CrO3, H+, H20
2nd degree alcohol to Ketone
PCC or CrO3, H+, H2O
Acid Chloride to Ketone
(CH3)2 CuLi
Carboxylic Acid to Ketone
CH3Li (2 eq)
R-CN to Ketone
CH3-MgBr (1 eq)
R-CN to Amide
H2O
amide to R-CN
SOCl2
1st degree alcohol to Aldehyde
PCC
Aldehyde to 1st degree Alcohol
LiAlH4
R-CN to Aldehyde
DIBAL (1eq)
Aldehyde to Carboxylic Acid
H3O+
Ester to Carboxylic Acid
H20, H+