Module 4: Carbonyl Group
0.0(0)
Card Sorting
1/43
Earn XP
Description and Tags
Last updated 1:56 PM on 3/8/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
1
New cards
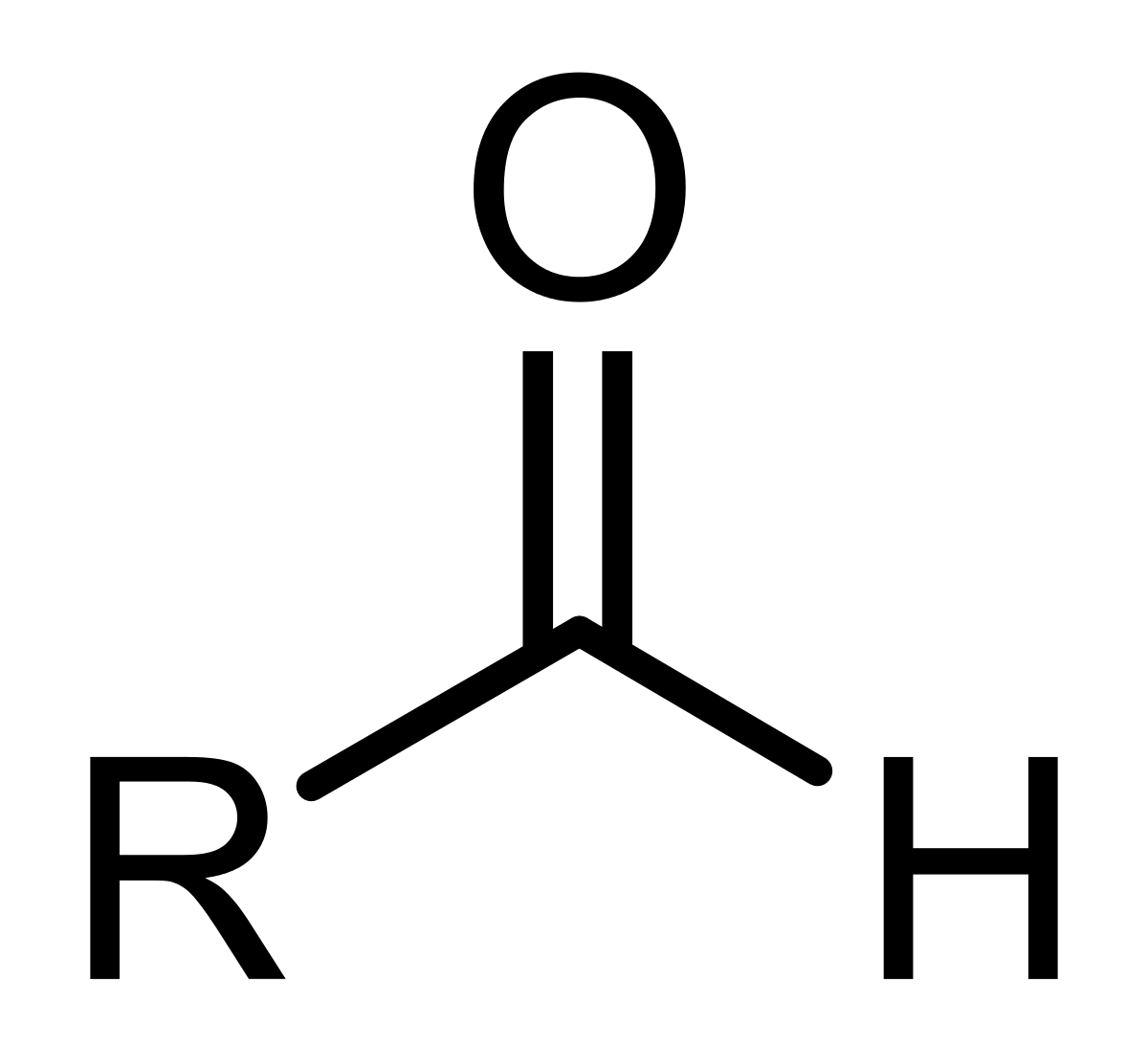
Has a general structure of R-CHO
Aldehyde
2
New cards
Attached to a Carbon with a Carbonyl (C=O) bond
Aldehyde
3
New cards
Special feature of aldehyde
It has Hydrogen
4
New cards
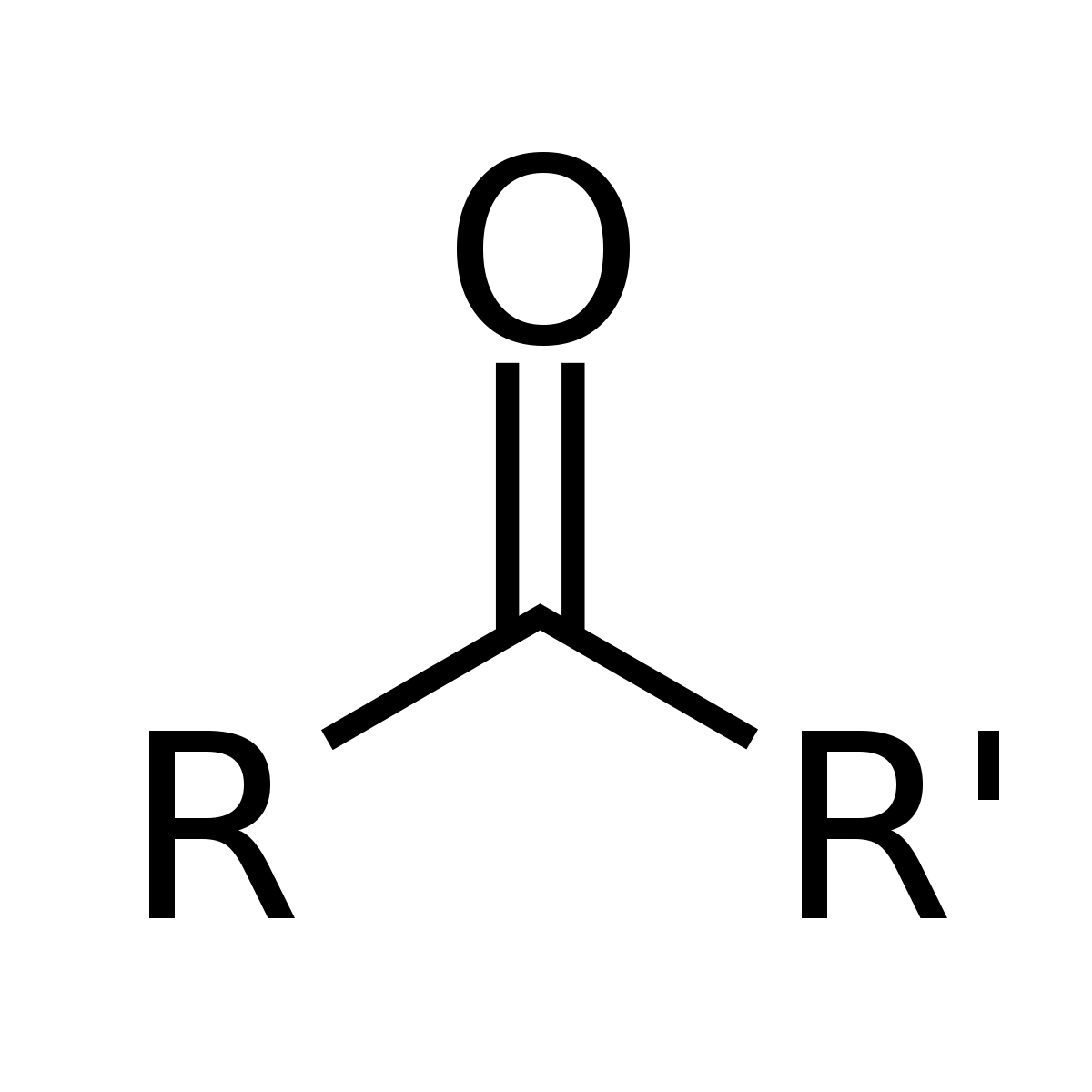
General structure R-CO-R
Ketone
5
New cards
Special feature of Ketone
Alkyl, alkane, or any Carbon-containing group
6
New cards
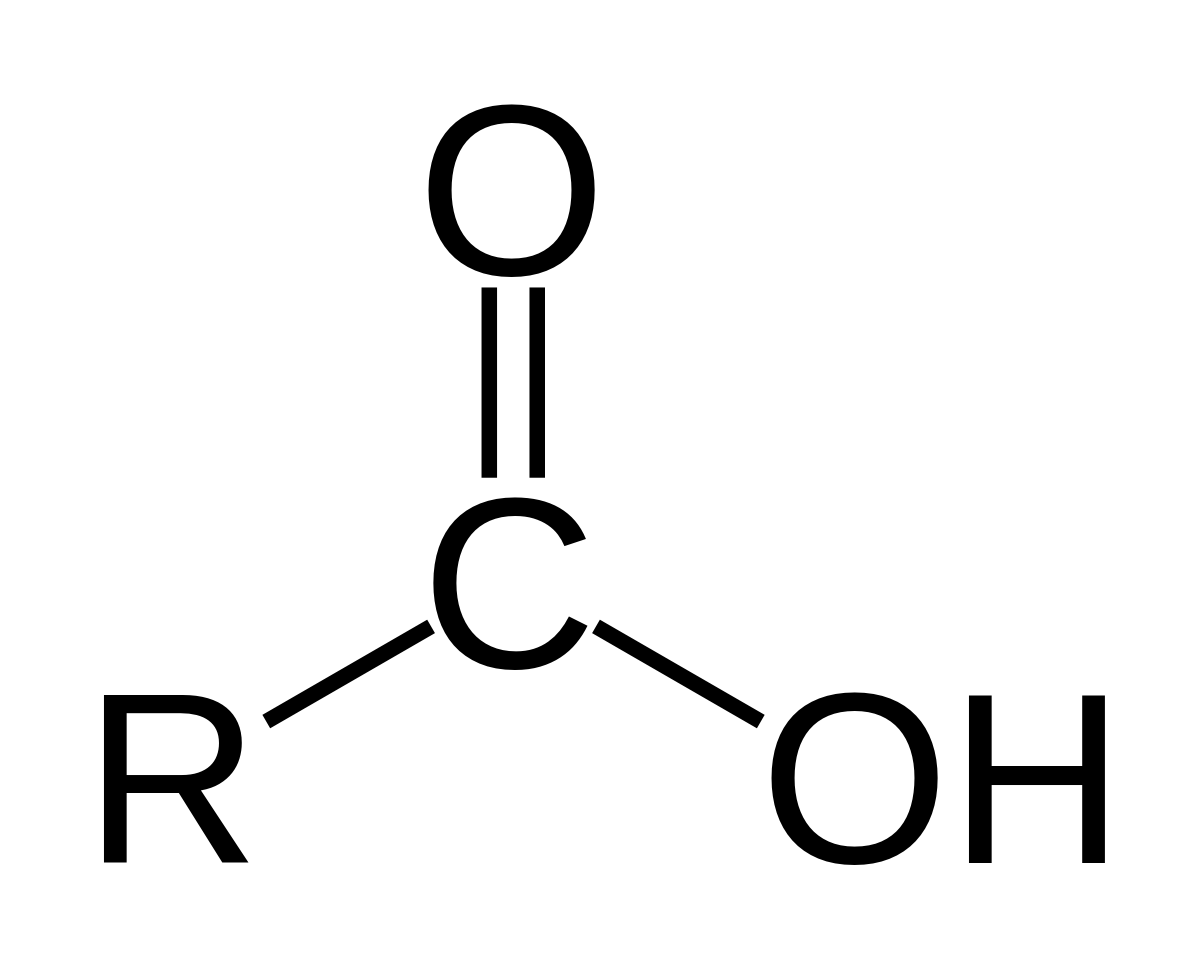
Has a general structure of R-COOH
Carboxylic acid
7
New cards
Special feature of Caroxylic acid
\
Hydroxyl (-OH) group
Hydroxyl (-OH) group
8
New cards
Carboxylic acid derivates
Ester, Anhydride, Amide
9
New cards
Similarities of Ester, anhydride, & amide
Carbonyl
10
New cards
The study of aldehydes and ketones help in the discussion of _______ since sugars contain either an aldehyde or ketone group.
Carbohydrates
11
New cards
The most common simple sugar in the body that is an aldehyde that has also several -OH groups.
Glucose
12
New cards
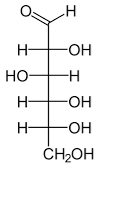
Name the structure
D-glucose
13
New cards
Are the most important organic compounds that show appreciable acidity.
Carboxylic acid
14
New cards
Carboxylic acids contain a carboxyl group (-COOH), attached to either an alkyl group or ____ group (Ar-COOH)
Aryl
15
New cards
True or False
\
Carboxylic acids are very polar compounds.
\
Carboxylic acids are very polar compounds.
True
16
New cards
True or False
\
Carboxylic acids form hydrogen bonds with other polar compounds like water.
\
Carboxylic acids form hydrogen bonds with other polar compounds like water.
True
17
New cards
Other term for Aryl groups
Aromatic groups
18
New cards
Derivatives of carboxylic acids that are NOT FOUND IN NATURE because of their reactivity
Acid chlorides & acid anhydrides
19
New cards
Names by identifying ***-ic*** (acid) to ***-yl*** (chloride) or the carboxylic acid ending with the carbonyl.
Acid chlorides
20
New cards
Has a general structure R-OCCl
Acid chlorides
21
New cards
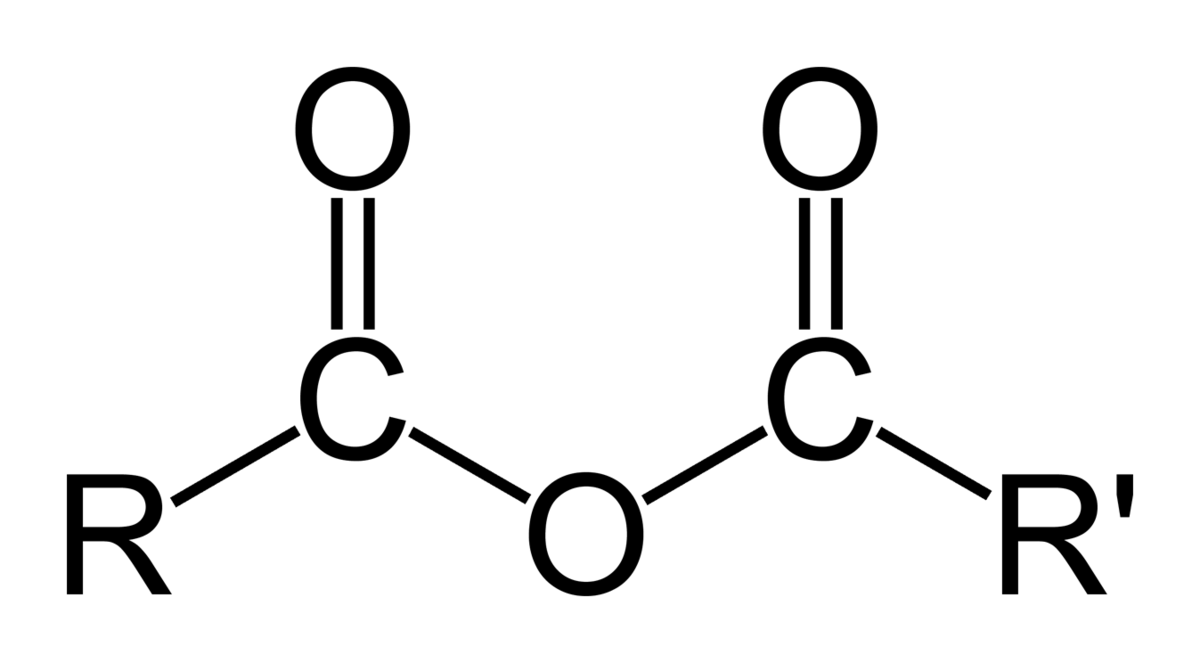
Has a general structure R-CO-O-CO-R
Anhydride
22
New cards
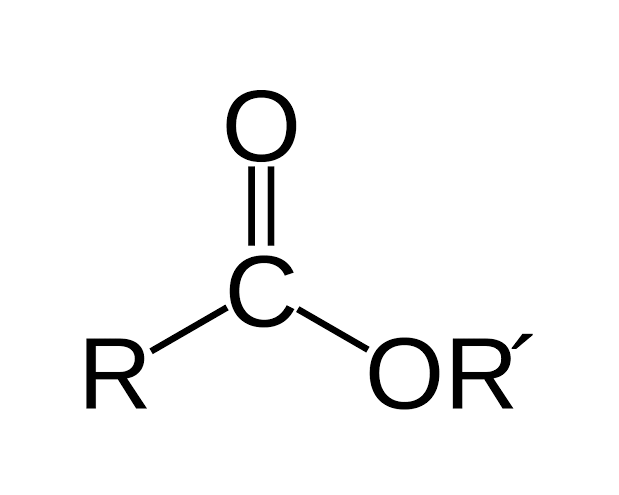
Has a general structure R-COOR
Ester
23
New cards
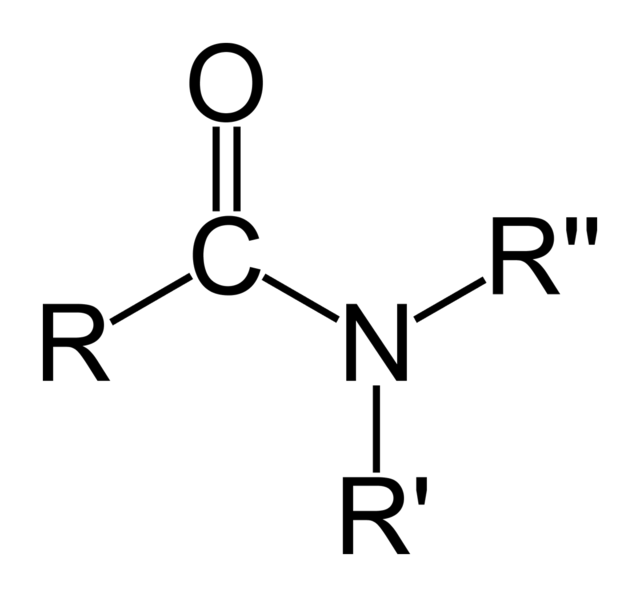
Has a general structure R-CO-NH2
Amide
24
New cards
Hs a functional group characterized by a carbonyl group (C=O) linked to a nitrogen atom (N) or any organic compound derived by the replacement of a hydroxyl group by an amino group.
Amide
25
New cards
The most stable of all the carbonyl functional groups.
Amide
26
New cards
Compounds in whicha hydrogen atom in nitrogen ftom ammonia or an amine is replaced by a metal cation are also known as amides or ________
Azanides
27
New cards
NH2- formula
Azanides
28
New cards
NH3 formula
Ammonia
29
New cards
NH4 formula
Ammonium
30
New cards
Organic compounds that have sweet, fragrant aromas and occur in flowers and fruits
Ester
31
New cards
True or False
\
Esters with high molecular weighs are solids. They do not vaporize readily; hence they are not pleasant-smelling.
\
Esters with high molecular weighs are solids. They do not vaporize readily; hence they are not pleasant-smelling.
True
32
New cards
Polyesters (synthetic fabrics) are substances with repeating ester groups and have very **low** molecular weights.
False - Low = High
33
New cards
The most abundant esters in our body
Triglycerides
34
New cards
Solid triglycerides from humans/animals are called
Fats
35
New cards
Liquid triglycerides from plants are called
Oils
36
New cards
True or False
\
Aldehydes are compounds that have only carbon and hydrogen atoms bonded to the carbonyl group.
\
Aldehydes are compounds that have only carbon and hydrogen atoms bonded to the carbonyl group.
True
37
New cards
True or False
\
Carboxylic acid, ester, and amide are compounds that contain a **electropositive** atom bonded to the carbonyl group.
\
Carboxylic acid, ester, and amide are compounds that contain a **electropositive** atom bonded to the carbonyl group.
False - positive = electronegative
38
New cards
True or False
\
Electronegative atoms are commonly **oxygen and nitrogen**
\
Electronegative atoms are commonly **oxygen and nitrogen**
True
39
New cards
True or False
\
The carbonyl carbon atom is **trigonal planar**, with angles of **130°**
\
The carbonyl carbon atom is **trigonal planar**, with angles of **130°**
False - 130° should be 120°
40
New cards
True or False
\
Carbonyl carbon atoms are sp² hybridize
\
Carbonyl carbon atoms are sp² hybridize
True
41
New cards
True or False
\
O is more electronegative than C, so the carbonyl group is **non-polar**.
\
O is more electronegative than C, so the carbonyl group is **non-polar**.
False - non-polar = polar
42
New cards
True or False
\
The carbonyl O is e- **poor** and the carbonyl is C e- **rich**
\
The carbonyl O is e- **poor** and the carbonyl is C e- **rich**
False - O is e- rich, C is e- poor
43
New cards
True or False
\
In acetaldehyde, C is **doubly bonded to O** and is **singly bonded to H**
\
In acetaldehyde, C is **doubly bonded to O** and is **singly bonded to H**
True
44
New cards
For acetone, C is **singly** bonded to 2 C's and **doubly** bonded to O
True