Organic Chemistry 3
1/94
Earn XP
Description and Tags
1-20 = Aromaticity and Benzene, 21-42 = Chirality, 43-65 = IR, Heterocycles 66-95
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
95 Terms
What is the aromatic stability of benzene?
Benzene is highly stable due to its conjugated π electron system.
It contains 6 π electrons which allows for electron delocalization.
Follows Huckel’s Rule, leading to low reactivity towards reactions that disrupt aromaticity.
What type of reactions does benzene primarily undergo?
Benzene primarily undergoes electrophilic aromatic substitution (EAS).
It avoids addition reactions that would break its aromatic nature.
What are common electrophilic substitutions that benzene undergoes?
Nitration: Introduction of a nitro group (NO2) using nitric acid in sulfuric acid.
Sulfonation: Introduction of a sulfonyl group (SO3H) using sulfur trioxide or concentrated sulfuric acid.
Halogenation: Adding halogens (Cl, Br) using halogenation agents like FeCl3.
Friedel-Crafts Alkylation: Formation of alkyl groups using alkyl halides and Lewis acids.
Friedel-Crafts Acylation: Formation of acyl groups (RC=O) using acyl chlorides with catalysts.
How do substituents on the benzene ring affect its reactivity?
Activating Groups (e.g., -OH, -NH2):
Increase electron density on the ring.
Make benzene more reactive toward electrophiles.
Deactivating Groups (e.g., -NO2, -CF3):
Decrease electron density.
Make benzene less reactive.
What is the role of resonance structures in benzene?
Resonance structures explain the stability of benzene.
They show electron delocalization over the ring.
Contributes to unique properties of benzene, such as lower reactivity.
Can benzene undergo addition reactions?
Benzene is generally resistant to addition reactions due to its aromaticity.
Under certain conditions (high temperature or strong reagents), it can form non-aromatic products.
What defines aromaticity in a compound?
Planar structure.
Cyclic nature with p orbitals.
Delocalized π electrons.
Follows Huckel’s rule (4n + 2 π electrons).
What is Electrophilic Aromatic Substitution?
A reaction where an electrophile replaces a hydrogen atom on a benzene ring.
Maintains aromaticity after the reaction.
Name some common functional groups associated with aromatic compounds.
A:
Toluene
Phenol
Aniline
Benzaldehyde
Benzoic acid
How does reactivity of benzene compare to alkenes?
Benzene is less reactive due to aromatic stability.
Primarily undergoes substitution reactions rather than addition.
What is halogenation in the context of aromatic compounds?
A type of electrophilic substitution where a halogen replaces a hydrogen atom.
It requires a catalyst (e.g., FeBr3 or AlCl3).
Describe the nitration process in aromatic compounds
Addition of a nitro group (NO2) to the benzene ring.
Reaction typically uses a mix of nitric acid and sulfuric acid.
What are activating substituents?
Groups that increase electron density on the benzene ring.
Make the ring more reactive toward electrophiles.
Examples include -OH, -NH2, -OCH3.
What are deactivating substituents?
Groups that decrease electron density on the benzene ring.
Make the ring less reactive toward electrophiles.
Examples include -NO2, -CF3, -COOH.
What does hydrogenation of cyclic compounds involve
The addition of hydrogen to unsaturated cyclic compounds.
Can lead to loss of aromaticity.
What is the importance of delocalized electrons in aromatic compounds
Contribute to stability of the benzene ring.
Allow for resonance, which distributes charge over the ring.
What is the significance of non-hindered π orbitals in aromatics?
Allow for effective overlap of p orbitals.
Help maintain aromatic stability.
Why are aromatic compounds resistant to addition reactions?
Adding to the structure would disrupt aromaticity and stability.
They favour substitution reactions instead.
How do substituents affect the rate of electrophilic substitution?
Activating groups increase reaction rate.
Deactivating groups decrease reaction rate.
What happens to a benzene ring during Electrophilic Substitution?
An electrophile attacks the ring, forming a carbocation.
The positive charge is delocalized throughout the ring.
What is chirality?
A property of molecules that lack mirror symmetry.
Chiral objects are not superimposable on their mirror images.
The mirror image is different
Example: left and right hands.
What is a chiral center?
A carbon atom bonded to four different substituents.
Responsible for a molecule’s chirality.
What are enantiomers?
Pairs of chiral molecules that are mirror images of each other.
Enantiomers can have very different biological effects.
A single enantiomer is termed enantiomerically pure.
What is the difference between a racemic mixture and a scalemic phase?
Racemic mixture:
Contains equal amounts of both enantiomers (1:1 ratio).
Scalemic phase:
Contains unequal amounts of the two enantiomers.
How do you identify chirality in organic molecules?
Look for chiral centers (usually carbon atoms).
Determine if the molecule can be superimposed on its mirror image.
Use methods like polarimetry and chiral chromatography.
Why is chirality important in pharmaceuticals?
Different enantiomers can have vastly different therapeutic effects.
One enantiomer may be therapeutic, while the other may be toxic or inactive.
The need for enantiomerically pure compounds in drug formulations.
What are examples of chiral molecules in nature?
Amino acids
(mainly found in the L-form).
Sugars
(mainly found in the D-form).
These chiral forms are crucial for biological functions.
What does the term asymmetric synthesis refer to?
A process to create chiral molecules with a predominant enantiomer.
Often involves reactions that favour one enantiomer over the other.
What is the significance of optical activity in chiral molecules?
The ability of chiral compounds to rotate plane-polarized light.
The direction of rotation (clockwise or counterclockwise) indicates the type of enantiomer:
dextrorotatory (right)
levorotatory (left).
What is a stereocenter?
Any atom that is attached to four different groups, resulting in stereoisomers.
All chiral centers are stereocenters,
But not all stereocenters are necessarily chiral.
What are diastereomers?
Stereoisomers that are not mirror images of each other.
They differ in configuration at one or more but not all of their stereocenters.
Generally have different physical properties compared to enantiomers.
How does chirality affect enzyme reactions?
Enzymes are chiral and often only bind to one specific enantiomer.
The chirality of substrates can influence the efficiency and outcome of biochemical pathways.
What is the R/S system used for?
A nomenclature system for assigning configuration to chiral centres.
R (rectus) indicates a clockwise configuration,
S (sinister) indicates a counterclockwise configuration
when viewed from a specific orientation.
What is chiral resolution?
The process of separating a racemic mixture into its individual enantiomers.
Techniques include:
chromatography,
distillation,
crystallization.
What role does chirality play in taste and smell?
Chiral molecules can produce different sensations; for example,
One enantiomer of a flavour may taste sweet,
while its mirror image may taste bitter.
Scent perception can also be influenced by chirality.
What is a prochiral molecule?
A molecule that can become chiral by a single modification (such as the addition of a substituent).
Prochiral molecules have parts that are indistinguishable but can turn into different enantiomers upon modification.
How is chirality relevant to agrochemicals?
Certain enantiomers of pesticides may exhibit different levels of toxicity to pests and wildlife.
Understanding chirality helps improve the efficacy and safety of agrochemicals.
What is a chiral pool strategy in organic synthesis?
A method that uses naturally occurring chiral molecules as starting materials to produce other chiral compounds.
This approach maximizes efficiency and selectivity in generating enantiomerically pure products.
What is enantioselectivity?
The preference of a chemical reaction to produce one enantiomer over the other.
High enantioselectivity during synthesis is often a goal in producing pharmaceuticals.
What methods can be used to determine the optical purity of a chiral substance?
Polarimetry:
measures how much a compound rotates plane-polarized light.
Comparing the optical rotation of a sample to that of a pure enantiomer provides a measure of optical purity.
How does chiral chromatography work?
A technique used to separate enantiomers based on their interactions with a chiral stationary phase.
This method allows for the isolation of specific enantiomers from mixtures.
How does chirality affect material properties?
Chiral materials may exhibit different
mechanical properties
optical properties
electrical properties
based on their helical structure.
Applications include liquid crystals and photonic devices.
What is Infrared (IR) Spectroscopy?
A technique used to identify and study chemicals by measuring the absorbance of infrared radiation by a sample.
Typically focuses on the mid-infrared range of wavelengths (4000 – 400 cm⁻¹).
How does IR Spectroscopy work?
Molecules absorb specific frequencies of IR radiation that correspond to their vibrational modes.
The resulting absorption spectrum provides information about
bond strengths,
functional groups
molecular structure.
What can affect the frequency of absorption in IR Spectroscopy?
The type of vibrational mode (stretching or bending),
bond strengths (as indicated by Badger's Rule),
Weight of the bonded atoms.
What types of molecular motions can IR Spectroscopy detect?
It primarily detects vibrational motions such as
stretching (symmetric and asymmetric) and
bending (scissoring, rocking, wagging, and twisting)
of bonds within molecules.
What is the significance of the IR signal classification?
IR signals can be classified based on their
intensity,
position,
shape,
which can indicate the presence of specific functional groups and the overall molecular structure
What is a characteristic of the absorption spectrum of a molecule?
Each functional group has a specific absorption frequency, allowing for the identification of functional groups within a compound.
What types of samples can be analyzed using IR Spectroscopy?
IR spectroscopy can be applied to
solids,
liquids,
gases,
making it a versatile tool in both organic and inorganic chemistry.
What is the Born–Oppenheimer approximation in the context of IR Spectroscopy
It is an approximation that separates molecular motion into
electronic and
nuclear motions,
allowing for the simplification of analysing molecular vibrations.
What is the importance of functional groups in IR Spectroscopy?
Specific functional groups absorb characteristic wavelengths in the IR spectrum,
making it possible to identify the functional groups present in a compound.
What limitations does IR Spectroscopy have?
It cannot provide information about molecular structure in terms of connectivity (i.e., cannot determine molecular geometry)
Or effectively identify non-polar molecules, as they might not exhibit significant IR absorption.
What are vibrational modes?
Different ways in which atoms in a molecule move relative to each other, typically detectable by techniques such as Infrared (IR) Spectroscopy.
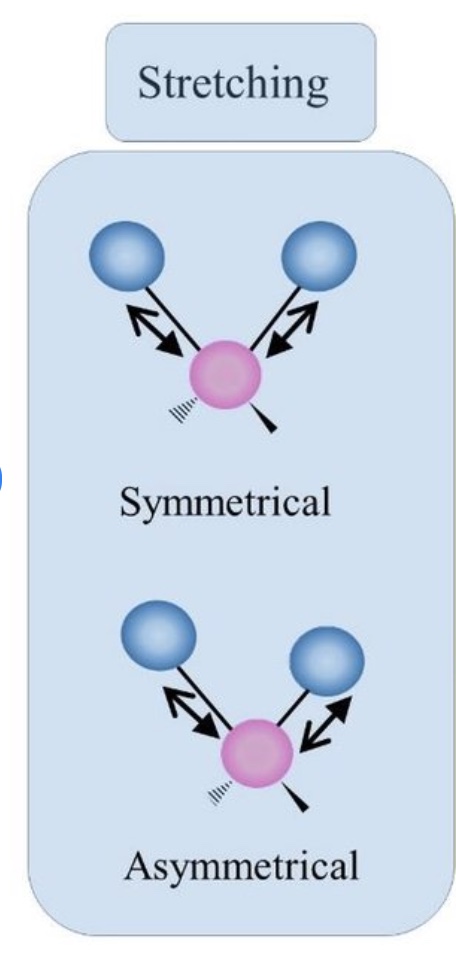
What are the two main types of stretching in vibrational modes?
Symmetric Stretching:
Bond lengths increase or decrease together.
-
-
-
Asymmetric Stretching:
Bond lengths change in opposite directions.
Name the subtypes of bending vibrational modes.
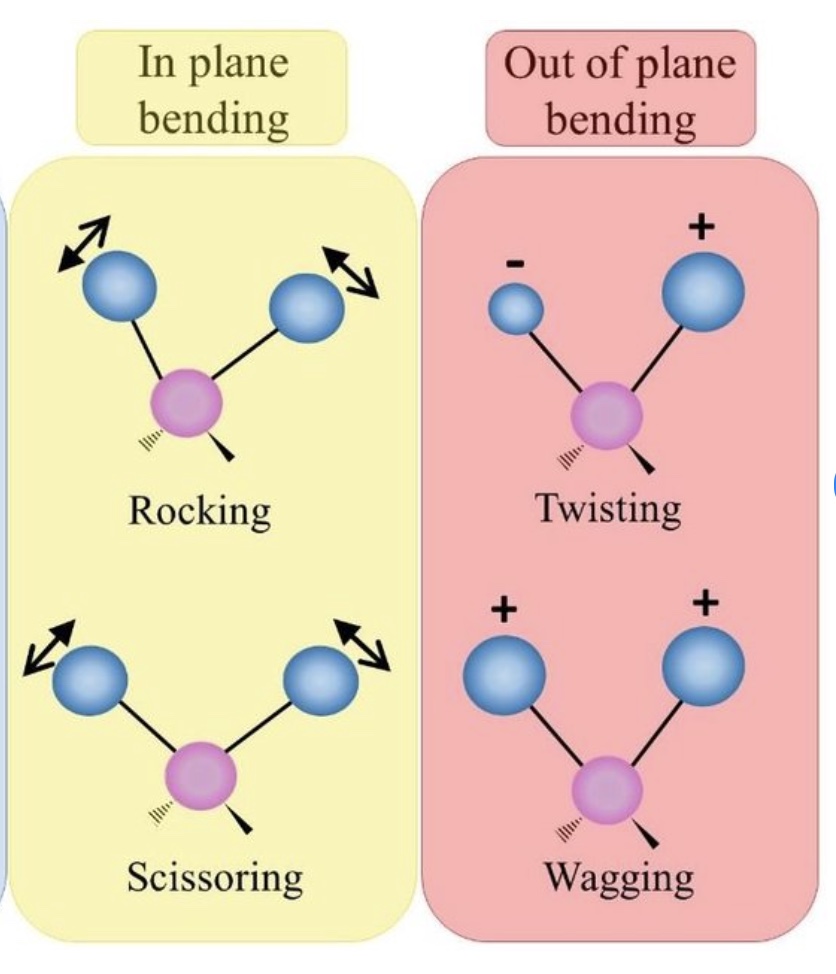
In-plane bending:
Scissoring: Angle decreases and then increases.
Rocking: Groups move back and forth in a plane.
Out-of-plane bending:
Wagging: Groups move up and down out of the plane.
Twisting: Groups rotate around the bond axis.
What factors affect vibrational frequencies?
Bond Strength: Stronger bonds vibrate at higher frequencies.
Mass of Atoms: Lighter atoms vibrate at higher frequencies.
Symmetry: Influences which vibrational modes are active in IR spectroscopy.
How do you calculate the number of vibrational modes in a molecule?
For linear molecules:
3N - 5
For non-linear molecules:
3N - 6.
(N = number of atoms)
What is the harmonic approximation?
A simplification that assumes molecular vibrations near the equilibrium positions of atoms behave like a harmonic oscillator.
Allows for easier analysis of vibrational modes.
How does the harmonic approximation treat molecular vibrations?
Models the potential energy of the system as a parabolic function of the atomic displacements from their equilibrium positions.
Leads to predictable vibrational frequencies.
What kind of potential energy function is associated with the harmonic approximation?
The Lennard-Jones potential is often referenced.
Describes potential energy between a pair of atoms as a function of distance, encompassing both attractive and repulsive forces.
What are the implications of the harmonic approximation for vibrational frequencies?
Allows for the calculation of vibrational frequencies using classical mechanics.
Frequency is related to the mass of atoms and the force constant (stiffness of the bond).
What are the limitations of the harmonic approximation?
Fails for
Large amplitude vibrations or
When potential energy significantly deviates from a parabolic shape.
Results in
Anharmonic effects that require more complex models for accurate description.
Why is the harmonic approximation beneficial in spectroscopy?
Simplifies the analysis of IR spectra and molecular vibrations.
Makes it feasible to predict spectral lines and understand molecular behaviour in a more manageable way.
What does the equation F = -kx represent?
F = -kx is Hooke's Law, where:
F: Restoring force (N)
k: Spring constant (N/m)
x: Displacement from equilibrium (m)
This law states that the force exerted by a spring is directly proportional to its displacement, with the negative sign indicating that the force acts in the opposite direction of the displacement.
Force increases as distance decrease
Force decreases as distance increases
What relevance does Hook’s Law have with IR?
In Infrared (IR) spectroscopy, the vibrational modes of molecules are modeled using Hooke's Law.
The stretching and bending of bonds in molecules can be seen as oscillations.
Different bond strengths (k values) lead to different vibrational frequencies.
This helps IR spectroscopy to identify and characterize molecular structures based on their vibrational transitions.
What are heterocycles?
Cyclic compounds containing one or more heteroatoms, Example
nitrogen,
oxygen,
sulfur
Can be aromatic or non-aromatic
Significant in various fields like pharmaceuticals, natural products, and materials science
What characterizes aromatic heterocycles?
Follow Hückel's rule (4n + 2 π electrons)
Exhibit delocalized pi electrons
Examples include
pyridine,
furan,
thiophene
Why are heterocycles important in pharmaceuticals?
Many drugs contain heterocyclic structures which enhance biological activity
Essential for the development of medicinal chemistry
Found in various natural compounds like alkaloids and vitamins
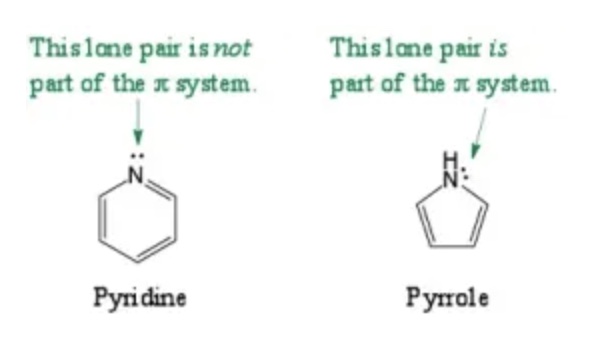
How does basicity vary in heterocycles?
Pyridine
has a lone pair on nitrogen, making it a weak base
Pyrrole
has nitrogen in the aromatic system, reducing its basicity due to electron delocalization
What are common reactions involving heterocycles?
Electrophilic Aromatic Substitution
in aromatic heterocycles
Nucleophilic Substitution
in non-aromatic heterocycles
Other reactions include
oxidation,
reduction, and
cyclization
What methods are used for the synthesis of heterocycles?
Cyclization reactions
from linear precursors
Multi-step synthetic pathways
for complex structures
Specific reactions include
Paal-Knoor synthesis for five-membered heterocycles
What defines non-aromatic heterocycles?
Lack delocalized pi electrons
Can be either saturated or unsaturated
Examples include
piperidine
morpholine
What role do heteroatoms play in heterocycles?
Influence the chemical properties and reactivity
Affect the basicity, electronegativity, and nucleophilicity of the compound
Participation in chemical reactions can differ from carbon-only cycles
List common examples of aromatic heterocycles.
Pyridine
Furan
Thiazole
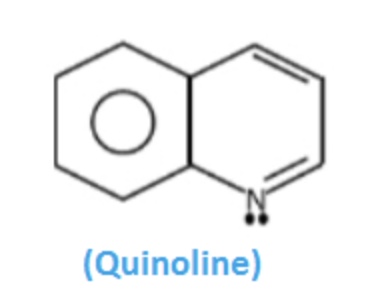
Quinoline
Pyridine
Furan
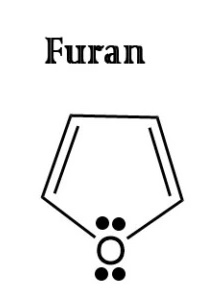
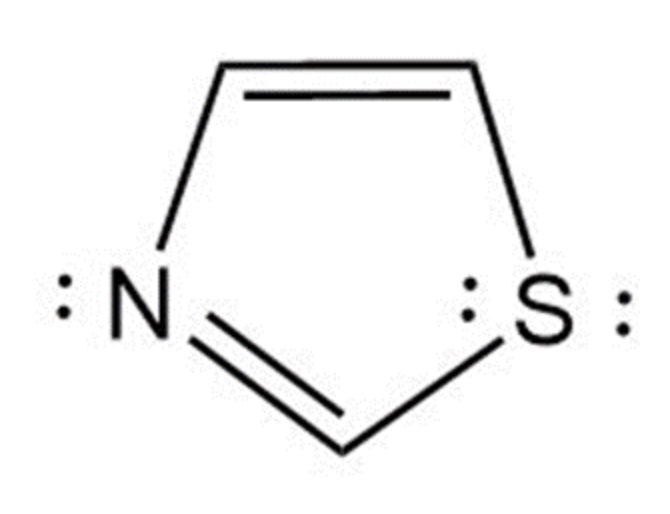
Thiazole
Quinoline
List of non-aromatic heterocycles
Pyrrolidine
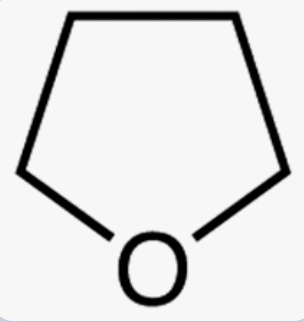
Tetrehydrofuran
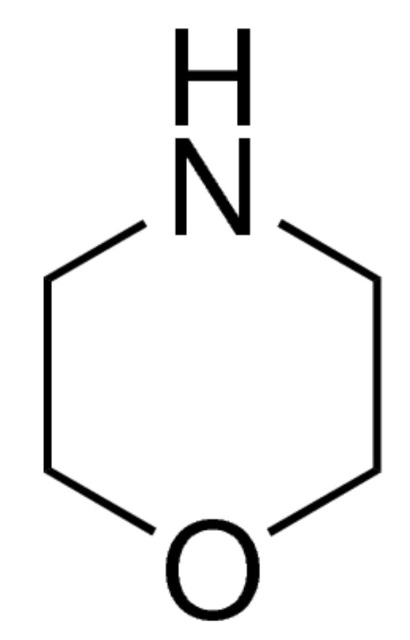
Morpholine
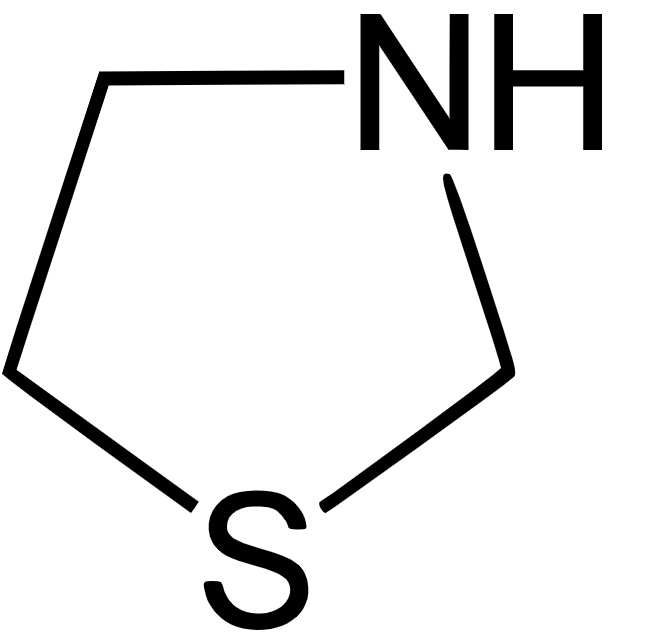
Thiazolidine
Pyrrolidine
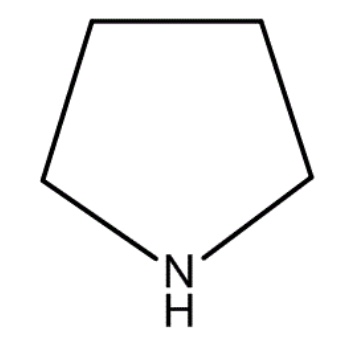
Tetrehydrofuran
Morpholine
Thiazolidine
Describe the Paal-Knoor synthesis.
Method for synthesizing substituted aromatic five-membered heterocycles
Involves the cyclization of 1,4-diketones with ammonia or amines
Can also use agents like phosphorus pentoxide (P₂O₅) and phosphorus pentasulfide (P₂S₅)
What is a key feature of aromatic substitution reactions in heterocycles?
One of the hydrogen atoms is replaced by another group
The reactions are facilitated by the stability of the aromatic system
Electrophiles are used frequently in these reactions
How do heterocycles contribute to material science?
Found in the production of new polymers
Used in materials with specific electronic properties
Contribute to the development of conductive materials and organic semiconductors
What are the characteristics of five-membered aromatic heterocycles?
Typically includes one or more heteroatoms like
nitrogen,
oxygen,
sulfur
Exhibits aromaticity and follows Hückel's rule
Examples include
pyrrole,
furan,
thiophene
What effects does substitution have on the stability of aromatic heterocycles?
Electron-donating groups can
enhance stability and
increase reactivity
Electron-withdrawing groups can
decrease stability and
affect the position of substitution
The nature of substituents greatly influences the chemical behavior of the compound
Name two methods used to study the reactivity of heterocycles.
Spectroscopic techniques such as
NMR and
mass spectrometry
Chemical tests to determine
acidity,
basicity,
reaction rates
What are reactivity patterns commonly observed in aromatic heterocycles?
Generally undergo electrophilic substitution reactions
May exhibit nucleophilic attack under specific conditions
Temperature
Catalyst
Reactivity is influenced by the nature of the heteroatom
What is the impact of substituents on the reactivity of aromatic heterocycles?
Electron-donating groups
enhance nucleophilicity
Electron-withdrawing groups
destabilize the aromatic system and affect reactivity
Substituents can direct the location of new groups added
ortho positions
meta positions
para positions
What are some common synthetic routes used for heterocycles?
Cyclization reactions
from precursor compounds
Multi-component reactions
that form heterocyclic systems
Utilizing cross-coupling methods
to build complex heterocycles
Describe fused heterocycles.
Heterocycles that share common bonds with other cyclic structures
Often exhibit unique stability and reactivity properties due to their fused nature
Examples include
indole (fused benzene and pyrrole) and
quinoline (fused benzene and pyridine)
How does solvation affect the properties of heterocycles?
Solvent polarity can influence the
rate and equilibrium of reactions
In polar solvents,
heterocycles can exhibit increased solubility
Interaction with solvent can alter spectroscopic signals
What is a common reaction pathway for synthesizing aromatic heterocycles?
Nitro substitution
followed by reduction to yield amines or other groups
Bicyclic synthesis
from precursors containing multiple functionalities
Aromatic substitution
to introduce necessary heteroatoms