Reductive ways to make Amines
0.0(0)
0.0(0)
Card Sorting
1/4
There's no tags or description
Looks like no tags are added yet.
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
5 Terms
1
New cards
NaN3; LiAlH4, H2O (or H2, Pd)
Azide (N3-) displaces LG. Then it is reduced to form an amine

2
New cards
NaCN; LiAlH4 then H2O
Nitrile displaces LG. It is then reduced to form an amine

3
New cards
H2, Pd ( or Fe/Zn/Sn with HCl)
Nitro group(-NO2) on benzene is reduced to an amino group (-NH2)
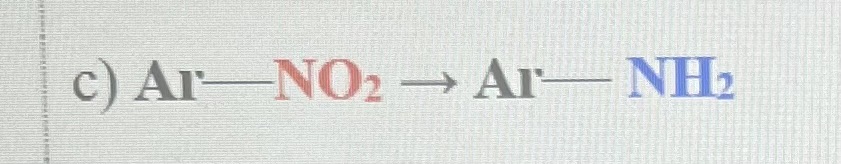
4
New cards
LiAlH4 then H2O (w/ amide
With an amide, you can reduce it to become a secondary amine. The O is turned to 2 H on C.

5
New cards
LiAlH4 then H2O (w/ imine)
An imine is reduced to a secondary amine. The double bond is broken to give 1 H to N and 1 to C
