3.2.1 Enthalpy change
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
What is enthalpy
Enthalpy, H, is a measure of the heat energy in a chemical system
enthalpy is sometimes thought of as the energy stored within bonds. Enthalpy cannot be measures, but enthalpy changes can
What does system refer to, and what does surroundings refer to
The CHEMICALS - The atoms, molecules or ions making up the REACTANTS/PRODUCTS
The surroundings is everything outside the chemicals eg the thermometer, the lab etc
The universe is everything- and includes both system and surroundings
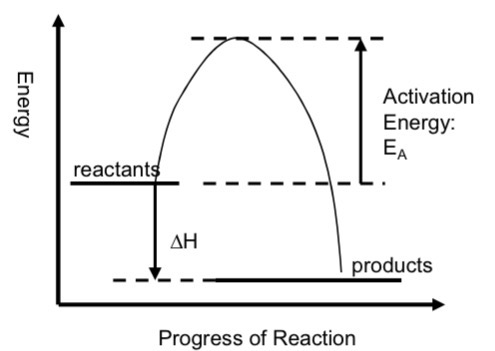
What is an exothermic reaction and draw the diagram
energy is transferred from the system (chemicals) to the surroundings. The products have less energy than the reactants
In an exothermic reaction the H change is NEGATIVE
Common oxidation exothermic processes are the combustion of fuels and the oxidation of carbohydrates eg glucose in respiration
Chemical system looses energy, surroundings gain energy, temp of surroundings increase
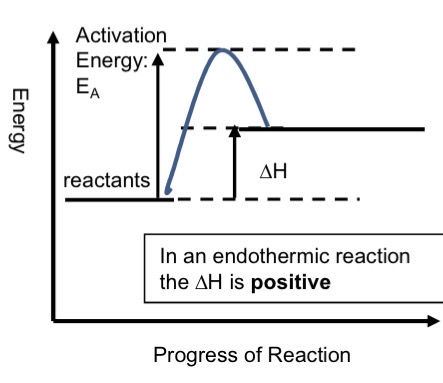
What is an endothermic reaction and give a diagram
Energy is transferred from the surroundings to the system (chemicals)
They require an input of heat energy eg thermal decomposition of calcium carbonate
The products have more heat energy than the reactants
The H change is POSITIVE
chemical system gains energy, surroundings loose energy, temperature of the surroundings decreases
Define activation energy
The minimum energy required for a reaction to take place
the energy input required to break bonds acts as a barrier to the reaction
Also known as the minimum energy which particles need to collide to start a reaction
On an enthalpy profile diagram, the arrow goes from the dotted line(that extends from the reactants) to the top of the curve
What are standard conditions and standard states
pressure is 100kPa - very close to the pressure of 1 atm, 101kPa
Temperature - 298K (25 degrees Celsius)
Concentration- 1 mol dm^-3 of solution
Standard state is the physical state of a substance under standard conditions
What is the enthalpy change of reaction
The enthalpy change that accompanies a reaction in the molar quantities shown in a chemical equation under standard conditions
or the number of moles of reactants as specified in the balanced equation react together
What is the enthalpy change of formation
The enthalpy change that takes place when 1 mole of a compound is formed from its elements under standard conditions
Therefore there should only be 1 MOL of products
Shown as the triangle symbol, little f then big H
What is the enthalpy change of formation of an element
0 kJ mol ^-1
as this refers to the formation of one mol of an element from its elements under standard- there is clearly no change
What is he enthalpy change of combustion
The enthalpy change that takes place when 1 mole of a substance reacts completely with oxygen under standard conditions, with all reactants and products in their standard states
Shown by the big triangle, little c and big H
What i the enthalpy change of neutralisation
The energy change that accompanies the reaction of an acid by a base to form one mole of H20 (l) under standard conditions, with all reactants and products in their standard states
symbol of big triangle, small neut and big H
What is the equation for heat energy given out or absorbed in a reaction, what is the equation for enthalpy change per mole
q=mc delta c
Enthalpy change per mole = q/ number of moles reacting
Define average bond enthalpy
The breaking of 1 mol of bonds in gaseous molecules
use mean because every single bond in a compound has a slightly different bond energy
The definition only applies when the substances start and end in the gaseous state
Does bond breaking release energy?
No bond breaking absorbs energy, bond making releases energy
If a section is endothermic, more energy is ‘-‘ when ‘-‘ bonds than is ‘-‘ when ‘- bonds
More energy is absorbed when breaking bonds than is released when making bonds
In an exothermic reaction, more energy is ‘-‘ when ‘-‘ bonds than is ‘-‘ when ‘-‘ bonds
More energy is released when making bonds than is absorbed when breaking bonds
What is the calculation to find enthalpy change using average bond enthalpy
Bond energies broken (in reactants) - bond energies made(in products)
What determines if a reaction is exothermic or endothermic
The difference between the energy required for bond breaking and the energy released by bond making
Hess’s law
States that total enthalpy change for a reaction is independent of the route by which the chemical change takes place
Why is Hess’es law used
To measure enthalpy change for a reaction that cannot be measured directly by experiments, instead alternative reactions are carried out that can be measured experimentally
Eg impossible to add the exact amount of water - and not easy to measure the temperature change of a solid
Describe a calorimetric method
Wshes the equipment (cup, pipettes etc) with the solutions to be used
Dry the cup
Put the polystyrene cup in a beaker - for insulation and support
Measure out desired volumes of solutions with volumetric pipettes to transfer to insulated co
Clamp thermometer in place - submerged in solution
Measure initial temperature of both solutions- every min for 2 mins, allowing both solutions to acclimatise
At 3 min transfer the second reagent to the first cup
If using a solid use the before and after method
Stir mixture - ensure all solution is the same temp
Record temp every mind for several mins
Why stir the solution, why polystyrene cup, why wash equipment with the solutions that are being used
Stir- ensure all of the solution is at the same temperature
Cup - insulation, reduce the temp lost to surroundings
Wash with solutions- prevent dilutions or contamination - may contain water that dilutes the reagent and affect concentration and results
Describes errors in this method
Energy transfer from surroundings (usually loss)
Approx in specific heat capacity of solution - the method assumes all the solutions have the heat capacity of water
Neglecting the specific heat capacity of the calorimeter - we ignore any energy absorbed by the apparatus
Reaction or dissolving may be incomplete or slow
Density of the solution is taken to be the same as water
If the reaction is too slow, what can happen that means the exact temperature rise is difficult to obtain - how can we counteract this
Cooling may occur simultaneously with the reaction
Counteract via taking readings at regular time intervals and extrapolate the temperature curve/line back to the time the reactants were added together
Also measure the temperature of reactants before they are added together t