Organic Chemistry - Hydrocarbons and Crude oil
1/56
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
Hydrocarbon
Hydrocarbons are compounds/molecules that are made up of carbon and hydrogen only.
Alkanes
saturated hydrocarbons
this means no more hydrogen atoms can be added
all carbon-carbon bonds are single covalent bonds
Rules for carbon and hydrogen bonding
Carbon - Max 4 covalent bonds (can be double)
Hydrogen - Max 1 covalent bond
General formula of an alkane
CnH2n+2
Homologus series
Group of organic molecules/compounds that have
Similar chemical properties
Due to them all having the same functional group/general formula
Properties of Hydrocarbons
→boiling point
→volatility
→viscosity
→flammability
What are properties of hydrocarbons dependent on?
the size of the molecule
Boiling point
temperature at which a liquid evaporates to form a gas
Viscosity
measure of a liquid's resistance to flow
(how thick or runny a liquid is)
Flammability
How easily a substance combusts.
If very flammable means it is easy to burn and catches fire easily
Small vs Long Hydrocarbons
Long
High BP
High Viscosity
Less Volatile
Less flamable - smoky flame (due to incomplete combustion)
Short
Low BP
Flow more easily
More volatile
More flammable
Complete combustion of hydrocarbons
hydrocarbon+oxygen —> carbon dioxide +water vapour (+ energy)
plentiful supply of oxygen
carbon and hydrogen in fuel completely oxidised
Prefixes for carbon and n0. correlation
Meth - 1
Eth - 2
Prop - 3
But - 4
Pent - 5
Hex - 6
Draw Alkanes Table
DRAW
Types of Formula
Displayed
Gernal
Molecular
Structural
Why are longer hydrocarbons less flammable?
There are strong intermolecular forces between the molecules, which require more energy to overcome.
This makes them less volatile, meaning they evaporate less easily
so they produce fewer flammable vapours to mix with the air and are harder to ignite
Incomplete combustion
→ limited supply of oxygen
→ carbon monoxide and/or carbon (soot) also produced
How to test for products of complete combustion of a hydrocarbon
→ Carbon dioxide turns limewater cloudy
→ Water turns blue cobalt chloride paper pink
Crude oil
It is finite resource
It is non renewable
It is a mixture of many different compounds (separated by fractional distillation)
Made up mostly of hydrocarbons (majority are alkanes)
Crude oil produces Many of the fuels on which we depend for our modern lifestyle
How was crude oil formed?
Formed by remains of PLANKTON and dead plants and animals
That were buried in mud (compressed under mud)
Over millions of years
These organic remain were then subject to compressed under extreme pressure and heat
The heat and pressure chemically changed the organic remains into crude oil
Fractional distillation process
Crude oil is heated
Hydrocarbons and liquids evaporate/vaporise into a gas
Which is passed into the fractionating column
That has a TEMPERATURE GRADIENT
When hydrocarbons reach a temperature lower than their boiling point they condense into a liquid
Fractions collect at different levels depending on their boiling point
(Longer condense at the bottom as it is hotter and shorter at the top where it is cooler)
What are fractions of crude oil used for (PETROCHEMICAL INDUSTRY)
Many useful materials on which modern life depends are produced by the petrochemical industry
Fuels
Feedstocks - used to make new compounds
Polymer
Solvent
Lubricants
Detergents
In terms of boiling point from lowest to highest list products of fractionating
LPG
Petrol
Kerosene
Diesel
Heavy Fuel
Bitumen
Cracking
Process by which you break down longer hydrocarbon chains into smaller more useful hydrocarbons
Thermal Decomposition Reaction
Why crack hydrocarbons?
→ some heavier fractions from fractional distillation of crude oil are not in high demand
→ these hydrocarbons are made up of large molecules that are difficult to vaporise and difficult to burn
→ this means that they are poor fuels
→ so are broken down into smaller, more useful hydrocarbons via cracking
Two types of cracking
→ catalytic cracking
→ thermal cracking
Catalytic cracking
long chain alkanes are heated until they vaporise into a gas
The gaseous alkanes are passed over hot aluminium oxide catalyst/zeolite catalyst at 500degrees (high temp)
Break the long chain alkanes into shorter chain alkane and alkene
Steam cracking
→ heat fraction/long chain alkane until the vaporise into a gas
→ mix vapour with steam at high temperature (800-900 degrees)
no catalyst involved
Product of Cracking
Alkanes + Alkenes
Isomers
Isomers are molecules that have the same molecular formula, but different structural formulas, this means they are made of the same atoms, but the atoms are arranged differently
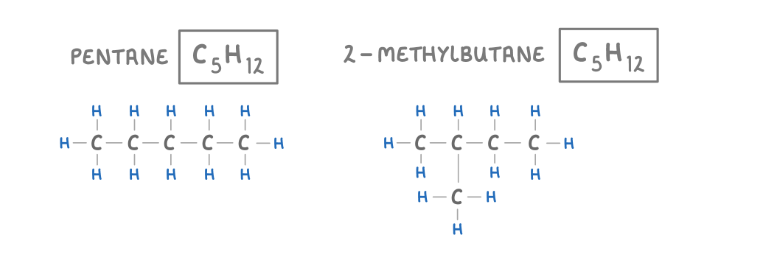
What does saturated mean when talking about hydrocarbons
Saturated means there are no carbon carbon double bonds
What are short chain alkanes used for?
Fuels
Why are hydrocarbons used as fuels?
They release a large amount of energy when combusted
Volatility
Refers to how easily a substance vaporises/evaporates into a gas
(very volatile means it easily evaporates and gives off fumes)
Is combustion an exothermic or endothermic reaction?
Combustion is an exothermic reaction as it releases heat energy to the surroundings
combustion of hydrocarbon fuels releases energy
Why does viscosity increase as chain length increases?
Viscosity increases because longer molecules can tangle together.
There are stronger intermolecular forces, so the molecules flow less easily.
Why does boiling point increase as chain length increases?
Boiling point increases as the number of carbon atoms increases
Because the intermolecular forces increase
These intermolecular forces increase as the size of the molecule increases
More energy is needed to overcome these forces.
Which hydrocarbons make good fuels?
Shorter hydrocarbons - Very flammable and volatile so easy to combust for energy
Longer hydrocarbons - Not very flammable or volatile so they are hard to combust for energy
These properties influence how hydrocarbons are used as fuels
Feedstocks
A feedstock is a raw material used to provide reactants for an industrial reaction.
Petrochemicals
A petrochemical is a substance made from crude oil, via chemical reactions.
What is the use of bitumen?
Road surfacing
As it is very viscous
What is the use of the fuels in CRUDE OIL
Heavy fuel oil - (fuel for ships)
Diesel Oil - Fuel for diesel vehicles
Kerosene - Aircraft fuel
Petrol - Fuel for cars
LPG - Bottled gas for heating and cooking
Why is there a vast array of natural and synthetic carbon compounds?
Carbon atoms form four covalent bonds.
They bond to other carbon atoms to form chains and rings.
This allows the formation of homologous series.
Members of a homologous series have the same functional group, same general formula, and similar chemical properties.
Therefore, carbon forms a very large number of different compounds.
Why are the products of cracking useful?
Shorter hydrocarbons make better fuels
Shorter alkanes can also be used as feedstock for the petrochemical industry to make polymers and detergents
What type of reaction is cracking an example of?
Cracking is a thermal decomposition reaction
Alkenes
Alkenes are also hydrocarbons and also an example of a homologous series.
Alkenes have a carbon double bond
Alkenes are unsaturated
Why cant there be an alkene called methne?
Methene is only one carbon atom and alkenes are made from two carbon atoms forming a double bond
What is the test for alkenes?
Bromine water test
Bromine water by itself is a bright orange colour
When alkenes are present it decolourises the bromine
Causing it to turn colourless
Which is more reactive alkanes or alkenes and why?
Alkenes are more reactive than alkanes.
Alkenes contain a carbon–carbon double bond (C=C).
The double bond makes them more reactive
What is the purpose of cracking?
Hydrocarbons can be broken down (cracked) to produce smaller, more useful molecules.
What are the products of cracking used for?
There is a high demand for fuels with small molecules and so some of the products of cracking are useful as fuels.
Alkenes are used to produce polymers and as starting materials for the production of many other chemicals.
How Modern Life Depends on Hydrocarbons
Cracking is useful because it breaks long-chain hydrocarbons into shorter-chain hydrocarbons, which are in higher demand as fuels. It also produces alkenes used to make polymers.
Modern life depends on hydrocarbons because they are used as fuels for transport, heating and electricity, and as feedstock for making plastics and other chemicals.
Why is cracking useful?
Breaks long-chain hydrocarbons
Produces short-chain fuels
Matches supply and demand
Produces alkenes
Alkenes make polymers
How does modern life depend on hydrocarbons?
Fuels for transport
Heating and electricity
Raw materials (feedstock)
Used to make plastics and chemicals
Why is there more demand for short-chain hydrocarbons?
They are more useful as fuels
More flammable
Lower boiling points
Used in petrol
First 4 Alkenes
Ethene
Propene
Butene
Pentene
Alkene General Formula
CnH2n