Unit 7: Quantum, Atomic, and Nuclear Physics
1/26
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
27 Terms
Quanta
Light being emitted as individual packets of energy called quanta.
Photon
A quantum of electromagnetic energy is known as a photon.
Photoelectric effect
Light behaves like a stream of photons, illustrated by the photoelectric effect.
Photoelectrons
The released electrons are known as photoelectrons.
Wave theory of light predictions
The significant time delay between the moment of illumination and the ejection of photoelectrons.
Increasing the intensity of the light could cause the electrons to leave the metal surface with greater kinetic energy.
Photoelectrons would be emitted regardless of the frequency of the incident energy, as long as the intensity was high enough.
Threshold frequency
Minimum frequency required to produce photoelectrons from a metal.
Work function
Energy required to impart to an electron on the metal surface.
Wave function
The probability that a particle will be measured to be at a particular position when the position is measured. That probability is related to a new physical parameter called the wave function.
Nuclear force
Force binding neutrons and protons in the nucleus.
Binding energy
Energy required to disassemble a nucleus into its constituent nucleons.
Nuclear fusion
Process of combining small nuclei at high temperatures.
Nuclear fission
Process of splitting a nucleus or emitting particles.
Alpha decay
Emission of an alpha particle from a nucleus.
Beta decay
Decay processes involving the transformation of neutrons and protons.
Gamma decay
Process where a nucleus relaxes and sheds energy in the form of a gamma ray.
Disintegration energy
Energy involved in nuclear reactions, determining if reactions are exothermic or endothermic.
Atomic Spectra
The light from a glowing gas, passed through a prism to disperse the beam into its component wavelengths, produces patterns of sharp lines called atomic spectra.
The wavelength of photon
Photon’s wavelength:
λ = wavelength
c = speed of light
f = frequency
h = Planck’s constant (6.626 × 10 -34 joule·s)
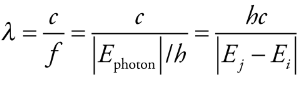
De-Broglie Wavelength
De-Broglie wavelength explains matter's wave-like behavior in quantum mechanics.
The equation is λ = h/p, where λ is De-Broglie wavelength, h is Planck's constant, and p is particle momentum.As momentum increases, the wavelength decreases.
This is important for understanding particle behavior at the quantum level.
Wave-Particle Duality
Electromagnetic radiation propagates like a wave but exchanges energy like a particle. This is known as wave-particle duality.
Theory of relativity
The results of physical experiments will be the same in any-nonaccelerating reference frames.The speed of light is constant.
Time dilation
Demonstrated by synchronized atomic clocks.
Length Contraction
To be consistent with time dilation, there must also be disagreement about distances. This is known as length contraction.
Isotopes
The nuclei that contain the same number of neutrons are called isotopes.
Mass number
The total number of nucleons (Z+N), is called the mass number, and is denoted by A
Atomic number
The number of protons in a given nucleus is called the atom’s atomic number denoted by Z.
Nuclear force
The strong nuclear force is a fundamental force which binds neutrons and protons together to form nuclei.