MTLBE_Week 07_4688-Clinical Laboratory Law
1/305
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
306 Terms
Clinical Laboratory
A facility that is involved in pre-analytical, analytical and post-analytical, where tests are done on specimens from the human body to obtain information about the health status of a patient for the prevention, diagnosis and treatment of diseases.
Familiarize and Memorize
2 years
All existing licensed CL shall be given ___ years to fully offer the additional services for each category with corresponding personnel and equipment from the date of effectivity of AO 2021-37
Tertiary
The serology/immunology section of this lab should provide the ff. additional service capabilities:
- Any machine-based serological and immunological testing such as, but not limited to: tumor markers, thyroid function tests and hepatitis profile
Section 2
This section of RA 4688 states that no license shall be granted or renewed unless such laboratory is under the administration, direction and supervision of an authorized physician
RA 4688
Republic Act of Clinical Laboratory Act of 1966
RA 4688: Clinical Laboratory Act of 1966
This regulation is applied to all laboratories collecting human samples and performing assays to help in the diagnosis, prevention, treatment and management of diseases and also to promote personal and public health.
8 sections
How many sections are there in RA 4688
True
True
In some circumstances if the lab always get failed mark at EQAP, they can't obtain license to operate
Familiarize

sanction/penalty
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
- Performing laboratory procedures beyond their authorized service capability; and,
- Giving and receiving any commission, bonus, kickback or rebate or engaging in any
split-fee arrangement in any form whatsoever with any facility, physician,
organization. agency or person., cither directly or indirecily, for patients referresd to a
CL licensed by the DOH.
Certificate of proficiency (SACCL)
Additional proof of trainings for rHIVda
Certificate of training in
bacteriology (RITM and other
RITM recognized institutions)
Additional proof of trainings for Bacteriology
Certificate of training in malaria
smear (RITM)
Additional proof of trainings for Malaria Smear
True
T or F
Refusal to participate in an External Quality Assurance Program given by
the designated NRL shall be one of the bases for suspension/revocation of license of the laboratory.
Clinical Chemistry
What is the scope of Lung Center of the Philippines (as NRL)
External Quality Assessment Program (EQAP)
A program where participating CL are given unknown samples for analysis
These samples should be treated as ordinary human specimens for the usual processing and examination
Primary
The hematology section of this lab should provide the ff. minimum service capabilities:
- Complete Blood Count (Hemoglobin, Hematocrit, Red Blood Cell Count, White Blood Cell Count with Differential Count, Quantitative Platelet Count)
- Forward and reverse ABO grouping and Rh (D) typing (tube method)
Pathologist
Who is the head of the CL?
1. Refusal to allow HFSRB/CHD-RLED authorized personnel to conduct inspection or monitoring visits of the clinical laboratory at any appropriate time;
2. Refusal or nonparticipation of any CL in an External Quality Assessment Program (EQAP) provided by a designated NRL or other local and international EQAP approved by the DOH;
3. Absence of action to improve the unsatisfactory or failed EQAP administered by a designated NRL or other local and international EQAP approved by the DOH;
4. Demonstrating incompetence or making consistent errors in the performance of CL examinations and procedures;
5. Deviation from the standard test procedures including use of expired reagents;
The Clinical Laboratory (CL) may be sanctioned or penalized upon commission of the following prohibited acts and violations: (1-5)
November 14, 2000
DO no. 393E s. 2000 was issued on what date?
NKTI
NRL for hematology including immunohematology, immunopathology and anatomic pathology (Consistent with D.O 301 -I series of 1999)
RA 4688: Clinical Laboratory Act of 1966
An act regulating the operation and maintenance of clinical laboratories and requiring the registration of the same with the department of health, providing penalty for the violation thereof, and for other purposes
Section 1
Section about Registration and Licensing
Section 2
Section about laboratory should be headed by a duly registered physician
Section 3
Section about secretary of health, through the Bureau of Research and Laboratories (BRL), now DOH, are the one who are responsible for issuance of rules and regulations
Section 4
Section about Punishment
-Imprisonment for not less than ONE MONTH but NOT
MORE THAN ONE YEAR
-Fine of NOT LESS THAN 1K nor MORE THAN 5K
Section 5
Section states that:
if any section or part of this Act shall be adjudged by any court of competent jurisdiction to be invalid, the judgment
shall not affect, impair, or invalidate the remainder thereof.
1. Government CL doing microscopy work for specific DOH programs such as but not
limited t; the following:
a. Malaria screening
b. Acid fast bacilli microscopy
c. Tests for STDs
d. Cervical cancer screening using Pap smears
2. An amendment to AO 2007-0027, which is AO 2007-0027-A, also spared the National Tuberculosis Reference Laboratory (NTRL) and its regional Reference Laboratories performing Tb cultures from securing license to operate a clinical laboratory.
3. Special clinical laboratories like research labs, labs for teaching purposes, genetics lab, immunohematology labs, and molecular labs are also be exempted from RA 4688 because either they are regulated by other laws or there is no regulation available at all
Special CLs that are not subject to administrative orders are required to register under the BHFS (Bureau of Health Facilities and Services) without being licensed under the provisions of this law:
Laboratories that are excluded from RA 4688
Acid Fast Bacilli Microscopy
It is a test under government CL that is conducted for tuberculosis
Laboratory the doesn't need LTO but needs to seek certificate of registration
Special CLs
AO 201 s 1973
Revised Rules and Regulations Governing the Registration, Operations and Maintenance of Clinical Laboratories in the Philippines
AO 290 s 1976
Amending Section ll Subsection A of Administrative Order No. 201, Series of 1973 Concerning Requirements for a Clinical Laboratory
AO 49-B s. 1988
Revised Rules and Regulations Governing the Registration, Operation and Maintenance of Clinical Laboratories in the Philippines
EO 102 s. 1999
Redirecting the Functions and Operations of the DOH
A0 59 s. 2001
Rules and Regulations Governing the Establishment, Operation and Maintenance of Clinical Laboratories in the Philippines)
AO 27 s. 2007
Revised Rules and Regulations Governing the Licensure and Regulation of Clinical Laboratories in the Philippines)
AO 27 s. 2007
Latest AO that is being followed under RA 4688
External Quality Assessment Program (EQAP)
Program where participating CL are given unknown samples for analysis. These samples should be treated as ordinary human specimens for the usual processing and examination. The quality of performance of the CL shall be assessed through the closeness of its results to the pre-determined value or reference value generated by the participating CL through peer group analysis.
Mobile Clinical Laboratory (MCL)
A laboratory testing unit capable of performing
limited CL diagnostic procedures. It moves from one testing site to another, and it has a
DOH-licensed CL as its main laboratory
National External Quality Assessment Scheme (NEQAS)
An EQAP activity conducted by the National Reference Laboratories to assess the quality of perfomance and accuracy of the results of laboratories.
National Reference Laboratory (NRL)
The highest level of laboratory in the country
performing highly complex procedures, including confirmatory testing, that is not
commonly performed by the lower level of laboratory. It is the responsible entity for facilitating NEQAS to ensure compliance to quality standards for regulation and licensing of all laboratories in the Philippines.
June 18 1966
Date when the Clinical Laboratory Law (RA 4688) was passed
Satisfactory (4/5 accurate and precise result of sample)
Grade you need to obtain in EQAP
True
T or F
Grades in EQAP
Satisfactory: 4/5 accurate and precise result of sample
Very Satisfactory: 5/5 accurate and precise result of sample
True
T or F
EQAP, NEQAS (determine performance of the laboratory; passed or failed) and NRL are related to each other
Physician's Office Laboratory (POL)
refers to a doctor's office/clinic wherein CL
examinations are per formed for the purpose of monitoring the doctor's patients only, wherein NO official results shall be issued. In this Order, POL within the premises of a DOH-regulated facility shall be under the supervision of the CL
Point of Care Testing (POCT)
refers to diagnostic testing done at or ncar the site of patient care rather than in the CL. It may be in the emergency room, operating suites,
wards, and ambulances.
Satellite Clinical Laboratory (SCL)
refers to an extension of the main CL located
within the facility's compound or premises. It shall have the same service capability as
the main laboratory.
Referral Tests
refers to CL tests that are either sent-out or outsourced to other DOH- licensed CL with the same or higher service capability
Point of Care Testing (POCT)
Drawback of this is that personnels are not medtechs, they have the skills but the results are not accurate (Ex. Glucometer)
Classification by:
1. Ownership
2. Institutional Chharacter
3. Function
4. Service Capability
According to AO No. 37 s. 2021, what are the classification of Clinical laboratories?
Government-owned
CL classification by ownership: owned,
wholly or partially, by national or
local government units
Private-owned
CL classification by ownership: are owned,
established, and operated by an individual, corporation, institutior association, or organization
True
T or F
The examples of government base hospitals
San Lazaro Hospital
Jose R. Reyes Memorial Medical Center
Hospital ng Maynila
UP Hosiptal (?)
True
T or F
The examples of private base hospitals
St. Lukes
FEU NRMF Hospital
Institution-based
Classification by Institutional Character: A laboratory that is located within the premises and operates as part of a DOH licensed health facility
Non-institution based
Classification by Institutional Character: A laboratory that operates independently and is not attached to any DOH licensed health facility.
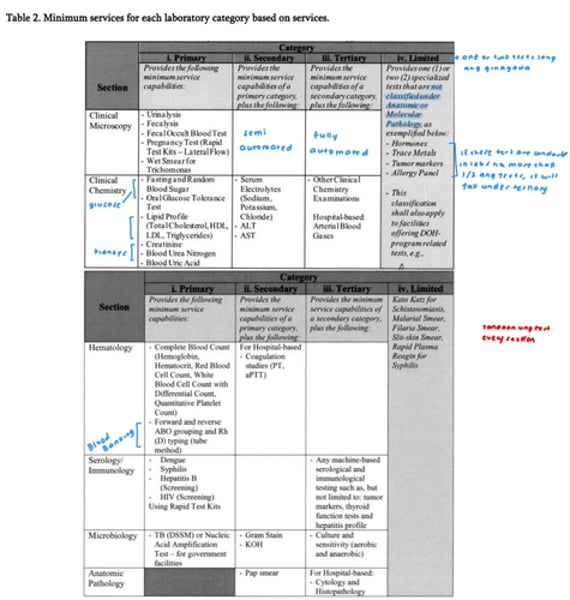
Clinical Pathology
Deals with the chemical and cellular analyses of blood and other body fluids (includes, but not limited to, clinical chemistry, clinical
microscopy, toxicology, therapeutic drug monitoring, immunology and
serology, hematology and coagulation), iden tification and examination of
microbes and parasites (bactcriology/parasitology/mycology/virology).
Anatomic Pathology
Provides processing and examination of surgical specimens as to the physical appearance and microscopic structure of tissues (and organs), such as, but not limited to, surgical pathology. cytopathology,
immunohistochemical techniques, autopsies and forensic pathology.
Anatomic Pathology
Concerned with the diagnosis of diseases through microscopic examination of tissues and organs
Molecular Pathology
Deals with the analysis of certain genes, proteins and other molecules in samples from organs, tissues or bodily fluids in order to diagnose discasc and/or to guide the prevention and trcatment of disease based
on the principles, techniques and tools of molecular biology as they are appled to diagnostic medicine in the laboratory.
Memorize and Familiarize
A. Permit to Construct (PTC)
1. A completely filled out application form for DOH-PTC (downloadable at
www.hfsrb.doh.gov.ph), whether manual or online, shall be submitted to the DOH
regulatory offices, as specified in Section V. B of this Order.
2. A DOH-PTC shall be required for construction of new CL and for renovation or
expansion of existing CL, including change in ownership and transfer of location.
3. The application shall be processed in accordance with the procedural guidelines set
forth in A.O. No. 2016-0042, also known as, "Guidelines in the Application for
Department of Health Permit to Construct (DOH-PTC)."
Procedural Guidelines for Permit to Construct
True
T or F
Inspection can be conducted ny the Director or 2 authorized representatives (HSFRB or CHD)
True
T or F
LTO shall be placed in an area that can readily be seen by the public at all times
False
Non-transferable
T or F
LTO is transferable
False
New license must be applied
T or F
Old license can still be applied if there are changes in ownership or trabsfer of location
True
T or F
Different branches = separate license
15 days
Changes in name, ownership, headship, DOH should be informed within _____
1 year
For diagnostic test, DOH-LTO is valid for how many year/s?
three (3) years
COR for CL that is operated and maintained exclusively for research and teaching purposes shall be required to register with the DOH-HFSRB every _______.
Health Facilities and Services and Regulatory Bureau
They set standards for the regulation of CL and strictly enforce the provisions of this
Order.
Health Facilities and Services and Regulatory Bureau
They disseminate regulatory policies, standards and forms for information and
guidelines of the DOH-CHDs.
Health Facilities and Services and Regulatory Bureau
They provide consuntation and technical assistance to stakeholders, including regulatory officers from the DOH-CHDs in line with the regulation of CL
Health Facilities and Services and Regulatory Bureau
They respond promptly to complaints relative to the operation of CL under its jurisdiction.
sanction/penalty
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
Refusal to allow HFSRB/CHD-RLED authorized personnel to conduct inspection or monitoring visits of the clinical laboratory at any appropriate time
sanction/penalty
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
Refusal or nonparticipation of any CL in an Extemal Quality Assessment Program
(EQAP) provided by a designated NRL or other local and international EQAP approved by the DOH
sanction/penalty
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
Absence of action to improve thE unsatisfactory or failed EQAP administered by a
designated NRL or other local and international EQAP approved byt the DOH;
sanction/penalty
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
Demonstrating incompetence or making consistent crrors in the performance of CL examinations and procedures;
sanction/penalty
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
- Deviation from the standard test procedures including use of expired reagents;
-Issuance of flaboratory report without the approval of the head of the laboratory;
-Transferring of laboratory results done by another laboratory to the result form of the
referring laboratory
revoke
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
Permitting unauthorized or unregistered personnel to perform technical procedures and
accESSTO laboratory records/data;
revoke
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
Lending or using the pame af the DOt-licensed CL or the head of the laboratory or
medical technologist to an unlicensed CL;
revoke
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
- Unauthorized use of the name and signature of the pathologist and RMT to secure
LTO;
- Issuance of fraudulent laboratory results, or tests not actuall y done or inaccurate
results,
revoke
State if the given statement below will fall to sanction/penalty or revoke of DOH LTO
- Changein-the-ownership, location, and-head-of the laboratory or laboratory personnel
without informing the HFSRB/CHD-RLED and
- Any material false statement in the application of LTO
three (3) years
All existing licensed CL shall be given ____ years to comply with the physical plant
requirements from the date of effecrtvity of this Order.
two (2) years
All existing licensed CL Shall be given _____ years to fully offer the additional
services for each category with corresponding personnel and equipment from the date
of effectivity of this Order.
True
T or F
For new CL, this Order shall be immediately applicable
True
T or F
For CL currently headed by Anatomic Pathologists with an associate Clinical Pathologist or Clinical Pathologists heading tertiary CL with Anatomic Pathology
services, such headships shall be retained until his/her eventual retirement, resignation or replacement. Thereafter, all CL shall be headed by a patbologist certified in Clinical
Pathology by the Board of Pathology of the Philippine Society of Pathologists, except for tertiary CL with anatomic pathology service which shall be headed by a pathologist
certified in both Anatomic and Clinical Pathology
Familiarize
True
T or F
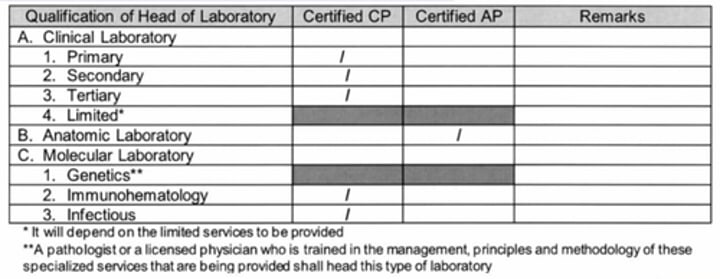
The laboratory shallbe managed by a LICENSED PHYSICIAN certified by the Philippine Board of Pathology
Secondary = Licensed Physician
Tertiary = Pathologist
True
T or F
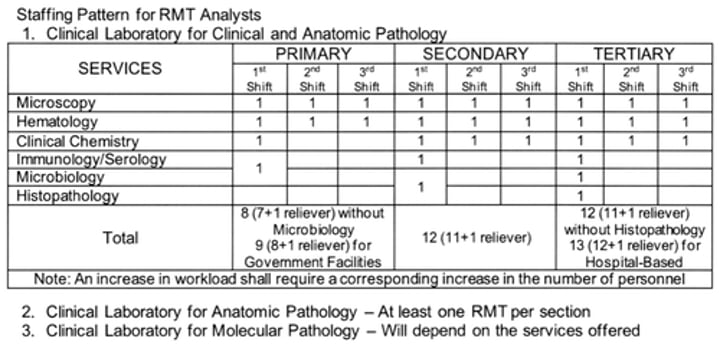
Staffing:
- Qualified and adequately train personnel
a. At least 1 RMT per shift to cover laboratory operation
- Staff development and appropriate continuing education program
Certificate of training on DSSM
(NTRL)
Additional proof of trainings for AFB microscopy
10
Working space in sq. m. for primary cl
20
Working space in sq. m. for secondary cl
60
Working space in sq. m. for tertiary cl
True
T or F
All clinical laboratories shall participate in EQAP given by designated NRL
and or/other recognized reference laboratories
True
T or F
In the renewal of license, the rating of the clinical laboratory should be at least SATISFACTORY PERFORMANCE rating given by the NRL
False
Laboratory requests shall be construed as consultation between the requesting physician and pathologist of the laboratory
T or F
Laboratory requests shall be construed as consultation between the requesting physician and the medical technician of the laboratory
True
T or F
All lab reposts shall bear the name of RMT and the pathologist.