enthalpy
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
heat (new definition)
change in thermal energy of a substance
enthalpy
H
total heat content in a system (at constant pressure)
therefore the difference of thermal energy from reactants to products
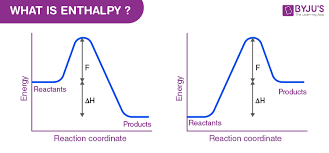
ΔH
change in enthalpy
measures the energy absorbed or released during a chemical reaction
Hreactants - Hproducts
Q vs. ΔH
both represent energy transfer
Q measures total heat added or removed in any process
ΔH heat of reaction (energy per mole) at constant pressure, to determine if endothermic or exothermic
at constant pressure, Qrxn = ΔHrxn
additive reaction enthalpies
uses Hess’s law
states that the enthalpy change of a physical or chemical process depends only on reactants and products
enthalpy change is independent of the pathway of the process and number of steps in the process
enthalpy change will be the same even if more steps are taken to get to the same product
summation of enthalpies
hess’s law
for any chemical change in several steps, net ΔH = sum of ΔH of separate steps
total enthalpy change for a chemical reaction is the same, regardless of whether the reaction occurs in one step or several steps
standard molar enthalpy of formation
quantity of energy associated with the formation of one mole of a substance from its elements in their standard states (at SATP)
ΔH°f
units: kJ/mol