chem chapter 6
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
reaction rate
the change in concentration of a reactant or product over time.
Reactants will have a negative reaction rate, as they are consumed
Products will have a positive reaction rate, as they are produced.
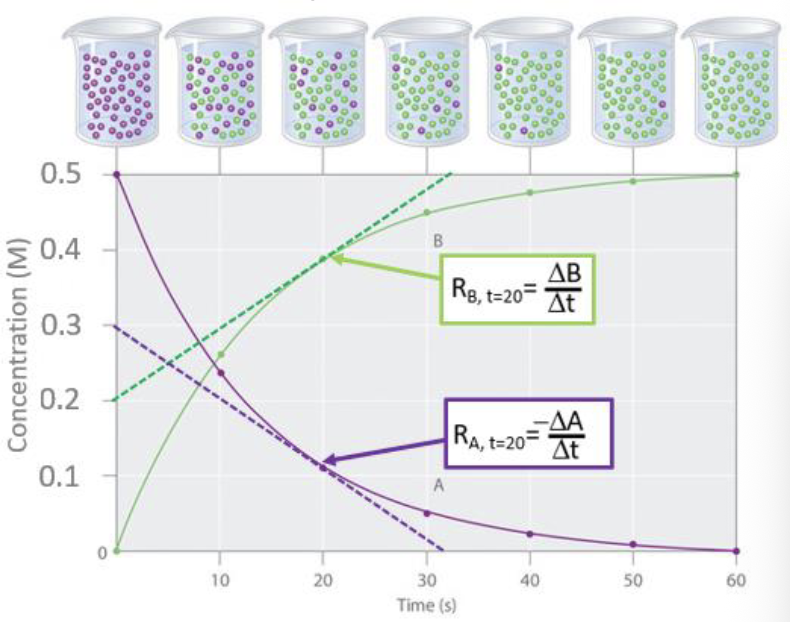
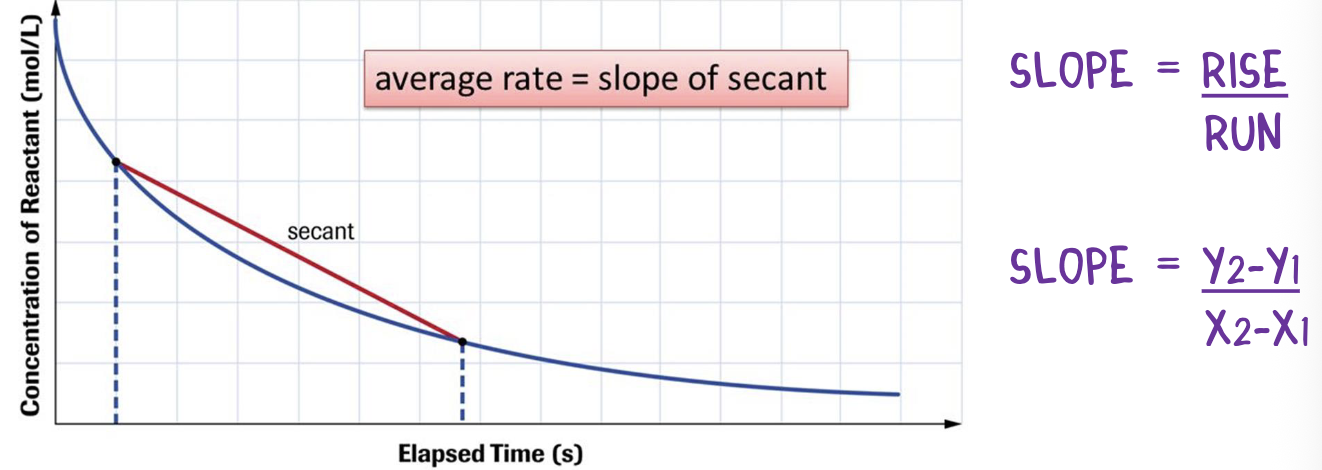
average rate of reaction
the average change in concentration over a given time interval
represented by the slope of a line that is drawn between 2 points on the curve (secant line)
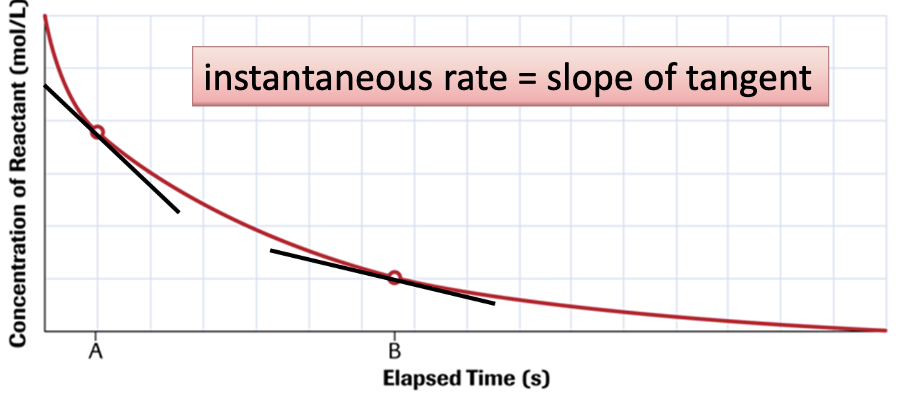
instantaneous rate of reaction
the rate of the reaction at a particular time
represented by the slope of a tangent line to the curve at that time
how to measure reaction rates
a chemist can monitor the rate of appearance of a product or disappearence of a reactant by monitoring observable changes
measuring reaction rates- monitoring colour
• in solutions that involve coloured reactants or products, colour changes can be observed, or its intensity can be measured with a spectrophotometer
e.g. The “iodine-clock reaction” is a reaction with a dramatic colorless-to-blue color change.
The color change occurs when iodine reacts with starch to form a dark blue iodine/starch comple
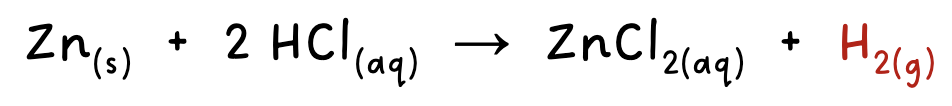
measuring reaction rates- monitoring mass
the decrease in mass can be measured in a reaction when a gas is produced in an open vessel
measuring reaction rates- measuring pH
changes in pH can be measured in reactions where an acid or base are involved
When a reactant is being consumed, the solution will become less acidic and pH will increase

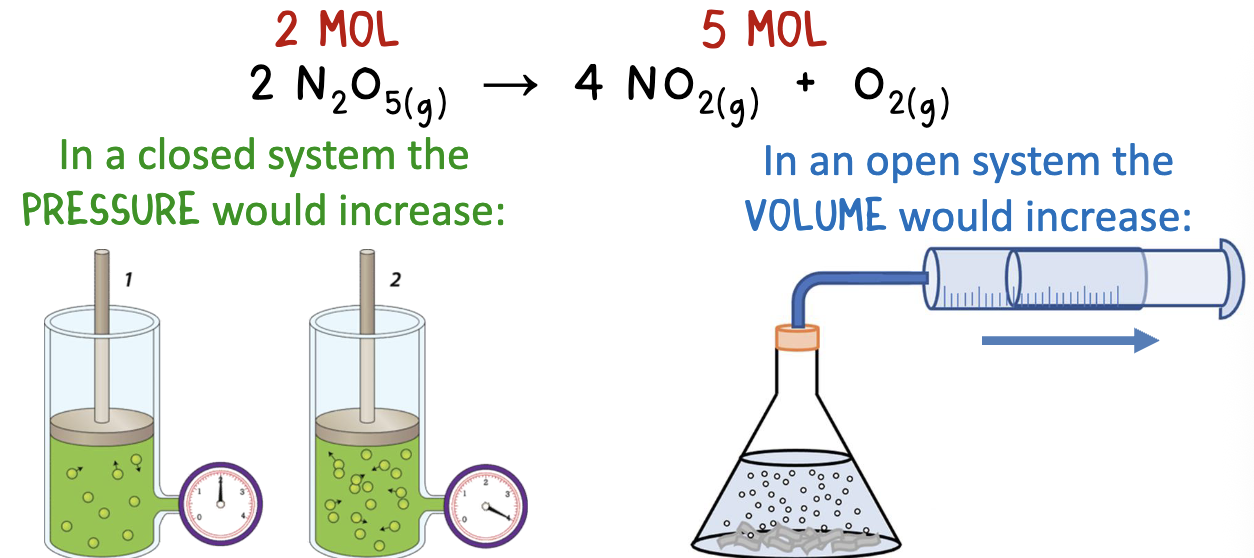
measuring reaction rates- monitoring pressure and volume
when a reaction involve gases, the change in volume or pressure can be measured
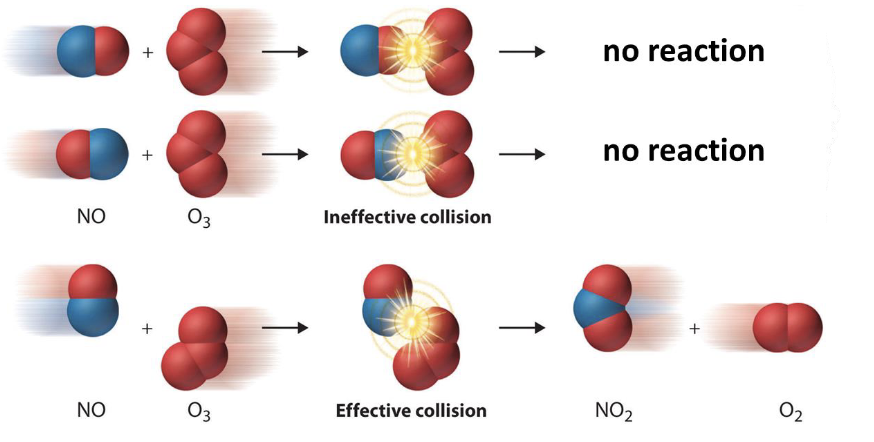
collision theory
a reaction occurs between 2 particles if they collide at the correct orientation and with certain minimum energy
What is needed for a effective collision
molecules must be in the correct orientation
there must be sufficient collision energy
transition state
as a chemical reaction occurs, there exists a transition state, where chemical bonds are partially broken and partially formed
this transition is very unstable
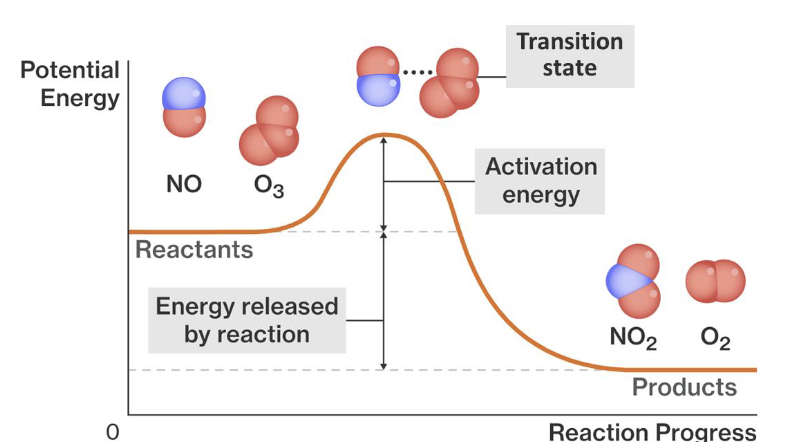
activation energy
the energy required to reach the transition state
the minimum energy required for a successful collision to take place
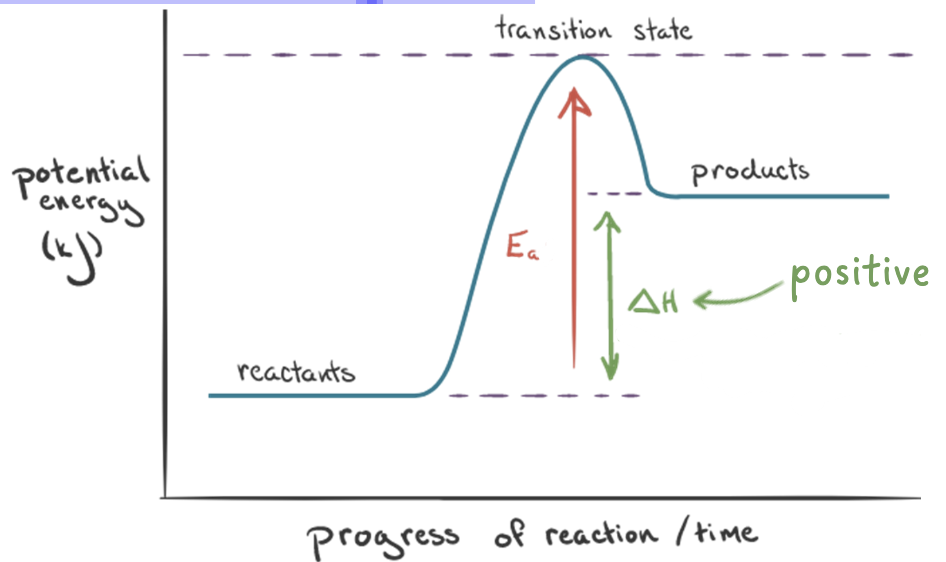
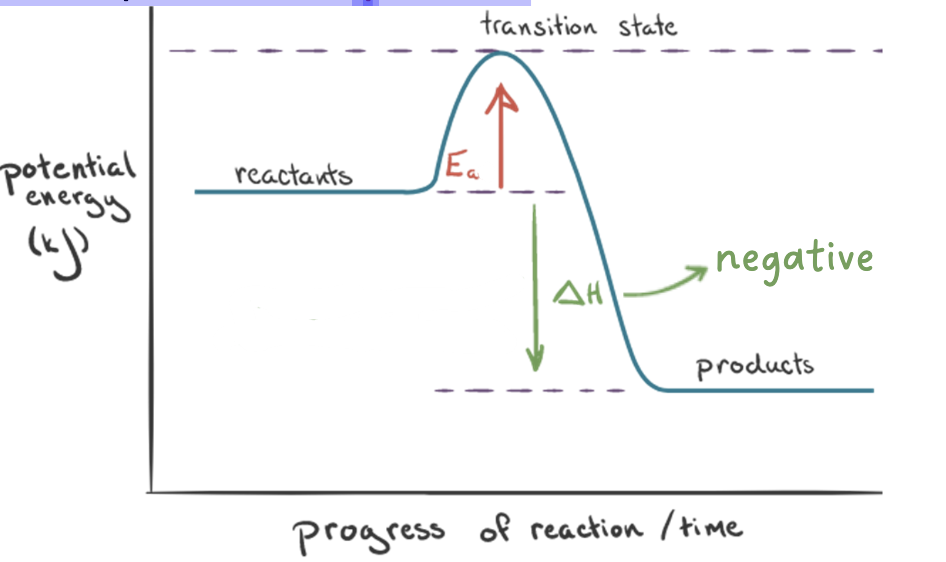
potential energy diagram
shows the relative potential energies of reactants, transition states, and products as a reaction progresses with time
• the enthalpy change, ΔH, is the difference in the energy between the reactants and the products
• the activation energy, Ea, is the difference in the energy between the reactants and the transition state
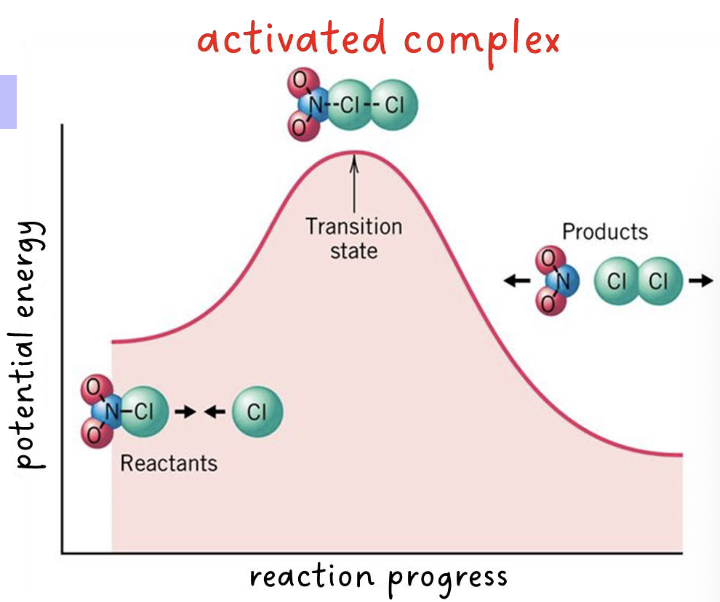
activated complex
the highly unstable structure that forms at the transition state
• results at the maximum energy point along the reaction path
• it is neither a reactant nor product
endothermic reactions
in an endothermic reaction, the reactants are at a lower energy level compared to the products
exothermic reactions
• in an exothermic reaction, the reactants are at a higher energy
level compared to the products
5 factors that affect the rate of a reaction
concentration
temperature
surface area
chemical nature of the reactants
catalysts
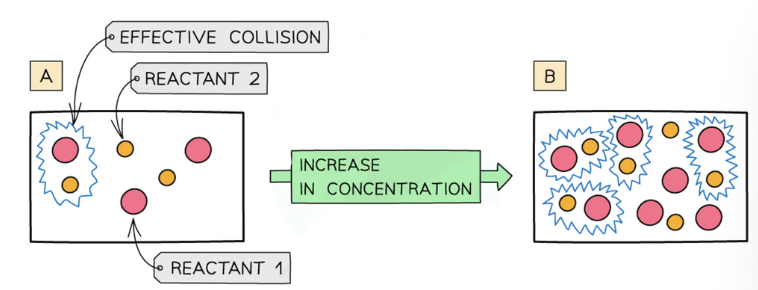
concentration
as concentration increases, the rate increases
when more particles are present, there will be more collisions
as reactants are used up, the reaction slows down
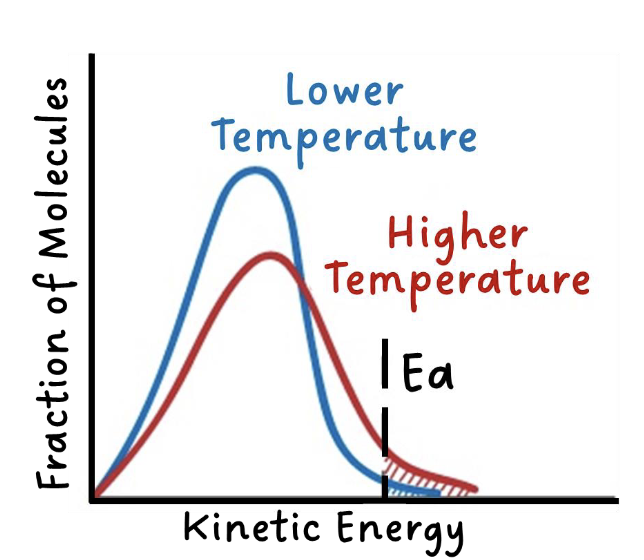
temperature
when temperature increases, the average kinetic energy of the particles increases
more particles have the minimum energy (activation energy) required to react
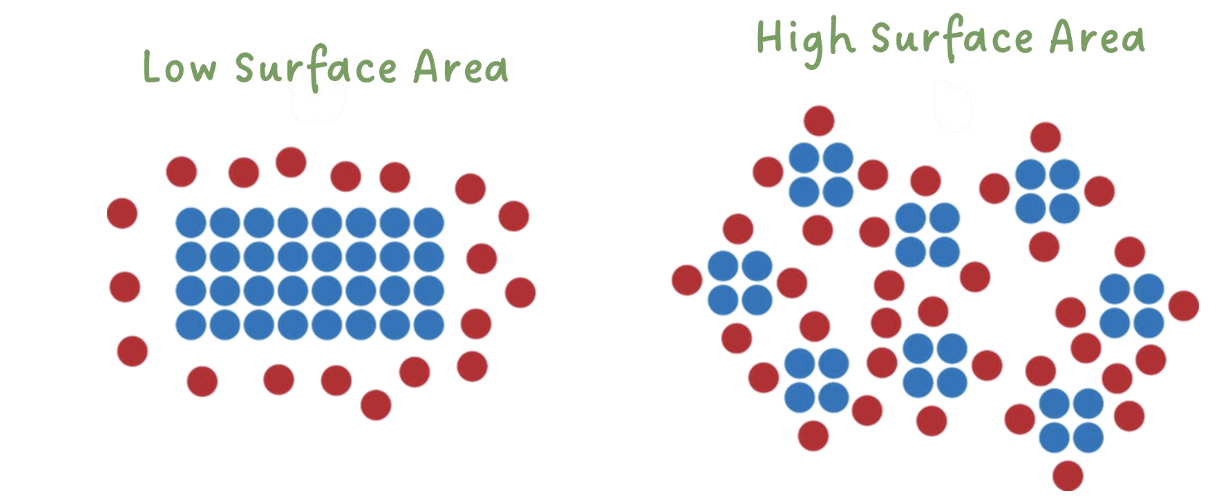
surface area
with greater surface area, there are more particles accessible for collisions
chemical nature of the reactants
a more complicated reaction will react slower than a simple one
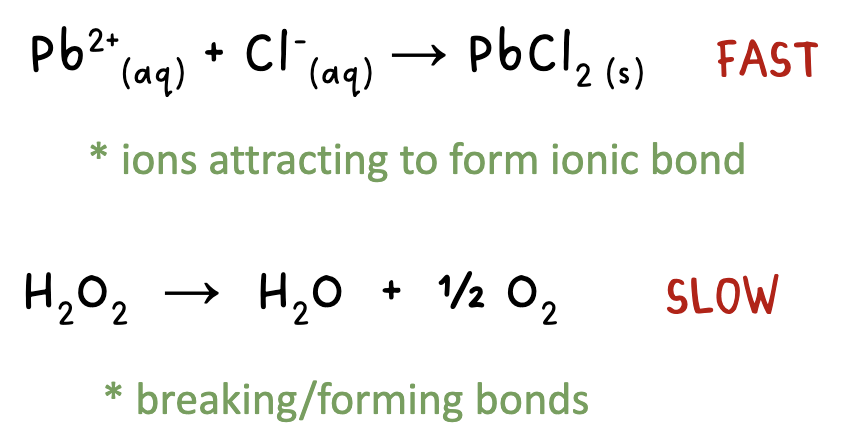
catalysts
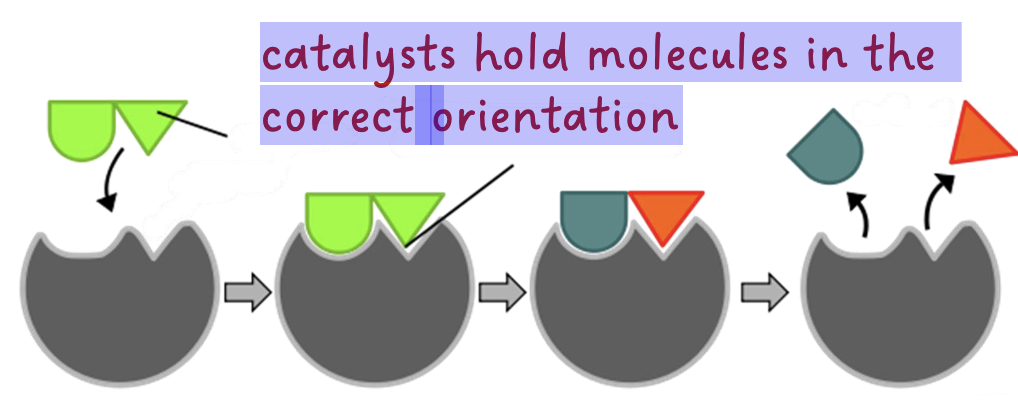
substances that increase the rate of reaction without being consumed
• enzymes are biological catalysts (e.g., lactase, protease) catalysts hold molecules in the correct orientation
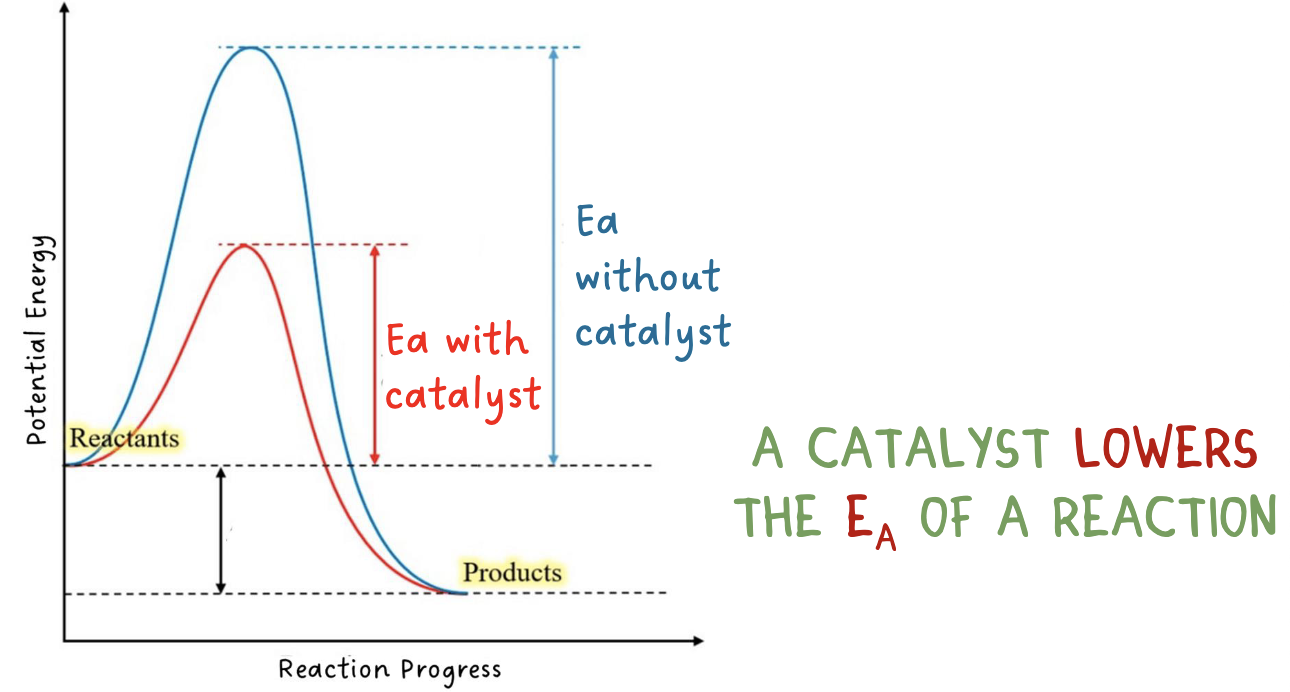
potential energy diagram- reactions with a catalyst
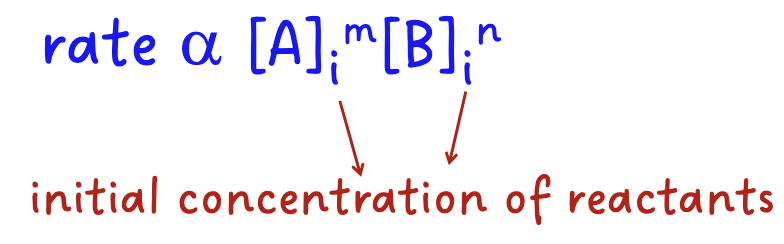
What is the rate law
The rate law is a mathematical expression that relates the rate of a reaction with the concentration of reactants.
The rate of a reaction is exponentially proportional to the initial concentrations of the reactants
reaction mechanism
a series of steps that make up an overall reaction
reaction intermediates
molecules that are formed in an elementary step and consumed in a subsequent elementary step
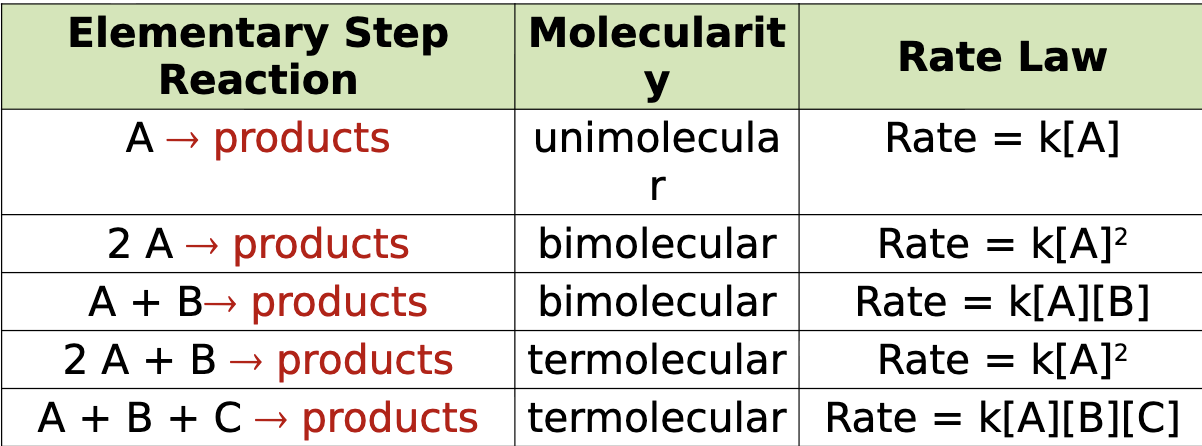
rate law for an elementary step
rate law for an elementary step is determined by its molecularity
requirements for a plausible reaction mechanism
the sum of the elementary steps must give the overall balanced equation for the reaction
the rate-determining step must predict the same rate law that is determined experimentally