Chemistry Unit 3 Flashcards
1/45
Earn XP
Description and Tags
Atomic Structure, Photoelectric Effect, Young's Double Slit, Schrodinger Wavefunction (n, l, ml, Ms), Electron Configurations, (Photoelectric Spectroscopy isn't included b/c u can interpret based off of prior knowledge)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
Photoelectric Effect
Ejection of electrons from the surface of a metal when light shines on it
Conclusions from the Photoelectric Effect
Light behaves both as a particle and a wave.
Not all colors produced current →there is a minimum energy to eject electrons
What did Young’s Double Slit experiment proved
wave-particle duality of light (Acts like a particle when observed but interference pattern suggests waves)
When do electrons emit light?
When dropping from a higher energy level to a lower energy level.
Excited state
When an atom absorbs energy and jumps up to a higher orbital
Ground State
state of lowest energy for an electron
How does the difference between the levels of an electron’s ground state and excited state influence the wavelength of light emitted?
A larger energy gap results in a shorter wavelength (higher energy) of light emitted, and vice versa.
Heisenberg Uncertainty Principle
Impossible to know both the speed and position of an electron
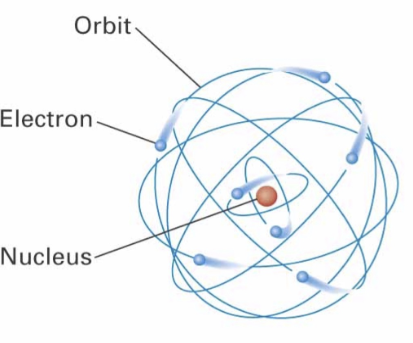
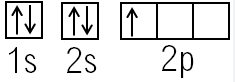
Identify this model:
Bohr Model
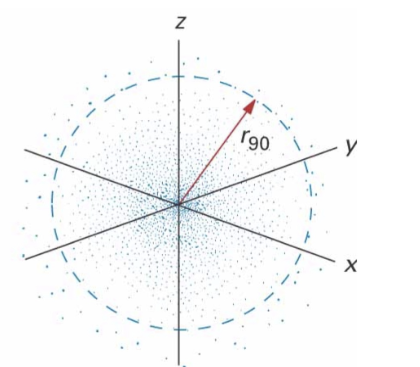
Identify this model:
Quantum Model
What are the 4 quantum numbers unique to each electron in an atom?
Principal quantum number
Angular momentum quantum number
Magnetic Quantum Number
Spin quantum Number

Principal quantum number
abbreviated as n
represents the distance from the nucleus and energy level of the electron (1s, 3d, etc)
as n increases, both orbital size and energy increases
Angular momentum quantum number
abbreviated as cursive l
shape of orbitals , calculated by using probabilities of electron “position
cursive l denotes the energy sublevels

Magnetic Quantum Number
orientation of the orbital in space (and shape)
refers to individual orbitals (each that can contain a max of 2 electrons)

Spin Quantum Number
designates the magnetic properties of an individual electron (spin)

Pauli exclusion principle
no two electrons in an atom can have the same four quantum number
Aufbau principle
When filling electron orbitals, lower energy orbitals fill first. All of the orbitals of the same sublevel (same energy) are filled before a new sublevel begins filling
Hund’s rule
When orbitals of identical energy are available (p, d, or f orbitals), these are 1st occupied singly with parallel spins rather than in pairs
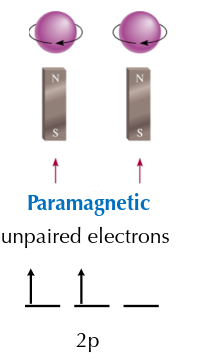
Paramagnetic Substance
weakly attracted by a magnetic field, usually the result of unpaired electrons
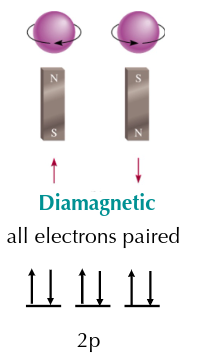
diamagnetic substance
not attracted by a magnetic field generally because it has only paired electrons.
Isoelectronic
same electron configuration
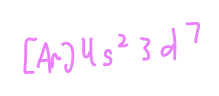
Name the element based on the electron configuration and name the valence electrons
Cobalt and the electrons in the 4s orbital are valence even though the electrons in 3d are written after
Excited state
when an electron configuration doesn’t follow Aufbau’s rule
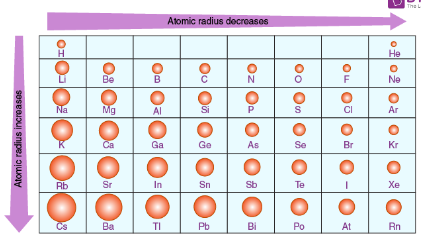
Describe the trend of atomic size as you move down and across the periodic table
Decreases as you move rightwards along period
Increases as you move down a group
According to Coluomb’s Law as the distance between two charges increases
Force of Attraction decreases
According to Coluomb’s Law as magnitude of charges increase
Force of Attraction increases
When using Coulomb’s Law, what does distance refer to?
principal energy levels
How does the Force of attraction between the nucleus and valence electrons change across the periodic table?
As you move down a group: Distance increases as sublevels are added. Effective Nuclear charge remains the same
As you move across a period: Same sublevel → distance is constant. Effective Nuclear charge increases as protons are added
Effective nuclear charge is defined as
number of protons - number of core electrons
(core electrons = total electrons - valence electrons)
What is ionization energy?
amount of energy required to pull off an electron
As force of attraction increases, what happens to 1st ionization energy
Stronger the force of attraction, 1st ionization energy increases
As atomic size increases, what happens to first ionization energy?
Coulomb’s Law states that as distance increases, attractive force decreases → first ionization energy decreases

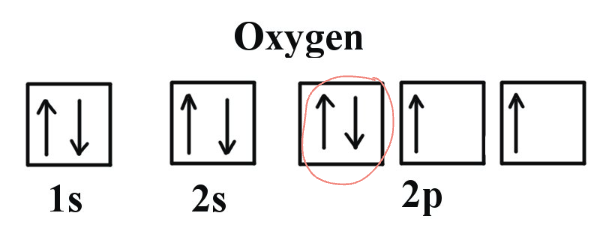
Why is the 1st ionization energy of oxygen lower than that of Nitrogen’s despite periodic trends implying otherwise
oxygen’s 1st ionization energy is lower due to there being electron electron repulsion when the first orbital of the 2s2p sublevel gains a 2nd electron, thus making it easier to remove said electron → lower ionization energy
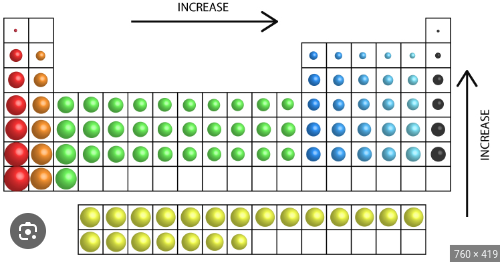
How does ionization energy trend as you move across a period and down a group?
Increases as you move across a period
Decreases as you move down a group
Why is the 1st ionization energy of boron lower than that of beryllium’s despite periodic trends implying otherwise
When finding the place for the 5th electron in boron, you jump up from the 2s sublevel to the 2s2p sublevel, this increase in energy level makes the electron more unstable, making it easier to rip away → decreasing 1st ionization energy
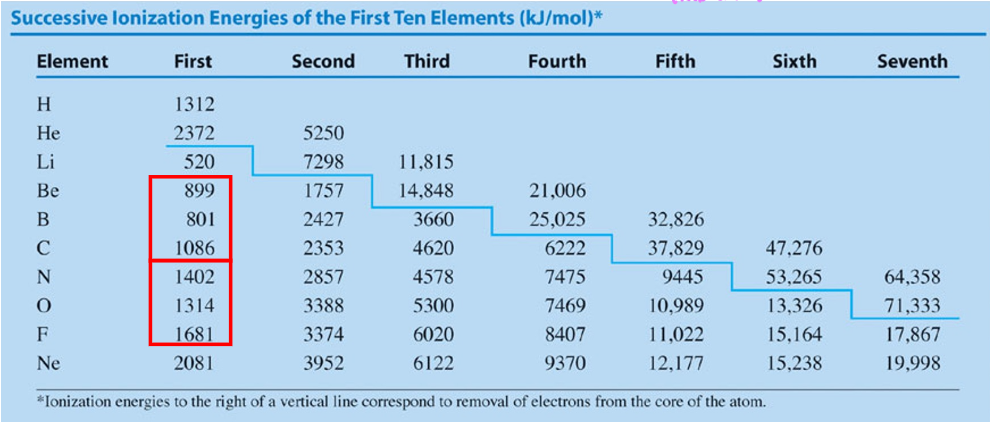
When looking at Nitrogen, why is there a jump in the order of magnitude of the ionization energies when going from 5th to 6th?
When going from taking away the 5th electron to 6th electron, nitrogen loses the 2s2p shell, distance decreases significantly → Force of attraction between the protons and remaining electrons increases → requiring significantly more energy to remove a 6th electron
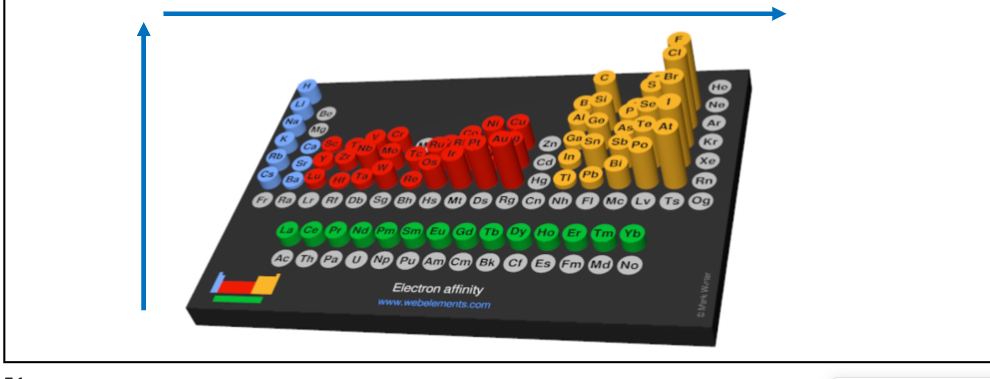
What is electron affinity?
the ability of an atom to accept an electron
The more negative the electron affinity value, the _____ an atom's affinity for electrons
higher
As Coulombic attraction increases, what happens to electron affinity
it increases
What happens to electron affinity as you move across a period and down a group?
Increases as you move rightwards along a period
Decreases as you move down a group
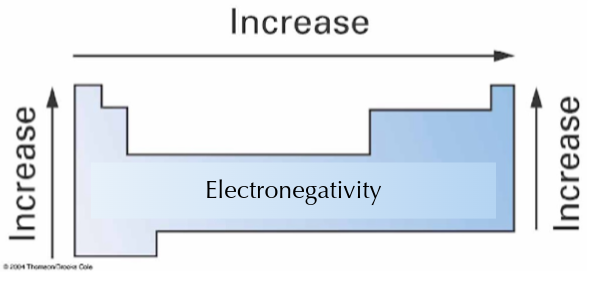
What is electronegativity?
Ability to attract electrons
What are the periodic trends for electronegativity?
Increases as you move rightwards and upwards
How do you calculate whether a bond is polar, no polar, or ionic
check the electronegativity difference between the elements
Cations are always ____ than the atom they were derived from
smaller
Anions are always _____ than the atom they were derived from
larger
Rank these ions in order from increasing size:
S2-, K+, Cl-, Ca2+
Answer: Ca2+, K+, Cl-, S2-
Notice that all these ions have the same electron configuration: [Ar]. Therefore, the only thing differentiating their effective Nuclear charge is the # of protons. More protons means greater attractive force according to Coulomb’s law, leading to a smaller atomic radius due to the attraction