chemistry unit
1/191
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
192 Terms
Neutralization Reaction
A double displacement reaction
occurs when acids and bases are mixed
neutralizes both substances
Forms water and a salt
The hydroxide ion (OH-) and hydrogen (H+) ions
react to form
Water
The metal cation and non-metal anion combine to form a
Salt
The goal of neutralization reaction
to from products with a pH closer to 7 (neutral)
Predicting the Products of Neutralization Reactions
If we know the reactants of a neutralization
reaction, we can predict the products by using the
double displacement model.
Chemical Spills and Neutralization Reactions
Sulfuric acid (H2SO4) and sodium hydroxide
(NaOH)
calcium oxide/ lime Ca0(s) was added
1.CaO(s) + H2O(l) → Ca(OH)2 (aq)
2. Ca(OH)2 (aq) + H2SO4 (aq) → 2H 2O(l) + CaSO4 (aq)
Antacids
Acidic stomach acid can irritate the stomach lining
Antacids contain hydroxide and carbonate neutralize stomach acid
Milk of magnesia Mg(OH)2 is not very soluble in water so it produces a low concentration of hydroxide atoms, so it doesn’t cause chemical burns but provides relief
What’s the PH of stomach
1.5
Neutral pH
A pH level of 7, indicating a neutral solution.
Double Displacement Model
A method used to predict the products of reactions between two ionic compounds.
Calcium Oxide (CaO)
A chemical compound used in the neutralization of sulfuric acid during chemical spills.
Milk of Magnesia
A common antacid that contains magnesium hydroxide (Mg(OH)2) and is used to neutralize stomach acid.
Products of Neutralization
Water and salt formed as a result of an acid-base neutralization reaction.
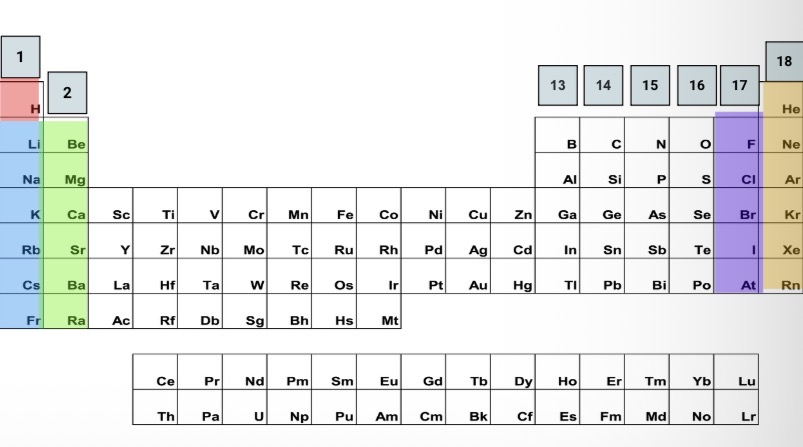
Element groups
Alkali metals
Highly reactive
Alkaline earth metals
Reactive
Halogens
Highly reactive
Noble gases
Not reactive/stable
What are the five main types of chemical reactions?
Synthesis, Decomposition, Single Displacement, Double Displacement, Combustion
What occurs in a synthesis reaction?
Two or more reactants combine to form a new larger product.
What is the general pattern for a synthesis reaction?
A + B → AB
What occurs in a decomposition reaction?
One compound breaks down into two or more smaller compounds or elements.
What is the general pattern for a decomposition reaction?
AB → A + B
What happens in a single displacement reaction?
One element takes the place of another element in a compound.
What is the general pattern for a single displacement reaction?
AB + C → AC + B
Provide an example of a single displacement reaction involving metals.
2HCl + Mg → MgCl2 + H2
What occurs in a double displacement reaction?
The metal ions of two different compounds exchange places.
What is the general pattern for a double displacement reaction?
AB + CD → AD + CB
What is a complete combustion reaction?
Reactions involving the burning of hydrocarbons with oxygen, producing carbon dioxide and water.
Blue, hottest flames, most efficient way to burn energy
What is the general pattern for a complete combustion reaction?
CxHy + O2 → CO2 + H2O
What is an incomplete combustion reaction?
Reactions involving the burning of hydrocarbons with limited oxygen, producing carbon, carbon monoxide, carbon dioxide, and water.
Produces yellow, sooty and cooler flames
What is the general pattern for an incomplete combustion reaction?
CxHy + O2 → C(s) + CO + CO2 + H2O
What is corrosion?
The breakdown of metal as a result of chemical reactions with its environment.
What is the Maillard reaction?
A series of chemical reactions between amino acids and sugars in food, leading to browning and flavor development.
What did Svante Arrhenius discover?
ions were present in aqueous solutions like acids and bases
What ions do acids produce when dissolved in water?
H+ (hydrogen ions)
What ions do bases produce when dissolved in water?
OH- (hydroxide ions)
What is the pH range for acids?
pH < 7
What is the pH range for bases?
pH > 7
What taste is characteristic of acids?
Sour taste
What taste is characteristic of bases?
Bitter taste
Feel of acids
Not slippery
Feel of bases
slippery
Acid reactivity
Reacts with most metals (usually to produce H+ gas)
Base reactivity
No reaction with metals
Acids and bases are
Soluble in water
Can be corrosive
Electrolytes present (forms charged ions in water = conductor of electricity)
What is a common use of hydrochloric acid?
Stomach acid and adjusting swimming pool acidity
What is the formula for hydrochloric acid?
HCl(aq)
What is a common use of sulfuric acid?
Car batteries, making detergents, and cleaning steel
What is the formula for sulfuric acid?
H2SO4(aq)
What is a common use of phosphoric acid?
Tart taste in soft drinks
What is the formula for phosphoric acid?
H3PO4(aq)
What is a common use of Ascetic acid?
vinegar
What is the formula for acetic acid?
HC2H3O2(aq)
What is a common use of sodium hydroxide?
making soap
removing grease
What is the formula for sodium hydroxide?
NaOH(aq)
What is a common use of magnesium hydroxide?
Antacid medicine
What is the formula for magnesium hydroxide?
Mg(OH)2(aq)
What is a common use of magnesium hydroxide?
Controlling the acidity of soil and pool water
What is the formula for calcium hydroxide?
Ca(OH)2(aq)
Acids usually
have hydrogen as the cation and some other anion
Binary acids
contain 2 elements
binary acid naming
Hydrogen becomes “hydro”
Non-metal’s name changes from “-ide” to “-ic”add “acid” at the end

oxyacids
acids with oxygen in the formula
associated with polyatomic ions
oxyacid naming
Drop the hydrogen
“-ate” ending on the polyatomic ion becomes “-ic”
add “acid” at the end

Bases always
Have hydroxide ions as the anion with some other metal as the cation
Base naming
Bases are named as any other ionic compound with an hydroxide ion
Other bases are unique → Ex. ammonia - NH3

What happens to the '-ate' ending of a polyatomic ion in oxyacids?
It becomes '-ic' when naming the acid.
Chemical Formulas of Acids and Bases
Chemical formulas are the same as its ionic compound counterpart, but the symbol (aq) is added to show that it has been dissolved in water.
What is an indicator?
A chemical that changes color in the presence of an acid or a base.
Each has a different range in which it changes colour
Natural Indicators
Tea, Litmus, Cabbage juice
Synthetic Indicators
Phenolphthalein, Bromothymol Blue, Methyl Orange
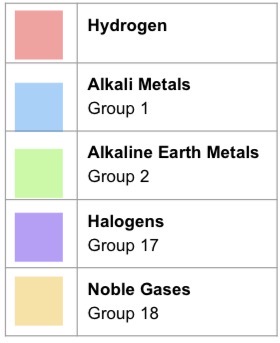
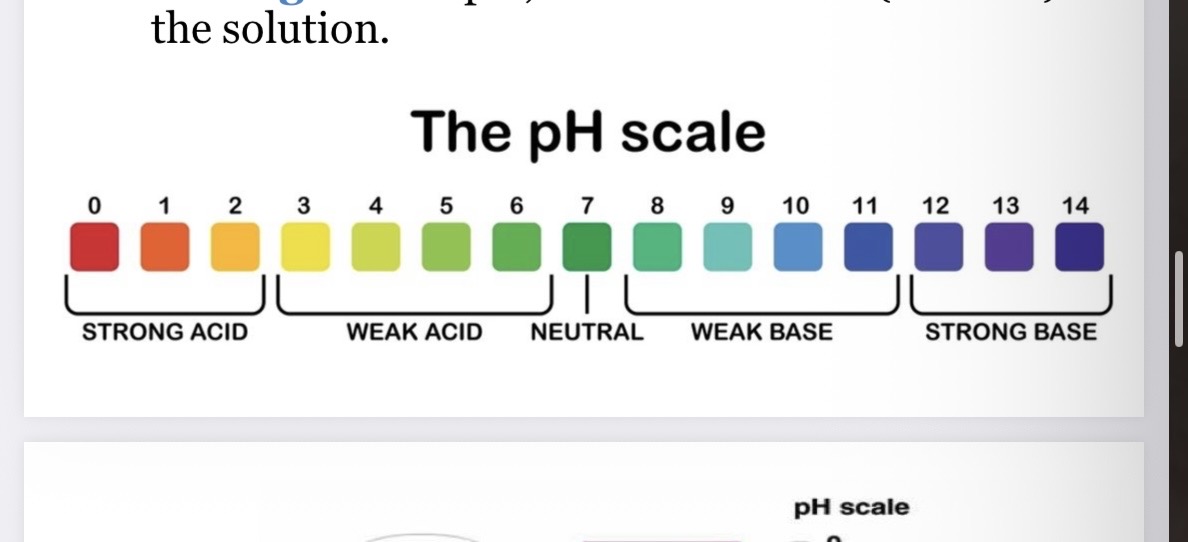
What does the pH scale measure?
The strength of acids and bases
0-14 indicates the level of acidity or alkalinity (basicity) of a solution
What determines a solutions pH value
The concentration of hydrogen ions relative to hydroxide ions disssolved in a solution
Lower pH
More acidic
Higher pH
More basic/alkaline
pH scale
pH stands for
Power of hydrogen
Strength of acids and bases
Depends on how well it can form ions when it dissolves in solution
A strong acid/base
Completely separates into its constituent ions
A weak acid/base
Partially separates into its constituent ions
What does a lower pH indicate?
A more acidic solution.
What does a higher pH indicate?
A more basic (alkaline) solution.
What color does bromothymol blue turn in an acid?
Yellow
What color does bromothymol blue turn in an base?
Blue
What color does phenolphthalein turn in a base?
Pink
pH is a logarithmic scale
A change in 1 pH unit represents a 10-fold increase or decrease in H+ ion concentration
What is the formula for nitric acid?
HNO3(aq)
What is the formula for potassium hydroxide?
KOH(aq)
What color does phenolphthalein turn in an acid?
Colorless
What color does phenolphthalein turn in a base?
Pink
What color does litmus paper turn in an acid?
Red
What color does litmus paper turn in a base?
Blue
Placement of things on the ph scale
types of chemical reactions
Synthesis
Decomposition
Single Displacement
Double Displacement
Combustion
What occurs in a synthesis reaction?
Two or more reactants combine to form a new larger product.
general pattern for a synthesis reaction
A + B → AB
What occurs in a decomposition reaction?
One compound breaks down into two or more smaller compounds or elements.
General pattern for a decomposition reaction
AB → A + B
What happens in a single displacement reaction?
One element takes the place of (displaces) another element in a compound.