23.5 predictions from electrode potentials
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
how to use standard electrode potentials to make predictions about feasibility
a reaction should take place between an oxidising agent and a reducing agent, provided that the oxidising agent has a more positive standard electrode potential value that the redox system of the reducing agent
which way are half cells written in the table
reduction is always the forwards reaction
how to gain the oxidation overall equation from two half equations
reversing the equilibrium then forming the equation
limits of making predictions using standard electrode potential values
some reactions may have a large activation energy
the concentration may not be exactly 1mol/dom³
other conditions may not be standard
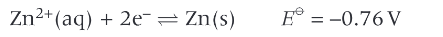
what happens if the concentration of Zn2+ is greater than 1mol/dm³
the equilibrium will shift to the right, removing electrons from the system making the electron potential less negative