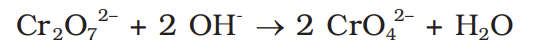08_d-f block elements (only reactions)
1/18
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
how to prepare potassium dichromate from chromite ore?
4Fe2CrO4 + 8Na2CO3 + 7O2 →
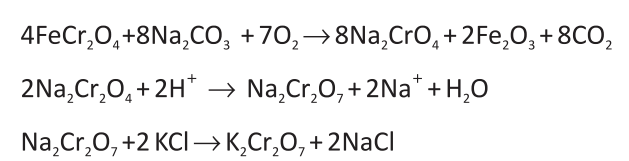
how to prepare potassium permanganate from pyrolusite ore?
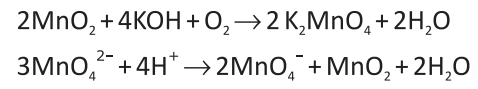
potassium permanganate is a ___________________ agent
strong oxidizing agent
iodine is liberated from potassium iodide
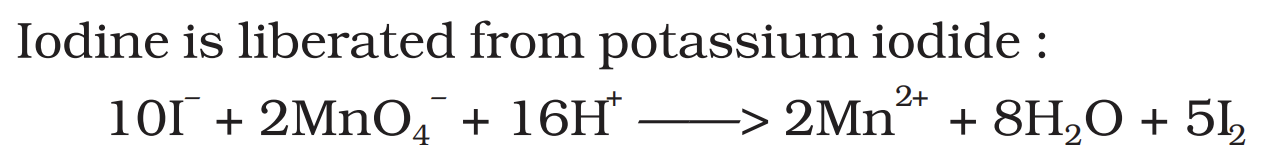
Fe2+ ion is converted to Fe3+
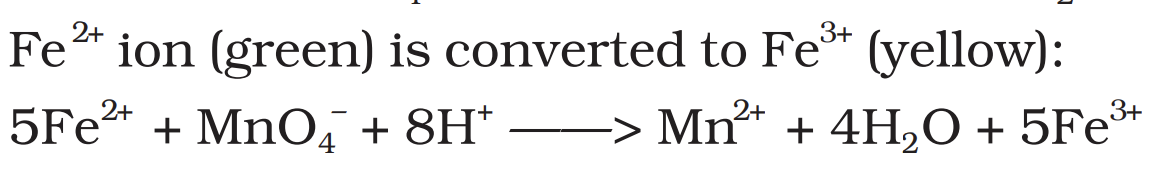
Oxalate ion or oxalic acid is oxidised at 333 K:
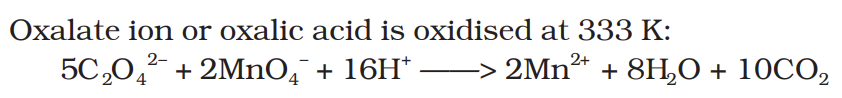
Hydrogen sulphide is oxidised, sulphur being precipitated:
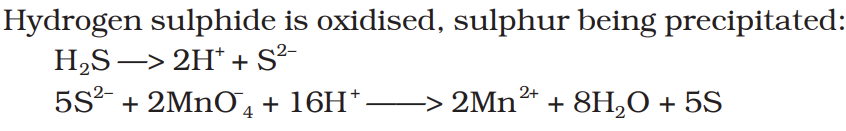
Sulphurous acid or sulphite is oxidised to a sulphate or sulphuric acid
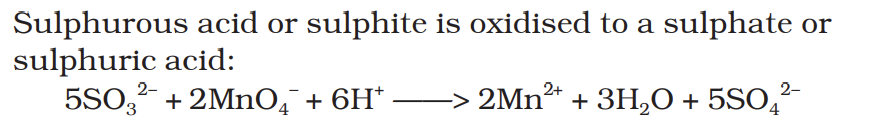
Nitrite is oxidised to nitrate:
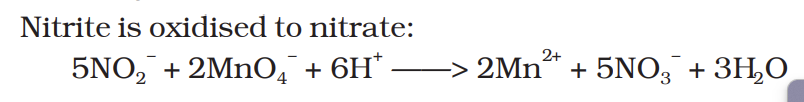
A notable reaction is the oxidation of iodide to iodate:
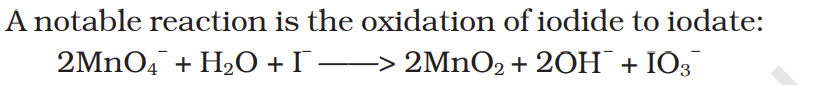
Thiosulphate is oxidised almost quantitatively to sulphate:
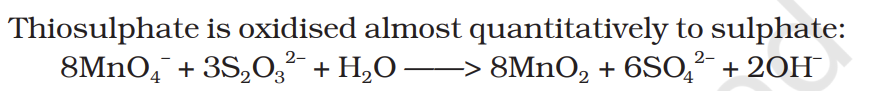
Manganous salt is oxidised to MnO2 ; the presence of zinc sulphate or zinc oxide catalyses the oxidation:
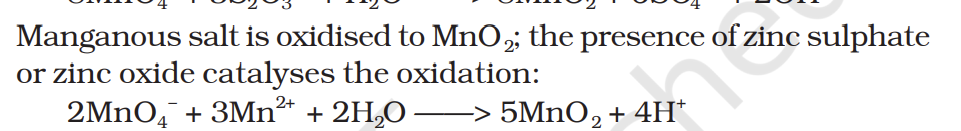
how to convert chromate to dichromate
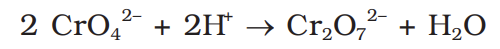
how to convert dichromate to chromate