Valence Bond Theory & Hybridization Theory
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
Valence bond theory
electrons in each bond act independently of the electrons in the other bonds
covalent bond forms when valence orbitals from 2 atoms overlap, unpaired electrons pair up making shared electron density
must have opposite spins
how valence bond theory works
when 2 valence orbitals overlap, potential energy lowers from increased attraction
at ideal distance (bond length) between nuclei, push/pull forces balanced
energy released when bond forms (Bond enthalpy)
Hybridization
introduced to explain molecular structure when the valence bond theory failed to correctly predict them
process of mixing atomic orbitals to form new orbitals with different shapes and energies compared to the originals (hybrid orbitals) better suited for covalent bonding
how orbital hybridization works
An electron may be promoted to create more unpaired electrons. Then, orbitals mix to form hybrid orbitals, which allow atoms to form stable, directional bonds.
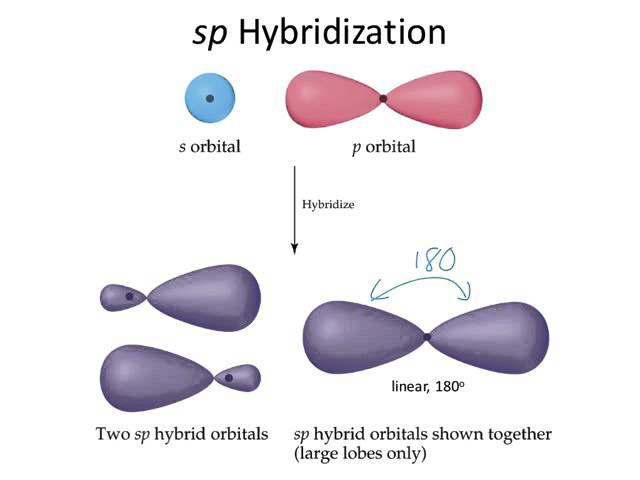
sp hybridization characteristics
1 s & 1 p orbital → 2 sp hybrid orbitals
180° linear fashion
½ s & ½ p character, energies exactly between s and p orbitals
sp² hybridization characteristics
1 s & 2 p orbitals → 3 sp² hybrid orbitals
120° trigonal planar
1/3 s & 2/3 p character
higher energy than sp orbital, closer to original p orbital
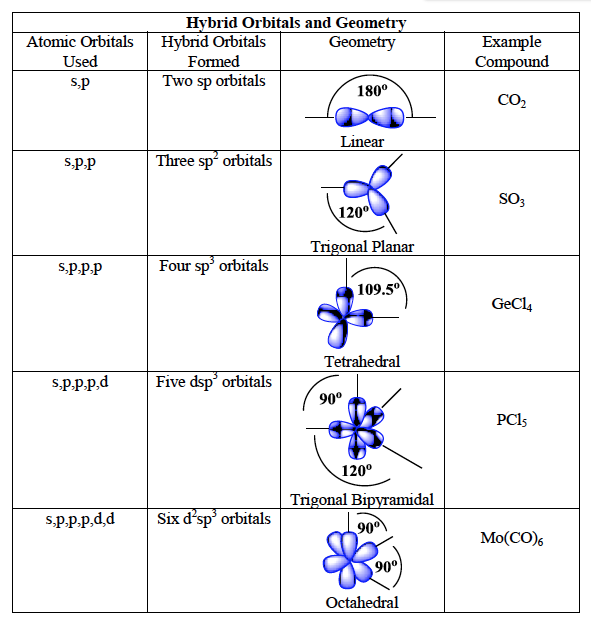
determining the hybridization
Count the number of atoms bonded to the central atom in Lewis structure
Count the number of lone pairs on the central atom and add it to the previous number
If the sum is 2, then it's sp hybridization; if 3, then sp² hybridization; if 4, then sp³ hybridization, and so on.
bond axis
an imaginary line that passes through the centers of two bonded atoms, representing the path along which the chemical bond is formed
Sigma (σ) bonds
formed by the overlap of orbitals in an end-to-end fashion, with the electron density concentrated between the nuclei of the bonding atoms
free rotation around the bond axis
Almost all single bonds are sigma bonds

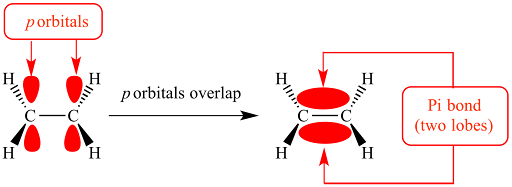
Pi (π) bonds
formed by the sideways overlap of unhybridized p orbitals with the electron density concentrated above and below the plane of the nuclei of the bonding atoms.
pi bond is weaker than a sigma bond
Pi bonds also restrict rotation around the bond axis