Chem Exam 2 Key Points
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Volume increases, Pressure decreases
side with more moles is favored
Volume decreases, Pressure increases
side with less moles is favored

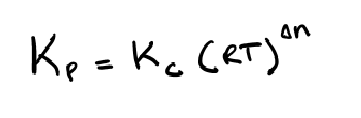
relationship used to convert between the two common types of equilibrium constants: Kp (pressure-based) and Kc (concentration-based).
For the reaction where [n = -1]…what happens after an increase in volume?
Q > K, so the reaction shifts left
For the rection where [n=0]…what happens after an increase in volume?
Q = K the reaction does not shift
For the reaction where [n = +1]… what happens after an increase in volume?
Q < K, the product shifts towards the product
pH =
-log(H+)
pOH =
-log(OH-)
pH + pOH = 14
Kw =
[H+] * [OH-]
[H+] =
10 ^ -pH
[OH-] =
10 ^-pOH
In pH scale… if number lower than 7
Acid = [H+] > [OH-]
In pOH scale… if number lower than 7…
Basic = [OH-] [H+]
In pH scale… if number bigger than 7…
Basic = [OH-] [H+]
In pOH scale…if number bigger than 7…
Acid = [H+] [OH-]
HF ← → H+ + F-
Ka = [H+] [F-] \ [HF]
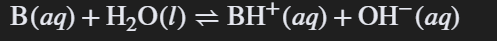
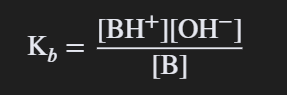
Ka * Kb = Kw
The relationship between the acid dissociation constant (Ka), base dissociation constant (Kb), and the water ionization constant (Kw).
Strong Acid, Strong Base
Neutral
Strong base, Weak Acid
Basic
Strong Acid, Weak Base
Acidic
The stronger the acid, the lower the pKa
Highly electronegative atoms, such as Chlorine, stabilize the conjugate base, making ionization occur more readily. The stronger the acid, the lower the pKa. Increasing the number if Cl atoms further lowers pKa, as does decreasing the distance between the Cl atoms and the site of ionization.
Reverse → flip (1/K)
Multiply coefficients → power (Kⁿ)
Add reactions → multiply Ks
Manipulating K