Bio unit 3
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
The first law of thermodynamics includes all the following except ___________________________
A)The total energy of the universe is constant
B)Energy can neither be created nor destroyed
C)Energy can be transferred or transformed
D)Entropy increases in the universe with every energy transfer or transformation.
D)Entropy increases in the universe with every energy transfer or transformation.
Which of the following is an anabolic process that is endergonic and endothermic?
A)ATP + H2O → ADP + Pi
B)C6H12O6 + 6O2 → 6CO2 + 6H2O + energy
C)CO2 + H2O + light → CH2O + O2
D)FADH2 → FAD + H2
E)CH4 + 2O2 → CO2 + 2H2O
C)CO2 + H2O + light → CH2O + O2
What is the relation between exergonic and exothermic reactions?
A)They are typically unrelated, but can be coupled with ATP hydrolysis
B)Every exothermic reaction leads to a subsequent exergonic one
C)Every exergonic reaction is also an exothermic one
D)Both result in a reduction of entropy in the universe
E)Exothermic reactions may be endergonic or exergonic processes
E)Exothermic reactions may be endergonic or exergonic processes
Melting of ice at room temperature results in the following changes in free energy (ΔG), enthalpy (ΔH), and entropy (ΔS).
A)+ΔG, +ΔH, +ΔS
B)+ΔG, -ΔH, +ΔS
C)+ΔG, -ΔH, -ΔS
D)-ΔG, +ΔH, +ΔS
E)-ΔG, +ΔH, -ΔS
D)-ΔG, +ΔH, +ΔS
Photosynthesis reactions are summarized below in the given equation.
6CO2 + 6H2O + light → C6H12O6 + 6O2
Which of the following changes will occur in this process with respect to free energy, enthalpy, and entropy.
A)+ΔG, +ΔH, +ΔS
B)+ΔG, -ΔH, +ΔS
C)+ΔG, +ΔH, -ΔS
D)-ΔG, +ΔH, +ΔS
E)-ΔG, +ΔH, -ΔS
C)+ΔG, +ΔH, -ΔS
If the Keq of a reaction is 10, when you increase the substrate concentration 2 fold, the Keq will _______________________
A)increase 2 fold also
B)decrease by 2 fold
C)stay the same
D)increase by 20 fold
E)decrease by 20 fold
C)stay the same
DNA replication using the high energy deoxy nucleoside triphosphates (dNTPs) as the monomers is a/an _______________________ reaction
A)endergonic
B)exergonic
C)both
D)neither
B)exergonic
ATP hydrolysis yields a ΔG of -7.3 Kcal/mol free energy, and it is coupled with many enzyme reactions that would not otherwise happen (endergonic). Which of the following reactions can be coupled with ATP hydrolysis to become a favorable or spontaneous reaction? A reaction with a ΔG value of _______ Kcal/mol.
A)-7.8
B)4.3
C)10.5
D)-5.3
E)7.3
B)4.3
Given the same concentration of substrate, which of the following reactions will yield the maximum amount of products?
A)Enzyme alone
B)Enzyme + substrate alone
C)Enzyme + substrate + competitive inhibitor
D)Enzyme + substrate + non-competitive inhibitor
E)Enzyme + Substrate + Activator
E)Enzyme + Substrate + Activator
In an enzyme-catalyzed reaction, which of the following does not change with or without enzyme?
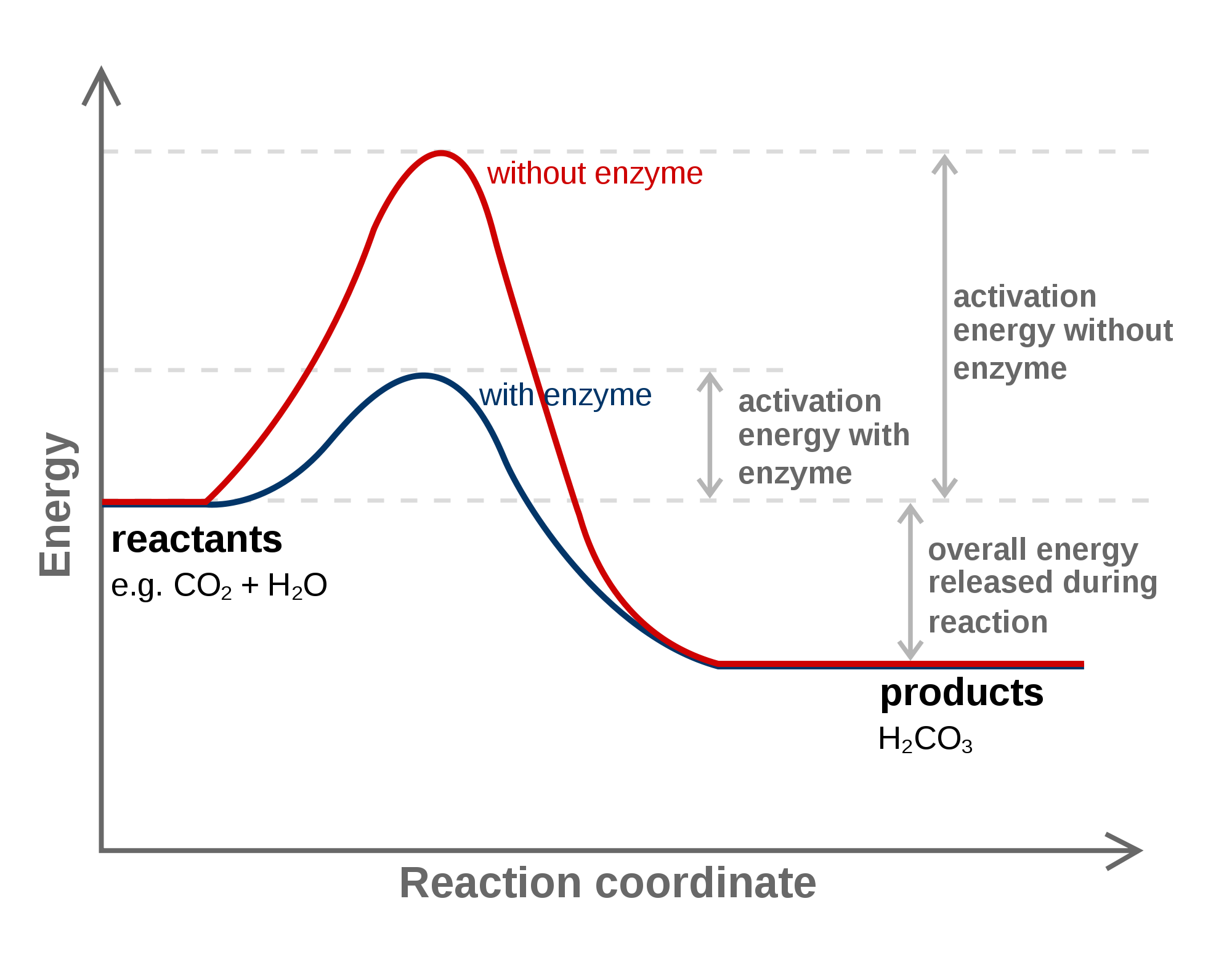
A)Reactants
B)Products
C)Change in activation energy
D)Net change in free energy
D)Net change in free energy
As you add more and more substrates, the velocity of enzyme reaction increases and as it reaches above a certain point, it plateaus because of what reason?
______
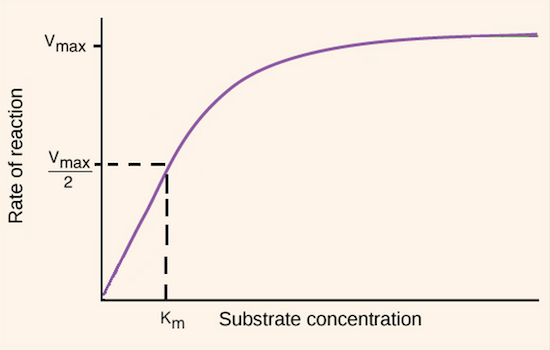
A)Enzyme is denatured and can no longer funcion effectively.
B)Substrates run out and enzymes can no longer convert them to products
C)All enzymes are busy catalyzing the reactions at saturating substrate concentration.
D)All things need to come to an end and so are the enzyme reactions.
E)Enzymes become inactive at saturating substrate concentration.
C)All enzymes are busy catalyzing the reactions at saturating substrate concentration.
An enzyme was tested with two different substrates A and B resulting in products X and Y. The Km for substrate A was 10 mM and the Km for substrate B was 100 mM. Which of the following statement is correct?
A)Substrate A is less effective than substrate B to have same amount of products formed.
B)Substrate B is less effective than substrate A to have same amount of products formed.
C)There is no difference between A and B in terms of products formed.
D)X is produced less than Y with same amount of substrate used.
E)Y is produced at same level as X with same amount of substrate used.
B)Substrate B is less effective than substrate A to have same amount of products formed.
If someone is accidentally poisoned by methanol, ethanol is given as an antidote.
Note: Ethanol is converted to acetaldehyde by Alcohol Dehydrogenase (ADH) and methanol is converted to formaldehyde by the same enzyme. Acetaldehyde can be detoxified but formaldehyde cannot be detoxified by humans.
How does ethanol serve as an antidote for methanol poisoning?
A)Ethanol binds to methanol and inactivates it by adding another -OH group.
B)Methanol reacts with ethanol and both becomes non-toxic.
C)Ethanol is an activator stimulating the synthesis of acetaldehyde and formaldehyde.
D)Ethanol is a preferred substrate that keeps the enzyme busy in making acetaldehyde and it prevents formaldehyde formation.
E)Methanol is a preferred substrate that keeps the enzyme busy in making formaldehyde and stores it for future use.
D)Ethanol is a preferred substrate that keeps the enzyme busy in making acetaldehyde and it prevents formaldehyde formation.
A biochemical pathway is depicted below with the chemicals made from the beginning to end.
A --> B --> C --> D --> X --> Y --> Z
Which of the following chemical if accumulates above certain level will inhibit the key regulatory enzyme and stop this pathway?
A)A
B)B
C)C
D)X
E)Y
F)Z
F)Z
The energy is stored in the form of ________ in the food we eat.
A)acetyl CoA
B)fructose
C)proteins
D)ATP
E)covalent bonds
E)covalent bonds
Overall, respiration is a/an____________________ and _______________________
A)oxidative........endergonic
B)oxidative.....exergonic
C)reductive.....endergonic
D)reductive....exergonic
E)endergonic.....redox process
B)oxidative.....exergonic
The following reaction, with reference to FAD, is a __________________ process and FAD is a/an ______________(oxidizing or reducing) agent.
FAD + X-H2 --> FADH2 + X
A)reduction…. …oxidizing
B)oxidation….….reducing
C)reduction… …reducing
D)oxidation…. …oxidizing
A)reduction…. …oxidizing
The reaction shown below is a/an ___________________ and it is catalyzed by _______.
Phosphoenolpyruvate + ADP --> Pyruate + ATP
A)reduction…. kinase
B)phosphorylation…..phosphatase
C)oxidative phosphorylation…….ATP synthase
D)substrate level phosphorylation…….ATP synthase
E)substrate level phosphorylation…….kinase
E)substrate level phosphorylation…….kinase
Which of the following are NOT needed to perform glycolysis? Select all that apply.
Select all that apply
A)Glucose
B)NAD+
C)ADP
D)Pi (inorganic phosphate)
E)NADH
F)ATP
G)Oxygen
E)NADH;
G)Oxygen
Which output of Glycolysis will enter the next step of Acetyl CoA formation?
A)ATP
B)Pyruvate
C)ADP
D)NADH
E)Ethanol
B)Pyruvate
Which key regulatory enzyme of glycolysis will be inhibited by ATP and citrate?
A)Hexokinase
B)Phosphofructrokinase
C)Triose isomerase
D)Aldolase
E)RuBisCo
B)Phosphofructrokinase
Which of the following processes can occur with or without oxygen?
A)Acetyl CoA formation
B)Glycolysis
C)Krebs Cycle
D)Oxidative phosphoprylation
B)Glycolysis
More free energy is released during the Krebs cycle than during glycolysis, but only 2 moles of ATP is produced for each mole of glucose undergoing glycolysis. Most of the remaining free energy that is produced during the Krebs cycle is used to _______________________
A)synthesize GTP.
B)reduce NAD+ and FAD.
C)release as heat.
D)oxidize pyruvate.
E)make ethanol.
B)reduce NAD+ and FAD.
When mice were fed with radioactive carbon in glucose (C6H12O6) all the 6 CO2 molecules will be released during which of the following process
A)Krebs cycle and Oxidative phosphorylation
B)Glycolysis and acetyl CoA formation
C)Glycolysis and Krebs cycle
D)Acetyl CoA formation and Krebs cycle
E)Glycolysis and oxidative phosphorylation
D)Acetyl CoA formation and Krebs cycle
When one consumes excess amounts of carbohydrates, they gain weight and store excess energy as fat. Conversion of carbohydrates to fat will involve the intermediate _________.
A)ATP.
B)Starch.
C)Citrate.
D)NADH.
E)Acetyl CoA.
E)Acetyl CoA.
Which of the following poisons will dissipate Δ [H+]?
A)A poison that would inhibit ATP synthase
B)A chemical that would inhibit citrate synthase
C)A chemical that would block the substrate level phosphorylation
D)A chemical that would make the inner mitochondrial membrane leaky to ions
E)A chemical that mimics glucose but is not metabolized by hexokinase
D)A chemical that would make the inner mitochondrial membrane leaky to ions
Which of the following is an exergonic process that directly helps the endergonic process of ATP synthesis in oxidative phosphorylation?
A)Acetyl CoA formation and krebs cycle
B)Ethanol fermentation and ATP synthase
C)Chemiosmosis and proton motive force
D)Glycolysis and pyruvate oxidation
E)Substrate level phosphorylation and Kinases
C)Chemiosmosis and proton motive force
What is the main role of oxygen in aerobic respiration?
A)It activates ATP synthase enzyme
B)It is required for Glycolysis and Krebs Cycle
C)It is an electron carrier to provide electrons to the elctron transport chain
D)It is an output of Oxidative Phosphorylation
E)It is the ultimate electron acceptor in Oxidative Phosphorylation
E)It is the ultimate electron acceptor in Oxidative Phosphorylation
When we exercise for a long time without rest, the muscle cells become deprived of oxygen. Which of the following is not produced in a muscle cell under such conditions?
A)NADH
B)Ethanol
C)ATP
D)Pyruvate.
E)Lactate.
B)Ethanol
How does the reduction of pyruvate to lactate during fermentation allow glycolysis to continue in the absence of oxygen?
A)Water is formed during this reaction.
B)This reaction is an endergonic reaction.
C)This reaction is coupled to the oxidation of NADH to NAD+.
D)This reaction is coupled to the formation of ADP and Pi.
E)This reaction is coupled to the reduction of NAD+ to NADH.
C)This reaction is coupled to the oxidation of NADH to NAD+.
Migratory birds store or carry limited amounts of stored energy or food but fly over long distances. Which of the following process is most utilized in such situations to make the most amount of ATP for their flying?
A)Glycolysis
B)Ethanol fermentation
C)Lactate fermentation
D)Krebs cycle
E) Oxidative phosphorylation
E)Oxidative phosphorylation
When the cells run out of oxygen, the Glycolysis and Krebs Cycle come to a grinding halt even though oxygen is not an input to either of these processes. Why do these processes stop in the abscence of oxygen?
A)Oxygen indirectly helps these processes by making water.
B)Oxygen is the electron carrier that will be essential for these processes to make CO2.
C)Without oxygen accepting the electrons, NAD+ and FAD are not generated.
D)Lack of oxygen produce excess of NAD+ and FAD
E)Lack of oxygen decreases the pH in matrix which inhibits the Glyucolysis and Kreb Cycle.
C)Without oxygen accepting the electrons, NAD+ and FAD are not generated.
The primary function of light energy in photosynthesis is to ____________________ .
A)make carbohydrates from CO2 and water
B)release O2 from splitting water
C)make ATP and NADPH
D)excite electrons in the photosystems I and II
E)fix CO2 into organic molecules
D)excite electrons in the photosystems I and II
The ultimate source of electrons used in photosynthesis is _________________.
A)CO2
B)NADPH
C)O2
D)H2O
E)CH2O
D)H2O
When does cyclic electron flow (at Photosystem I) occur instead of non-cyclic electron flow?
A)When there is abundant ATP.
B)When there is shortage of NADPH.
C)When there is shortage of ATP.
D)When there is abundance of NADPH.
E)When there is equal amount of ATP and NADPH.
D)When there is abundance of NADPH.
During photophosphorylation, the pH of the thylakoid interior will be _______________
A)higher than the pH of stroma since protons move into thylakoid
B)lower than the pH of stroma since protons move into thylakoid space.
C)same as the the pH of stroma since protons go in and out of thylakoid space
D)higher than the pH of stroma since protons move into stroma from thylakoid space
E)lower than the pH of stroma since protons move into the intermembrane space from stroma
B)lower than the pH of stroma since protons move into thylakoid space.
Suppose a thylakoid is somehow punctured so that the interior of the thylakoid is no longer separated from the stroma. This damage will have the most direct effect on which of the following processes?
A)the absorption of light energy by chlorophyll
B)the splitting of water
C)the flow of electrons from photosystem II to photosystem I
D)the reduction of NADP+
E)the synthesis of ATP
E)the synthesis of ATP
The electrons lost by PS I is replaced by the electrons coming from PS II. The electrons lost by PSII are replaced by the electrons coming from the ____________________________.
A)carbon dioxide coming through the stomates
B)glucose stored in the leaves.
C)redox reactions of mitochondrion.
D)splitting of water in the non-cyclic photophosphorylation.
E)from the electron carriers NADH and FADH2
D)splitting of water in the non-cyclic photophosphorylation.
The water used in light reactions reach the mesophyll cells in the leaves via ________________ transported from the roots through pressure gardient.
A)stomates
B)plasmodesmata
C)aquaporins
D)phloem
E)xylem
E)xylem
Which outputs of light reactions enter Calvin-Benson cycle?
A)ATP
B)NADPH
C)O2
D)A and B
E)A, B and C
D)A and B
When a suspension of algae is incubated in a flask in the presence of light and CO2 and then transferred to the dark, the reduction of 3-phosphoglycerate to glyceraldehyde 3-phosphate is blocked. This reaction stops when the algae are placed in the dark because
A)the reaction requires CO2.
B)the reaction is exergonic.
C)the reaction requires ATP and NADPH
D)the reaction requires O2.
E)chlorophyll is not synthesized in the dark.
C)the reaction requires ATP and NADPH
CAM plants can keep stomata closed in daytime, reducing water loss. This is possible because they __________.
A)fix CO2 into organic acids during the night by PEP carboxylase
B)use photosystems I and II during day and night.
C)use the enzyme phosphofructokinase, to fix CO2 more effectively
D)fix CO2 into sugars in the bundle-sheath cells.
E)fix CO2 into pyruvic acid in the mesophyll cells
A)fix CO2 into organic acids during the night by PEP carboxylase
When RuBisCO enzyme was modified to reduce the binding of oxygen to minimize photorespiration, the enzyme activity to fix CO2 to carbohydrate was also decreased. A possible explanation to this result is _______________.
A)carbon fixation reaction requires O2.
B)modified enzyme prefers O2 and not CO2.
C)modified enzyme prefers CO2 and not O2.
D)modified enzyme lost the binding to both O2 and CO2.
E)carbon fixation reaction requires light and water.
D)modified enzyme lost the binding to both O2 and CO2.
The sugars made by photosynthesis are transported to other parts of the plants via ____________
A)Xylem
B)Ploem
C)Plasmodesmata
D)Stomates
E)Mesophyll cells
B)Ploem
Which of the following outputs of Calvin Cycle become inputs for Light Reactions? Select all that apply.
Select all that apply
A)ATP
B)NADPH
C)ADP, Pi
D)NADP+
E)NAD+
F)FAD
G)NADH
H)FADH2
C)ADP, Pi
D)NADP+
What is the main precursor derived mostly from carbohydrates to start fat biosynthesis?
A)Pyruvate
B)Citrate
C)Acetyl CoA
D)Oxaloacetate
E)ATP
C)Acetyl CoA
Which of the following enzymes is the key regulatory enzyme in the fatty acid biosynthesis pathway?
A)Pyruvate dehydrogenease
B)Citrate synthase
C)Fatty acid synthase
D)Acetyl CoA carboxyalse
E)Phosphofructokinase
D)Acetyl CoA carboxyalse
What is the key regulatory enzyme for steroid biosynthesis in animals?
A)Phosphofructokinase
B)Hexokinase
C)ATP Synthase
D)HMG CoA Reductase
E)PEP Carboxylase
D)HMG CoA Reductase
What is the main process of fat break down to generate acetyl CoA?
A)Glycolysis
B)Beta oxidation
C)Krebs cycle
D)Susbtrate level phosphorylation
E)Chemosynthesis
B)Beta oxidation
Which of the following cell structures makes all the other membrane structures?
A)Lysosome
B)Smooth ER
C)Vauole
D)Peroxisome
E)Rough ER
E)Rough ER
How is the LDL cholesterol taken up and broken down in the cells?
A)Receptor mediated endocytosis
B)Phagocytosis
C)Pinocytosis
D)Osmosis
E)Facilitated diffusion
A)Receptor mediated endocytosis
What is the precursor to start steroid biosynthesis?
A)Acetyl CoA
B)Pyruvate
C)Fattyacids
D)Glucose
E)Fructose
A)Acetyl CoA
What is the precursor to Retinol, the pigment in the eye?
A)Cholesterol
B)Fatty acid
C)Phospholipid
D)Beta carotene
E)Acetyl CoA
D)Beta carotene
Which of the following cells and/or structures divide by binary fission? Select ALL answers that apply.
Select all that apply
A)Bacteria
B)Chloroplast
C)Protist
D)Fungi
E)Mitochondria
F)Plant cells
A)Bacteria
B)Chloroplast
E)Mitochondria
All the cells in human body except those cells that make sperm and eggs divide by _____________
A)Binary fission
B)Mitosis
C)Meiosis
D)A and B
E)A, B and C
B)Mitosis
Which of the following statements about mitosis is true?
A)The chromosome number in the resulting cells is halved.
B)DNA replication is completed in prophase.
C)Crossing over occurs during metaphase.
D)Replicated Nuclear DNA is equally divided in to two cells
E)It consists of two nuclear divisions and single cell division.
D)Replicated Nuclear DNA is equally divided in to two cells
Chromosomes move to the opposite poles by the ___________________________.
A)microfilaments pulling them to wards the poles.
B)microfilaments pushing them towards the center.
C)Kinetochore microtubules pull them towards the poles.
D)non-kinetochore microtubulues push them towards the poles.
E)motor proteins attached to the microtubules help in the migration.
E)motor proteins attached to the microtubules help in the migration.
THE MECHANISM OF COLCHICINE INHIBITION OF MITOSIS I. Kinetics of Inhibition and the Binding of H~-Colchicine EDWIN W. TAYLOR, Ph.D. 1965. From the Committee on Biophysics, The University of Chicago
ABSTRACT Ha-colchicine of high specific activity (2.5 curies per mM) was prepared in order to study the mechanism of colchicine inhibition of mitosis in cultures of human cells, strain K.B. No direct effects on the duration of the cell cycle or macromolecular synthesis were demonstrable at a concentration of colchicine which completely inhibited mitosis. The radioactive compound was bound to the cells at a rate proportional to colchicine concentration. The binding appeared to be reversible since the radioactivity of the cells reached a maximum value for a given concentration and was slowly lost after resuspension of the cells in fresh medium. A suitable exposure to colchicine produced accumulation of metaphase-blocked mitoses after the colchicine was removed from the medium. An exposure of 6 to 8 hours at l0 -7 M was sufficient to block essentially all the cells in metaphase, thus indicating that colchicine is bound to the majority of interphase cells. The data are in quantitative agreement with a mechanism involving reversible binding of colchicine to a set of cellular sites. Based on the correlation between the time of first appearance of blocked mitoses and the radioactivity per cell, it is suggested that if a critical fraction (3 to 5 per cent) of the sites are complexed, the cell is unable to form a functional mitotic spindle.A microtubule polymerization inhibitor, Colchicine is added to actively growing cells. Most of the cells would be found with their ______ phase arrested.
A)Prophase
B)Prometaphase
C)Metaphase
D)Anaphase
E)Telophase and cytokinesis
C) Metaphase
The initiation of the S phase and the M phase of the cell cycle depends on _______ and _______ to make _______.
A)actin; myosin; fibers.
B)Cdk; cyclin; MPF.
C)ligand; enzyme; complex.
D)insulin; receptor; G-protein.
E)ADP; Pi; ATP.
B)Cdk; cyclin; MPF.
The specialized nerve and muscle cells are found in ___________ stage of cell cycle?
A) G0
B)S
C)G1
D)G2
E)M
A)G0