Quiz Review: Molecular Compounds - Chemistry
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
When looking at chemical formulas, how can you tell if a substance is an ionic compounds or a molecular compound?
Molecular - Nonmetals, Binary
Ionic - Metal + nonmetal, polyatomic ion
When looking at a chemical names, how can you tell if a substance is an ionic compound or a molecular compound?
Ionic - Roman Numerals
Molecular - prefixes
SO2
Molecular
CF4
Molecular
NiO
Ionic
CuI2
ionic
PCI5
Moelcular
ZnF2
Ionic
Sulfur trioxide
SO3
Selenium tetrachloride
SeCl4
Nickel (II) Bromide
Ni2Br
Xenon hexaflouride
XeF5
Barium Sulfate
BaSO4
Hydrophosphoric Acid
H3PO4
Sulfurous Acid
H2SO4
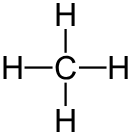
Single Bond
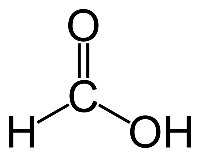
Double bond
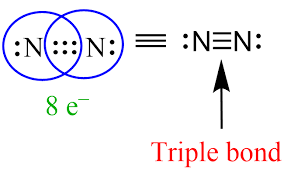
Triple Bond
True/ False
In molecular compounds, electron are shared between elements
True
True/ False
Molecular compounds have high melting points compared to ionic compounds
False
True/ False
Electrons are given away or taken when ionic bonds are formed
True
True/ False
Ionic bonds form between two metals
False
True/ False
Covalent bonds form between a metal and nonmental
False
True/ False
Ionic compounds make crystalline structure that are solids at room temperature
False
True/ False
The particle created by covalent bonding is called a molecule
True
True/ False
Molecular compounds contain covalent bonds
True
True/ False
Molecular compounds can be found in any state at room temperature
False
True/ False
Ionic compounds are named using numerical prefixes
False
Why do atoms make bonds?
Atoms make bonds to have a complete octet of 8 electrons, to make the compounds more stable, and electrons are shared because both atoms have similar attraction for electrons
How is bond angle of a molecule affected by lone pairs on the central atom?
Lone pairs repel electrons. Bond angle is reduced by lone pairs.
What subatomic particles make up bonds? Are bonds attracted to lone electron pairs? Why or why not?
Electrons. Bonds and lone pair repeal. Like charges repel. Opposite attract.
What is an octet?
A set of 8 valence electrons in the outer most energy level of an atom
How does the octet rule apply to molecular compounds and covalent bonds?
In molecular (covalent) compounds atoms share electrons so each atom can reach an octet (8 valence electrosn)
Explain how to write the names of H3N, HNO3, HNO2
H3N - Binary
Hydronitric acid
HNO3 - nitrate ion
nitric acid (ate - ic)
HNO2 - nitrite ion
Nitrous acid (ite - ous)
Linear
180 bond angle
Nonpolar Molecular
2 domain
2 Bond
o lone pairs
Trigonal Planar
120 bond angle
Nonpolar Molecular
3 domain
3 bonds
o lone pairs
Tetrahedral
109.5 bond angle
Nonpolar molecule
4 domain
4 bonds
o lone pairs
Trigonal pyramidal
107 bond angle
Polar molecule
4 domain
3 bonds
1 lone pairs
Bent
104.5
Polar Molecular
4 domains
2 bonds
2 lone pairs