19/20) Geochemical Kinetics 1&2
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
difference between kinetics and thermodynamics
thermo: study of energy transfer, tells what should happen, not when or how fast it will happen
excludes time
kinetics: introduces time, describes how fast reactions occur
water rock reactions are much __ than speciations reactions
slower
when are kinetics significant?
not very important when rate constants are fast: speciation and complexation reactions, dissolution of salts, high temperature
tend to be important
dissolution of aluminosilicate minerals (very), dissolution of other minerals, gas exchange reactions, redox
what do we want to achieve with kinetic theory
predict how fast a mineral will dissolve or precipitate as a function of geochemical parameters
pH, T, saturation state (go towards eqlm things go towards 0)
need a way of measuring rates in the lab so we can calculate them in the field
types of surface area
BET surface area (brunauer, emmett, teller): total surface area (crushed rock), quantified using physical adsorption of gas molecules to the mineral surface
geometric surface area: surface area of a simple geometry (sphere, cube) that approximates the general shape of the mineral grain
difference between transport limited and reaction limited
transport limited: solution equilibrates with the mineral quickly, so reaction rate is determined by new solution coming in
reaction limited: solution is slow to equilibrate with the mineral, the reaction rate is determined by the innate reaction rate (kinetics)
in limestones and sandstones, which is transport limited and which is reaction limited
limestones are transport limited
sandstones are reaction limited
what are different rate dependences?
pH, temperature
what are 2 kinetic experiments
batch: mineral + water in container sampled over time
flow-through: mineral + water, solution washing in and out, measuring change in concentration as fluid moves through the reactor
requires pumps, need a way of controlling flow rate and measure flow rate
control very precisely the solution that goes in
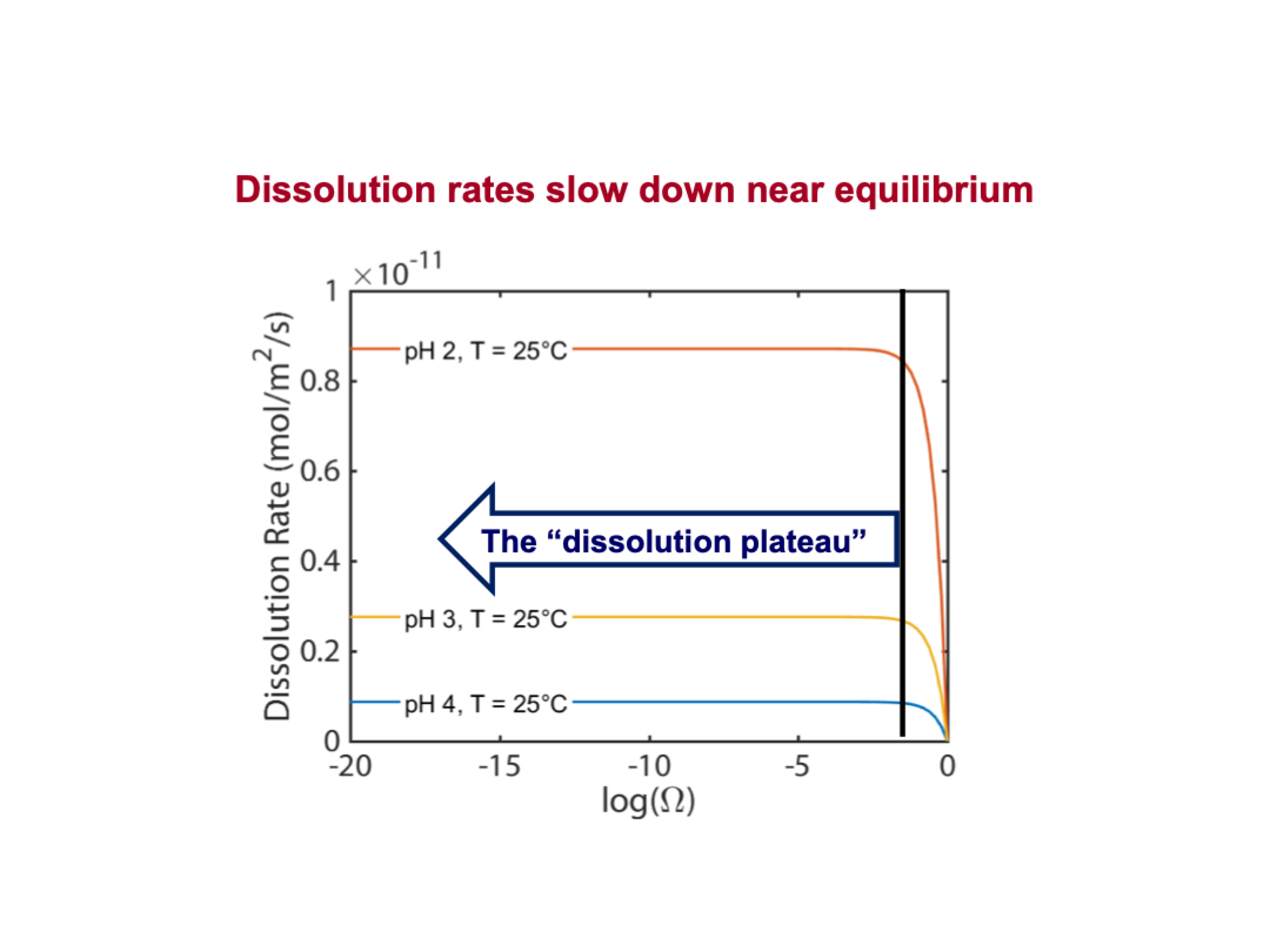
explain this
To start, there is basically no mineral in solution, so adding more mineral, the solution is still so undersaturated that the reaction rate is unchanging, it’s going as fast as it can at that particular pH, until it reaches a point where it's close to equilibrium but still undersaturated, and adding mineral begins to decrease the reaction rate as it moves closer to eqlm. Eventually eqlm must happen, so the reaction rate reaches zero
it’s easy to do experiments on dissolution plateau, because the rate is the same no matter what the saturated state is
K-feldspar dissolution rates ___ with increasing pH and the approach to equilbrium
slow down
why might mineral lifetimes be underestimates
reactive surface are is not equal to effective surface area
pH is generally more neutral
temperature is lower
if omega is less than one then what?
-rtln(omega) is negative
∆G is positive
dissolution
if omega is greater than one then what?
then -rtln(omega) is positive
∆G is negative
precipitation
what is transition state theory
explains chemical reaction rates by proposing that reactants form high-energy, unstable "activated complexes" at the peak of the energy barrier before becoming products, assuming a quasi-equilibrium between reactants and this state
mineral precipitation rates depend on both growth mechanism and saturation state
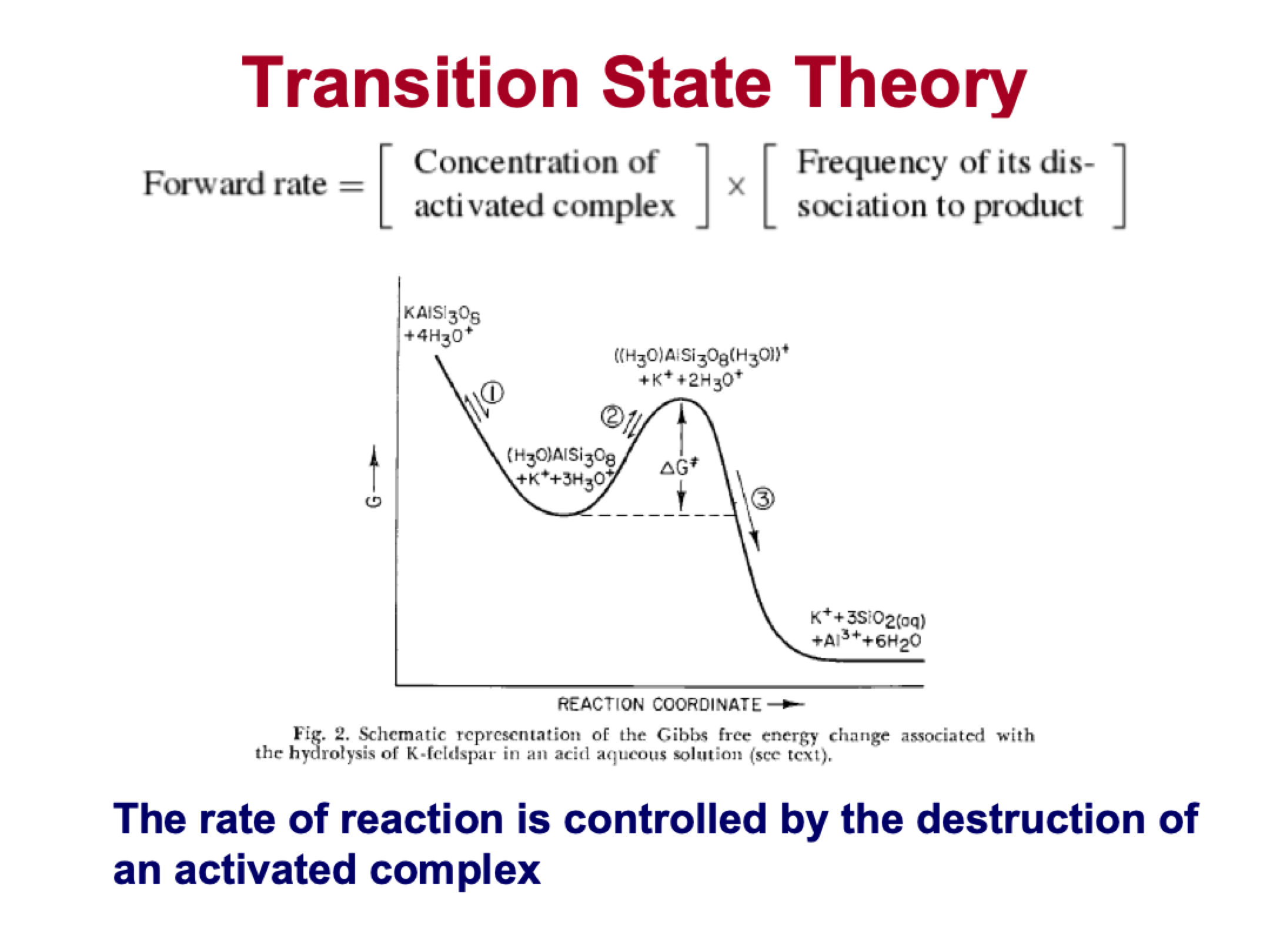
explain this
1) is reversible, the H+ are booting out a potassium, the charge balance is maintained. this happens all the time at mineral surfaces
2) is reversible, adding another H+ and now there is a charge on the surface, this is the activated complex
3) is irreversible, destruction of the kspar structure, the constituents are in solution
the overall rate of kspar destruction is controlled by reaction 3, the other reactions are instantaneous
what is the problem with affinity factors p and q
assuming p and q are 1 is inaccurate in many cases
details on batch expt
pros: cheap, easy, effective
fewer places to leak from, no pumps
easily repeatable and to vary parameters
cons: difficult to control fluid chemistry, change temperature
cannot prevent secondary minerals from precipitating
no control over solution chemistry once reaction has begun, once pH starts changing, it’s very quick
when would you need parr type reactors
elevated temperatures with gases
gas escape is low, maintaining constant pressure during sampling requires a constant pressure pump
maintaining constant pressure during CO2 injection requires fluid removal
details on gold
details on nucleation on glass
rough laser etching on the bottom of glass makes different nucleation patterns
nucleating CO2 bubbles out of supersaturated solution
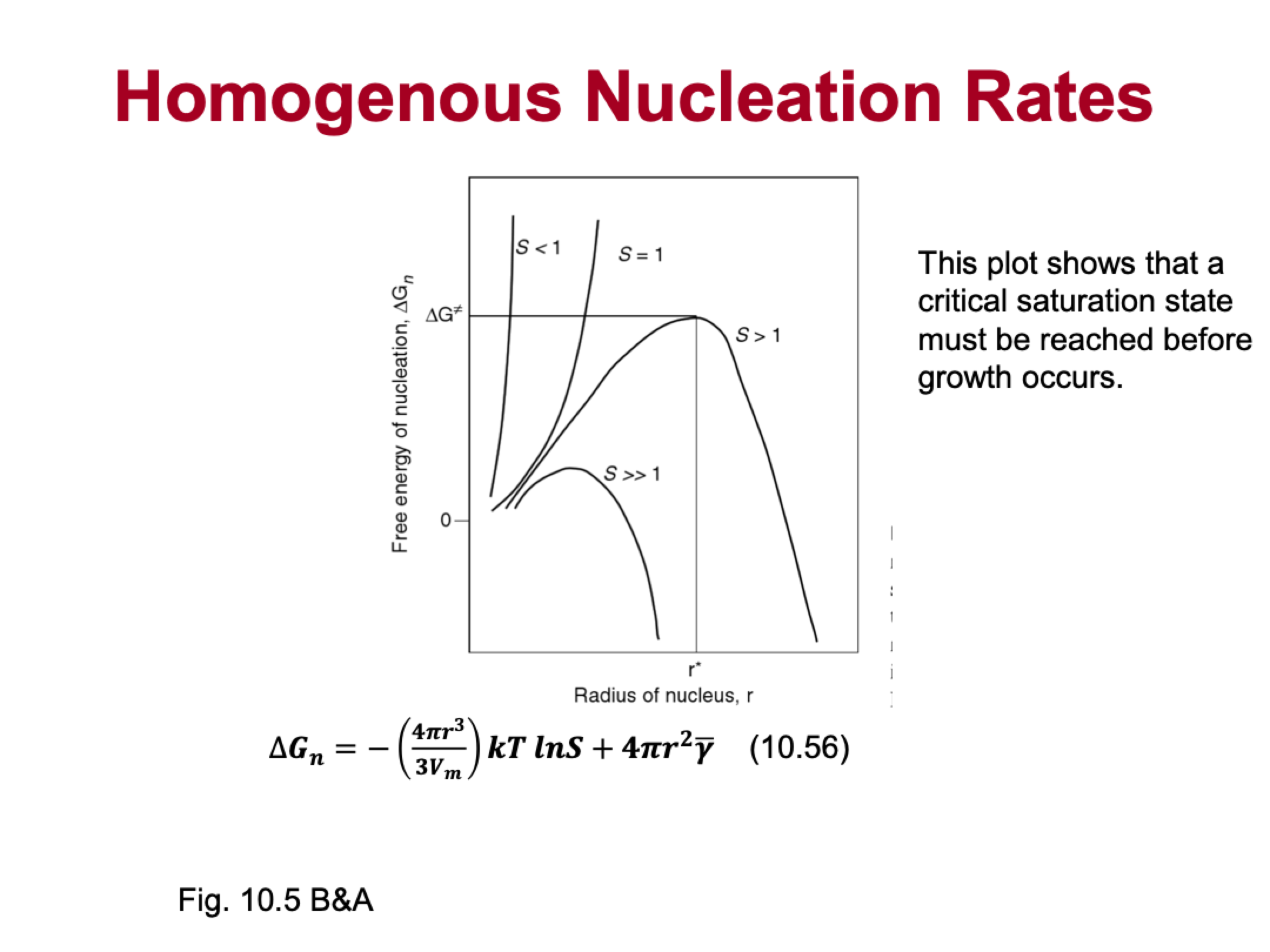
explain this
critical saturation state must be reached before growth occurs
high threshold of nucleation, make some bubbles then rate goes up drastically
very low threshold of nucleation, only require a little bit of supersaturation, then can make big nuclei right away
types of growth on mineral surfaces
mononuclear
polynuclear
screw dislocation: some part of mineral is raised above the rest, it grows in a spiral
crystal cube showing edge and kink
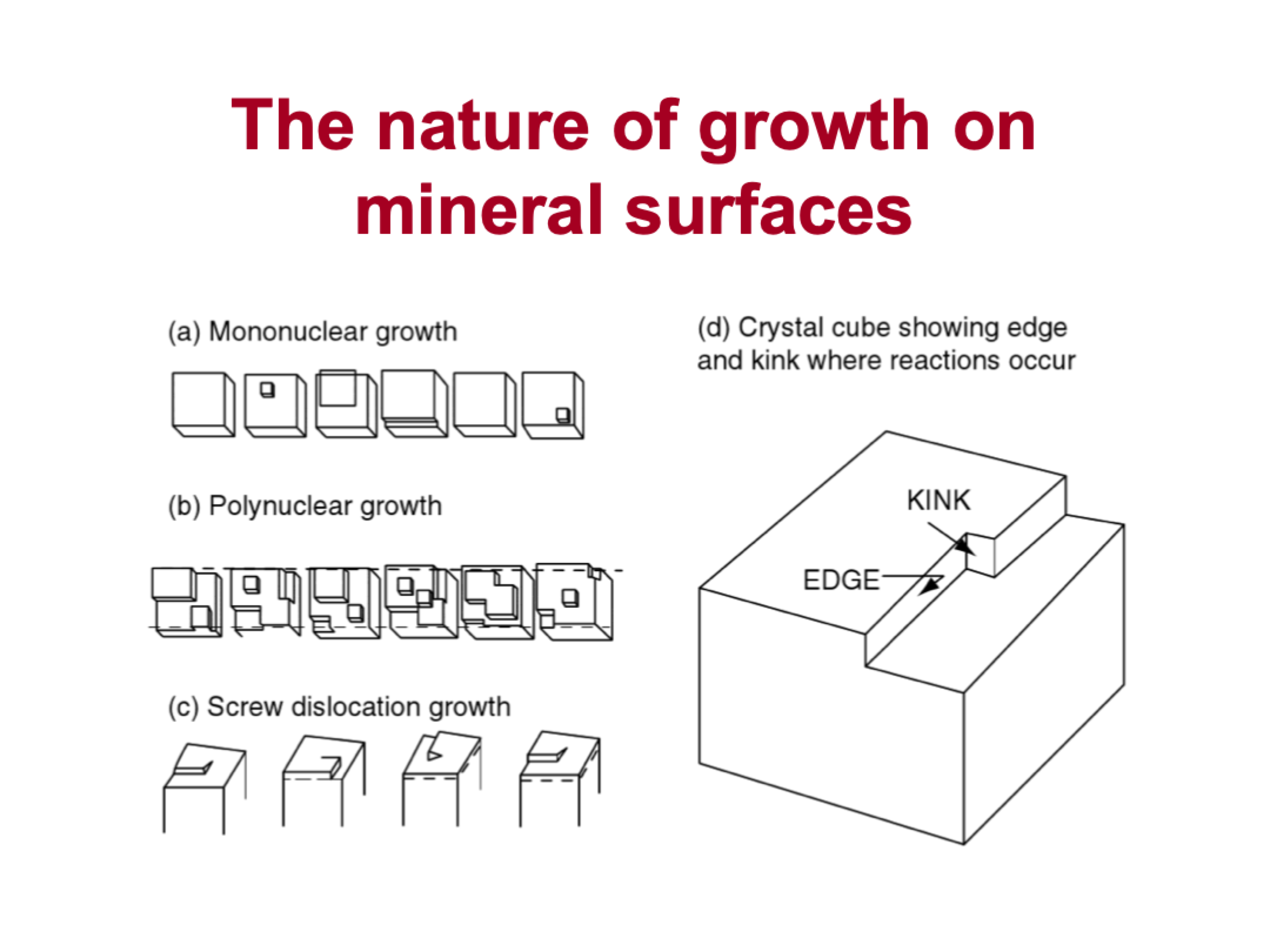
why is it difficult to predict precipitation on calcite
impossible to know how many screw dislocation points there are on calcite
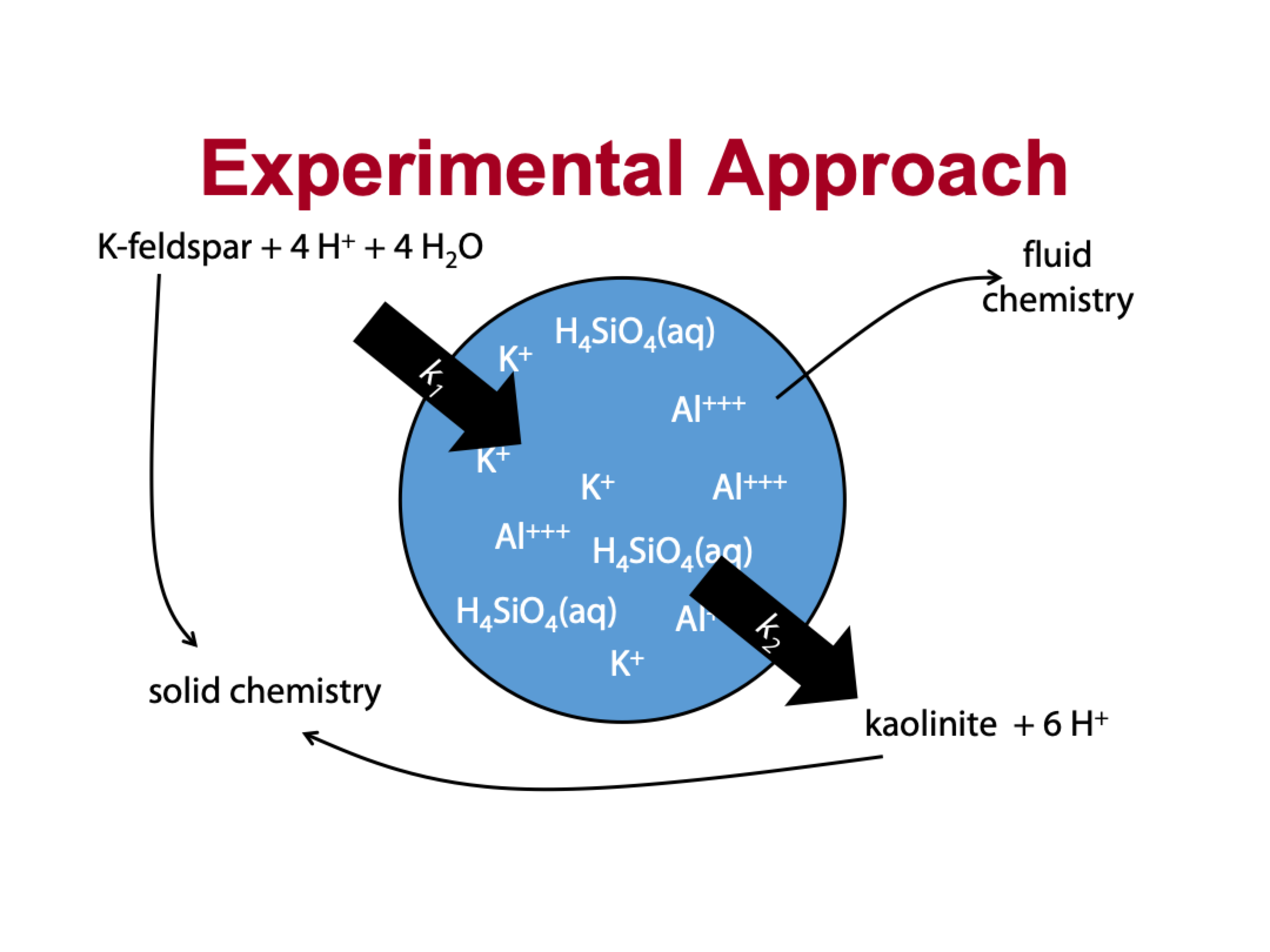
explain this
if K2 is really fast, then everything leaves solution and we are really far from eqlm