Reactions of Organic Chemistry II
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
H3O+ (hydronium ion) and a nucleophile like CN or OH
What are some typical reagents for acid catalyzed hydration
H2O, an electrophile like CN
What are some reagents for base catalyzed hydration
LiAlH4, NaBH4, Ni, Pd/C & H2
What are some reduction reagents for ketones/aldenhydes?

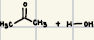
These are the reactants of the addition of HCN, what is the product?
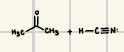
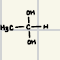
These are the reactants of the addition of H2O, what is the product?
Aldenhydes
What do primary alcohols oxidize to?
Ketones
What do secondary alcohols oxidize to?
Cr2O7, CrO3, PCC/CH2Cl2, MnO2
What are some reagents for oxidation overall?
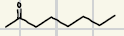
What is the product for a secondary alcohol with these reagents?
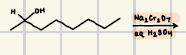
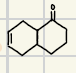
What is the product for a secondary alcohol with these reagents?
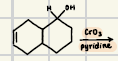
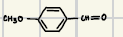
What is the product for a secondary alcohol with these reagents?
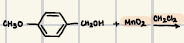
AlCl3,H2O
What are the reagents for Friedel-Crafts Acylation?
Hg2+, H2SO4
What are the reagents for the hydration of alkynes?
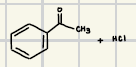
What is the product for this Friedel’s Craft Acylation
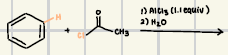
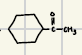
What is the product for the Hydration of Alkynes?
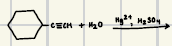
THF, H2O2/OH
What are the reagents for the hydroboration-oxidation of alkynes
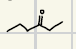
What is the product for the Hydroboration-oxidation of Alkynes?
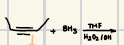
LiAlH4, NaBH4
What are some reduction reagents that should be known in aldehydes and ketones?

What is the product for this reduction reaction (ketone)

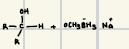
What is the product for this reduction reaction (ketone)
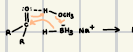
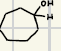
What is the product for this catalytic Hydrogenation
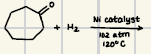
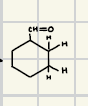
What is the product for this catalytic Hydrogenation
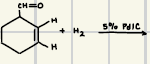
Pd/C, Ni Catalyst
What are some reagents for catalytic hydrogenation?
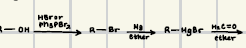
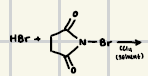
HBr or Ph3PBr2 along with Mg/ether
What reagents can help a Grignard reagent form?
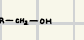
It will turn it into an alcohol
How will an MgBr react with a ketone?
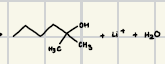
They also form alcohols
How do orgolithium reagents react with ketones?
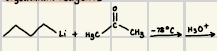
How do acetylide reagents react with ketones?
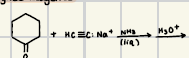
With the reaction of an aldenhyde or a ketone & an alcohol with a prescence of a strong acid
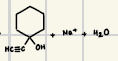
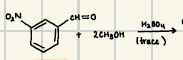
How are acetals formed?
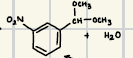
What does this acetal formation reaction form?
They are strong protecting groups
Why are acetals good in synthesis?
AcO, heat
What are the reagents for imine and enamines formation
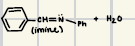
What is the reaction product for this imine formation
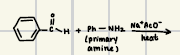
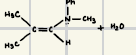
What is the reaction product for this enamine formation
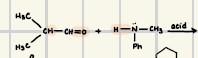
Acid
What are the reagents for enamine formation?
Wolff-Kischner Reaction/Clemmensen Reaction
These reactions can be used to reduce aldenhydes and ketones to a methylene
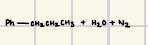
What is the reaction product for this wolff-kischner reaction?

H2N-NH2, KOH, and heat
What are the reagents for Wolff-Kischner Reaction?
Zn/Hg, HCl
What are the reagents for Clemmensen Reaction?
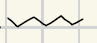
What is the reaction product for this clemmensen reaction?
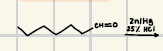
Wittig Alkene Synthesis
This is a good reaction to prepare alkenes from aldenhydes and ketones
Ph3PCH3
Reagents for Wittig Alkene Synthesis?
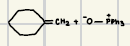
Reaction product for Wittig Alkene Synthesis?

KMnO4/NaOH, H2Cr2O7, and Ag2O
What are some good reagents for the oxidation of aldehydes?
Yes espeically if ortho para groups are added
Are allylic and benzylic radicals are good at reacting?
NBS Reactions
This is a reaction where there is a slower release of bromination as Br2 concentration remains low
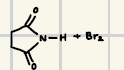
What are the products for this NBS reaction
H2CrO4,H2O or AgO and KMnO4
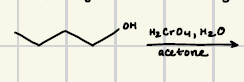
What are the reagents for the oxidation of alcohols and aldenhydes?
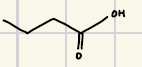
What is the reaction product for this oxidation of primary alcohol and aldenhyde?
CH3OH or CH2N2 & H2SO4
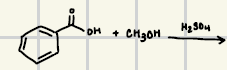
What are the reagents for an acid/fischer esterfication
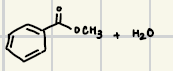
What is the product of this Fischer Reaction?
SOCl2 & PCl5
What are the reagents for Acid Chloride formation?
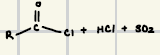
What is the reaction product of the synthesis of acid chlorides?
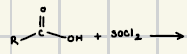
Through the treatment of sodium salts with PCl5
How are sulfonyl chlorides prepared?
LiAlH4
How can carboxylic acids be reduced to a primary alcohol?
CO2
What does the carboxylic acid lose during decarboxylation?
SN1 or SN2
What are the reactions that vinylic or phenols can’t really react to?
They would have to rehybridize and also steric hinderance being an issue with the bond angle changing from 120 to 90
Why can’t vinyls or phenols really react in SN reactions?
L type ligands
These are the type of ligands that can be removed with its bonding electron pair and obtain a neutral molecule
X type ligands
Those that we can remove with its bonding pair having formal charge
X type ligand
This ligand can be removed with its bonding pair that has a molecule with formal charge
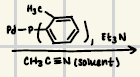
Pd(OAc)2
Reagents for the Heck reaction
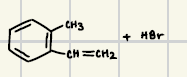
Reaction product for heck reaction
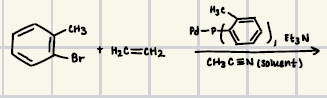
Pd(OAc)2, PPh3, Na2CO3
What are the reagents for suzuki coupling
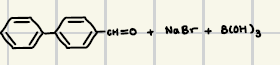
What is the product for this suzuki coupling?
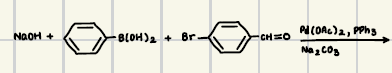
Adding an ortho para directing group to the phenol
What makes phenol extremely reactive?
Acid chlorides and anhydrides
These will react rapidly with water even in the absence of bases
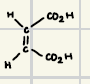
Reaction product for hydrolysis of acid chloride and anhydrides
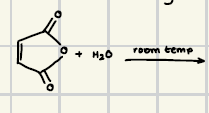
Hydrolysis of esters/saponofication
This reaction is irreversible unlike the acid version of this reaction.
Through an acid catalyst and when the OHs meet up
How can lactones be formed?
Through strong acids and heat
How can nitriles and amides be hydrolyzed to carboxylic acids?