Chemistry Unit 1, topic 1, QCE 2025
1/54
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Determine the name and symbol of the element with this electron configuration:
a) 2
b)2,7
c) 2,8,3
d) 2,5
e) 2,8,7
a) He, Helium
b) F, Fluorine
c) Al, Aluminium
d) N, Nitrogen
e) Cl, Chlorine
Describe the electron configuration of the following elements:
a) Ca
b) Sr
c) Na
d) Cl
a) 2,8,8,2 or 1s²,2s²,2p^6, 3s², 3p^6,4s²
b) 2,8,8,18,2 or 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s²
c) 2,8,1 or 1s² 2s² 2p⁶ 3s¹
d) 2,8,7 or 1s² 2s² 2p⁶ 3s² 3p⁵
Oxygen has 2 known isotopes. One has 9 neutrons and the other has 10 neutrons. Write their symbols using using atomic symbol notation.
Isotope with 9 neutrons:
Protons: 8
Neutrons: 9
Mass number (A): 8 + 9 = 17
Symbol: ¹⁷O
Isotope with 10 neutrons:
Protons: 8
Neutrons: 10
Mass number (A): 8 + 10 = 18
Symbol: ¹⁸O
Explain the similarities and differences between isotopes of an element.
Same: Number of protons (same element), same chemical properties.
Different: Number of neutrons (different mass), potentially different nuclear stability (radioactivity) and different physical properties.
State the relative energies of the s, p and d orbitals
In atoms with many electrons, the energy of an electron's path is determined by how far it is from the center, the shape of its path, and how it pushes away from other electrons. Because s-orbitals tend to spend more time very close to the center, they have the lowest energy. Then, p-orbitals, which have a different shape, have a higher energy than s-orbitals. Finally, d-orbitals, which have even more complex shapes and are further away, have the highest energy among these three. This energy difference is caused by the different shapes of the paths, how much time electrons spend near the center, and how electrons block each other from feeling the full pull of the center.
State the number of electrons in each shell for the following atoms.
a) 5 electrons
b) 12 electrons
c) 20 electrons
d) 35 electrons
a) 2,3
b) 2,8,2
c) 8,8,2
d) 2,8,18,7
Draw the Bohr diagram for the an atom of Argon.
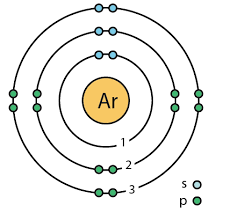
Identify two elements that have two electrons in their valence shell. Describe another similarity between these elements.
Both beryllium and magnesium are located in Group 2 of the periodic table, also known as the alkaline earth metals. Another similarity between these elements is that they both tend to form ions with a +2 charge when they participate in chemical reactions, as they readily lose their two valence electrons to achieve a stable, full outer shell configuration.
An atom has two electrons in the first shell, six electrons in the second shell and eight electrons in the third shell. Identify the atom and state why this electron arrangement is unexpected. Propose a reason for this.
The atom described, with two electrons in the first shell, six in the second, and eight in the third, is sulfur. This electron arrangement is unexpected because the typical electron configuration for sulfur is 2, 8, 6. The described configuration suggests an additional two electrons have been added to the third shell, pushing it to a full octet. This is likely due to the atom existing as a sulfide ion (S²⁻), having gained two electrons to achieve a stable, full outer shell, similar to the noble gas argon.
Identify the correct name for an atom with seven protons and seven neutrons. Write its nuclear symbol notation.
An atom possessing seven protons and seven neutrons is identified as Nitrogen, and its nuclear symbol notation is written as ¹⁴N
Different elements sometimes have the same number of neutrons but never the same number of protons. Identify any examples of this in the first 10 elements and use these examples in your answer.
It is possible for different elements to have the same number of neutrons, as shown by Boron-12 and Carbon-13, which both contain seven neutrons, but it is impossible for them to have the same number of protons, because the number of protons defines the element.
Determine full and condensed electron configurations for atoms and ions up to Z = 36
To determine full electron configurations, we systematically fill electron shells and subshells according to the Aufbau principle, starting with the lowest energy levels. 1 For example, potassium (K, Z=19) has a full configuration of 1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹. In contrast, condensed electron configurations utilize the preceding noble gas symbol in brackets to represent the filled inner shells, simplifying the notation. 2 Potassium's condensed configuration becomes [Ar]4s¹. When forming ions, electrons are added or removed from the outermost shell. 3 For instance, the chloride ion (Cl⁻, Z=17) gains an electron, resulting in a full configuration of 1s² 2s² 2p⁶ 3s² 3p⁶, or a condensed configuration of [Ar]. Similarly, the calcium ion (Ca²⁺, Z=20) loses two electrons, yielding a full configuration of 1s² 2s² 2p⁶ 3s² 3p⁶, or a condensed configuration of [Ar]. This method effectively portrays the electron distribution in atoms and ions up to atomic number 36.
Identify the electron configuration of Cr and Cu as exceptions.
Chromium and copper deviate from the predicted electron configurations due to the enhanced stability associated with half-filled and fully-filled d subshells. In the case of chromium, instead of the expected 4s²3d⁴, an electron is promoted from the 4s orbital to the 3d orbital, resulting in a 4s¹3d⁵ configuration, where the 3d subshell is half-filled. Likewise, copper, rather than having a 4s²3d⁹ configuration, adopts a 4s¹3d¹⁰ configuration, achieving a fully-filled 3d subshell. This electron promotion from the 4s to the 3d orbital demonstrates that the stability gained from these specific d subshell arrangements overrides the standard filling order.
Explain how successive ionisation energy data is related to the electron configuration of an atom
Successive ionization energy data reveals the electron configuration of an atom by showing how much energy is needed to remove each electron, one after another. As electrons are removed, the remaining electrons are held more tightly, so each successive ionization energy is higher. Large jumps in ionization energy occur when an electron is removed from a new, inner shell, indicating the number of electrons in each shell and therefore the electron configuration.
State that isotopes can be represented in the form AX (IUPAC) or X-A
Isotopes can be represented in the form ᴬX, according to IUPAC notation, where A represents the mass number and X represents the element's symbol. Alternatively, they can also be represented as X-A, where X is the element symbol and A is the mass number
Identify that isotopes of an element have the same electron configuration and possess similar chemical properties but have different physical properties
Isotopes of a particular element maintain an identical electron configuration, which directly results in their exhibiting remarkably similar chemical properties. However, due to their differing numbers of neutrons, isotopes possess variations in their physical properties, most notably in their mass and density
Explain that the relative atomic mass of an element is the ratio of the weighted average mass per atom of the naturally occurring form of the element to 1/12 the mass of an atom of carbon-12
The relative atomic mass of an element is a comparison of its average atomic mass to a standard. Specifically, it's the ratio of the weighted average mass of all the naturally occurring isotopes of that element to one-twelfth of the mass of a carbon-12 atom. This means that instead of giving the absolute mass of an atom, which is incredibly small, it provides a relative value that allows us to compare the masses of different elements. The "weighted average" part is crucial because it accounts for the different abundances of isotopes in a naturally occurring sample. Carbon-12 is used as the standard because it's a stable and well-defined isotope.
Define isotopes
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
Recall whether the atomic number for atoms of the same element ever changes.
The atomic number for atoms of the same element never changes. If the atomic number changes, the element itself changes.
Explain why the relative atomic mass of an element shown in the periodic table is generally not a whole number.
Identify the number of protons, electrons and neutrons in a neutral atoms of ^37Cl
Identify that the structure of the periodic table based on increasing atomic number.
Describe the key features of the periodic table
Determine the name and symbol of the following elements. In addition, identify the shell configuration of each element.
a) second element in group 14
b) second element in group 2
a)
b)
Identify that the periodic table is arranged into four blocks associated with the four sub-levels — s, p, d and f.
Define the term ‘effective nuclear charge’ of the an atom and determine the effective nuclear charge of an atom of carbon.
Define the first ionization energy of an atom
Explain the term ‘shielding effect’
Determine the electron configuration of the following atoms or ions.
a) Ca
b) Al ³+
a)
b)
Explain why ionization energy increases from left to right across a period.
Compare and contrast the trends in atomic and ionic radii in the periodic table using specific examples to illustrate your explanation.
Sort the following in order of increasing atomic radius: based on your understanding of the trends in the periodic table.
N, B, Ga. Al, Cl
Predict whether Mg2+ is larger than F- using the periodic table. Explain your choice based on the structure of the two ions.
Deduce why the number of subatomic particles in an atomic increases across a period but the size of the atom decreases.
Describe the relationship between the structure of the periodic table and the electronic configuration of atoms.
Define atomic radius.
Refers to the size of an atom. Fluorine is smaller than lithium even though is has more protons and electrons.
Define first ionization energy
It’s how much energy it takes to remove an electron from an atom.
Define effective nuclear charge.
List any differences between the lithium and fluorine atoms - visualized using Bohr’s model
The lithium atom has three protons and three electrons, with its electrons, with its electrons, with its electrons arranged as 2 in the first shell and 1 in the second shell. Fluorine has nine protons and nine electrons, with 2 electrons in the first shell and 7 in the second shell. That’s why fluorine is way more eager to grab an extra electron to fill its outer shell, while lithium is eager to loose one.
Explain how these differences account for the different atomic radii of lithium and fluorine.
As you go across the periodic table, atoms gain more protons, which means a stronger positive charge in the nucleus. This stronger positive charge pulls the electrons in closer, making the atomic radius smaller. Even though fluorine has more electrons, the pull from the increased number of protons makes it smaller than lithium.
Explain how these differences account for the different first ionization energies of lithium and fluorine.
Lithium has that one electron in its outer shell, and it really wants to get rid of it to have a full inner shell. This means it doesn’t take much energy to pull that electron away. However, fluorine already has 7 electrons in its outer shell and really wants one more to complete it. So, it’s holding onto electrons super tightly, meaning it takes a lot more energy to remove one.
What is the smallest unit of element that still carries the properties of that element?
Atom
The Bohr model of the atom is primarily known for its description of…
the quantisation of electron energy levels.
Consider the following isotope of argon: A#18, AM40 Ar
The number of protons is 18 and the number of neutrons is 22
Which of the following statements is true about isotopes of an element?
Isotopes have different numbers of neutrons but the same number of protons
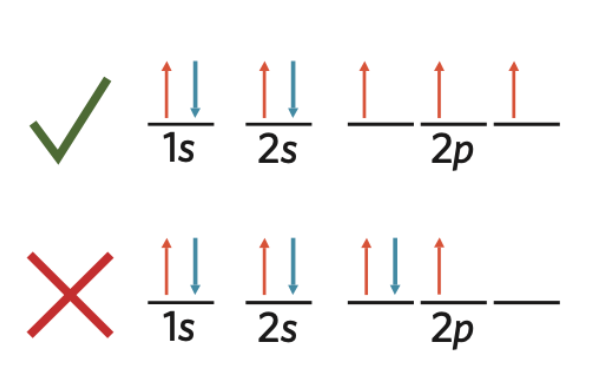
True or False, is this the best representation of Hunds Rule?
True
Why do chromium (Cr) and copper (Cu) have exceptions to the usual electron configuration filling order?
They have more stable electron configurations with half-filled or fully-filled d orbitals.
What is the full electron configuration for sodium (Na)?
1s2 2s2 2p6 3s1
Identify the condensed electron configurations for the element with atomic numbers 28 and 29
28: [Ar] 4s2 3d81
29: [Ar] 4s1 3d102
Why does the 4s subshell fill with electrons before the 3d subshell? Explain this phenomenon in terms of the relative energies of the 3d and 4s subshells.

The 4s subshell has a lower energy than 3d. According to the Aufbau principle the subshells are filled in order of their energy increasing. This means that the 4s orbital is more stable and fills with electrons before the 3d orbital.
Describe the essential difference between the shell model and the subshell model of the atom in terms of energy levels.
In short, the shell model has electrons in energy levels based on distance from the nucleus, while the subshell model refines this, showing that within each shell, energy levels are further split by orbital shape (s, p, d, etc.).
Identify an electron configuration of a sulfur atom that has been heated so that its electrons are no longer in the ground state.
An excited state electron configuration for sulfur could be: 1s² 2s² 2p⁶ 3s¹ 3p⁵ 3d¹ because excited electrons move up one level.
Define ground state.
The ground state is the lowest energy configuration of an atom's electrons (normal electron configuration)
Define excited state
The excited state occurs when an electron absorbs energy and moves to a higher energy level.