electrolysis part 2
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
reactivity series
Potassium
Sodium
Calcium
Magnesium
Aluminium
Carbon
Zinc
Iron
Tin
Hydrogen
Copper
Silver
Gold
more reactive metals are
strongest reducing agent , loses electrons readily + oxidised
more reactive metal reduces the ions of / can displace a less reactive metal
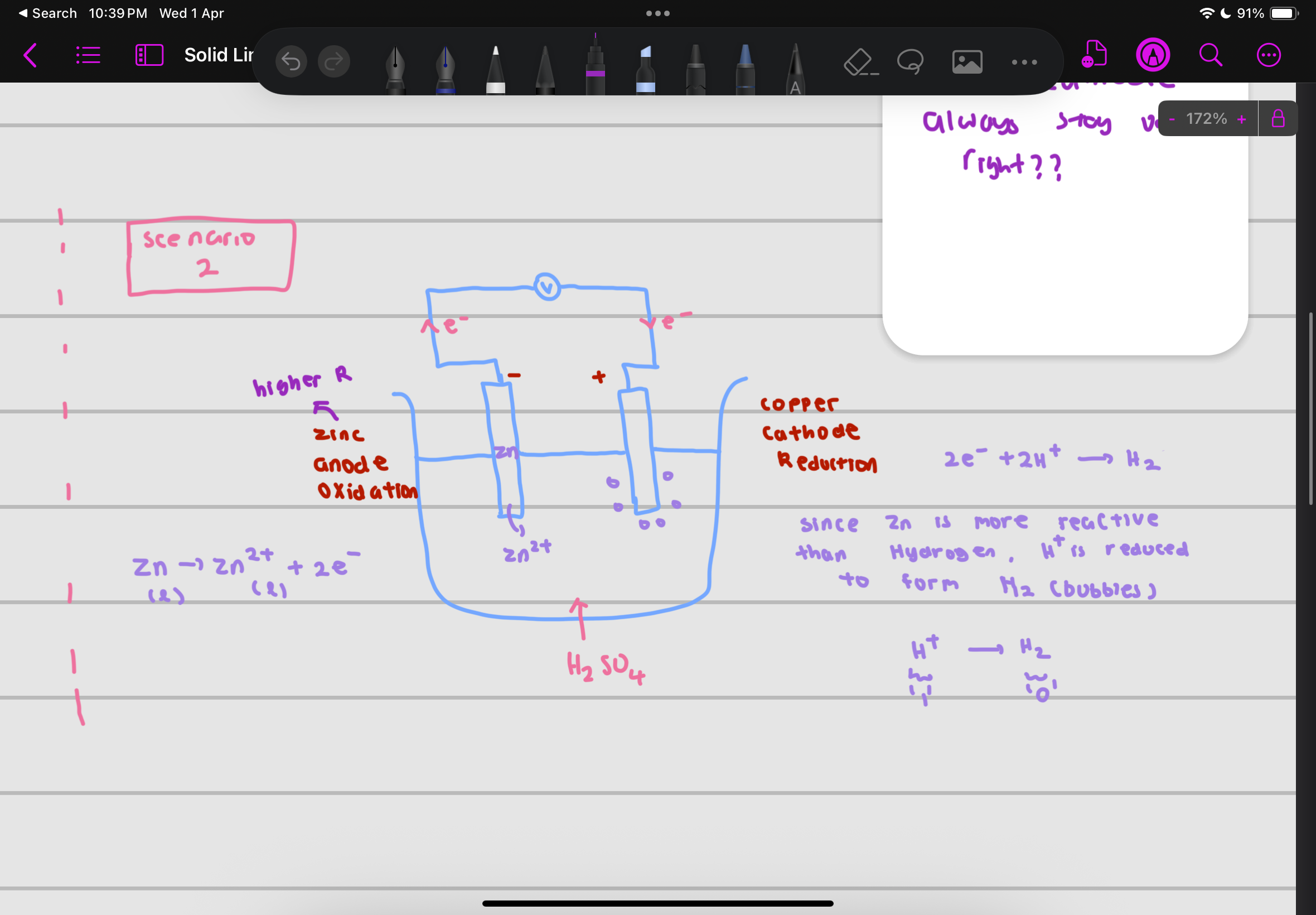
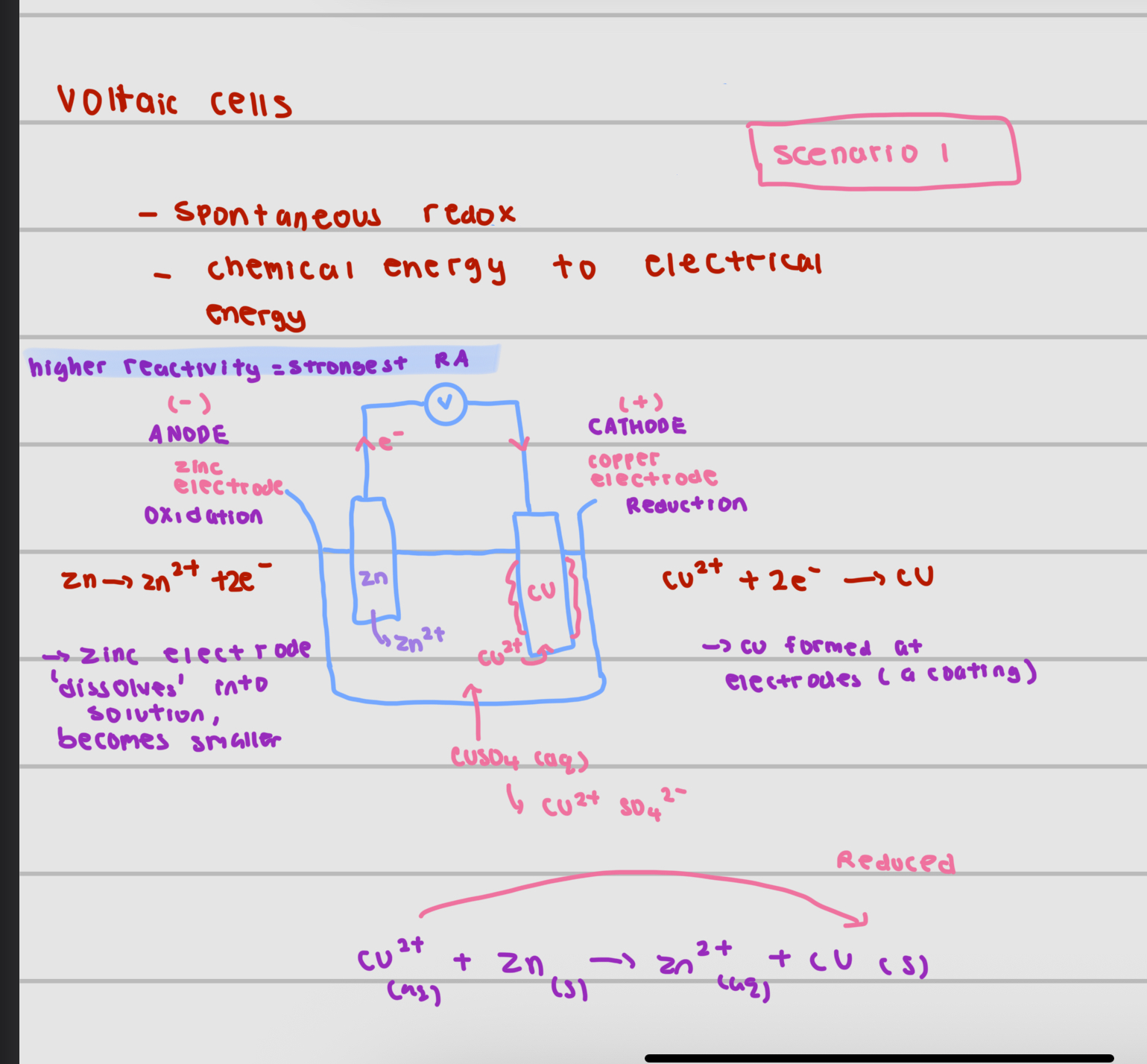
voltaic cell
serves like a battery, converts chemical energy to electrical energy
in a voltaic cell, the electrical energy is produced from a spontaneous redox reaction in which electrons are transferred through an external circuit
a simple voltaic cell consists of
2 electrodes made of metals with different reactivities , immersed in an aqueous electrolyte solution
solution should contain ions of less reactive metal
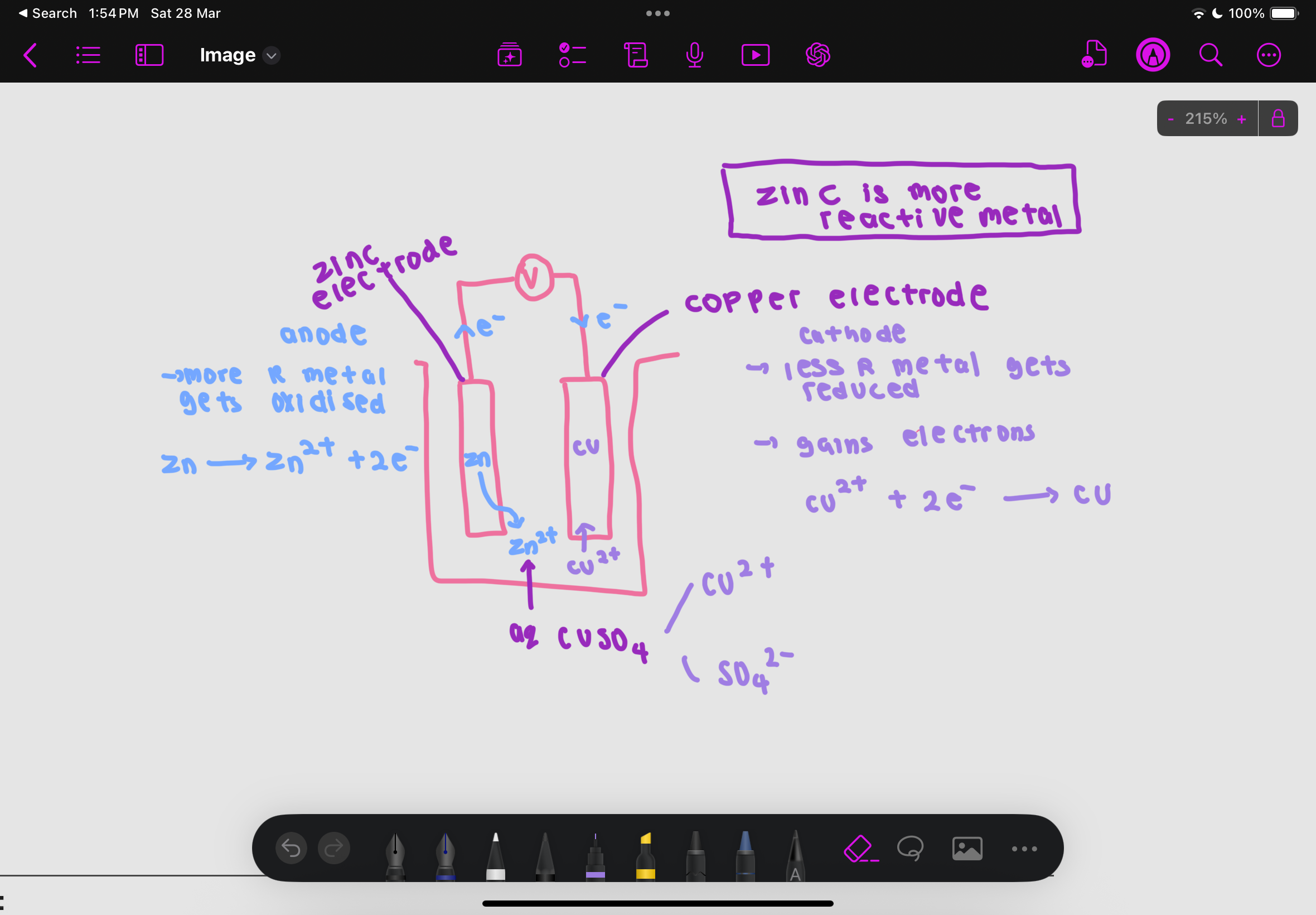
what is at the anode
the metal higher in reactivity series as it is oxidised and loses electrons to form positive ions
How does electrons flow in the voltmeter
electrons flow through the external circuit (wire) to the less reactive metal at the cathode (from more reactive to less reactive)
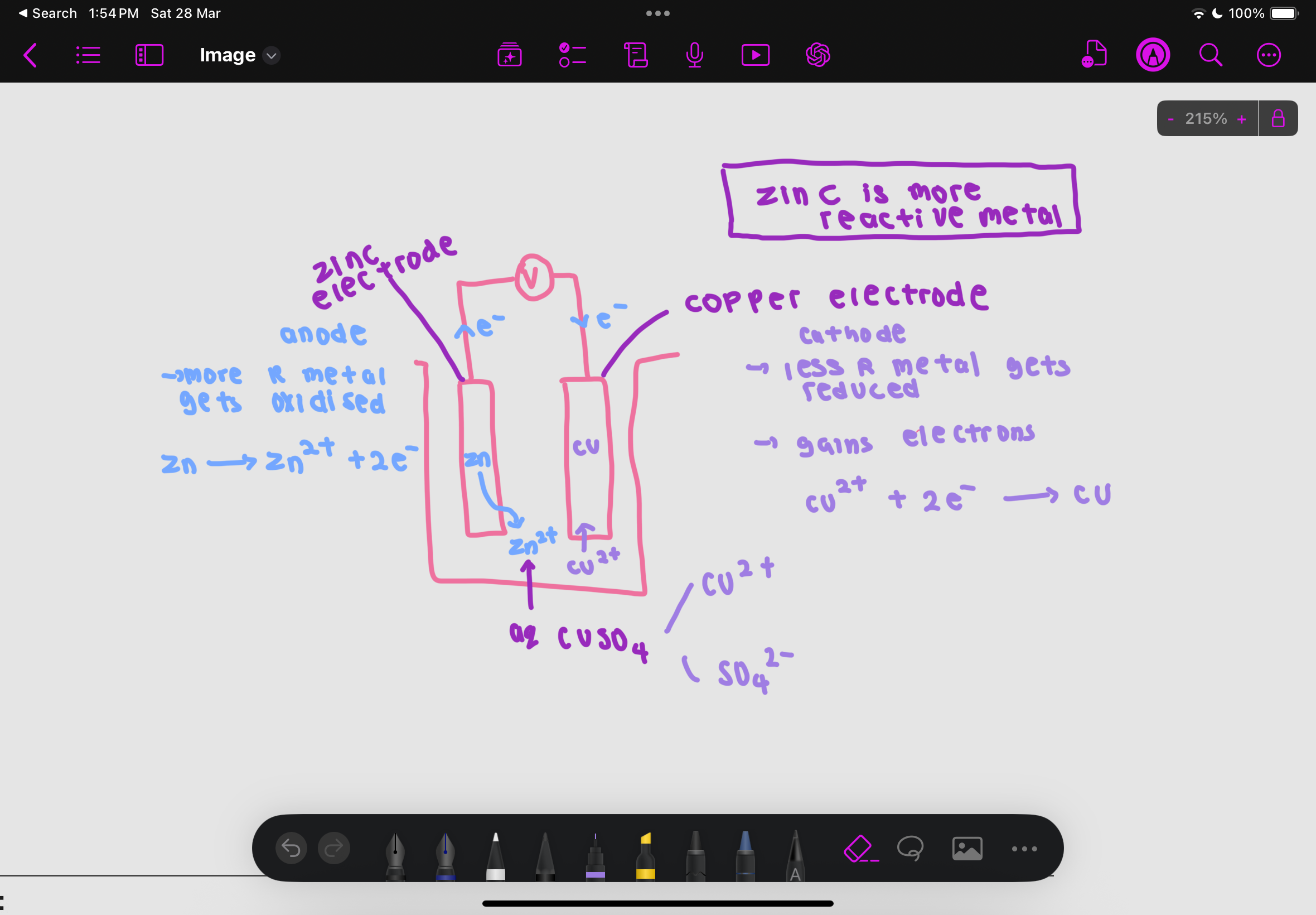
what is at the cathode
the lower in reactivity series metal acts as the cathode
At the cathode, positive ions in the electrolyte gain electrons and are reduced to form neutral atoms
the farther apart 2 metals are in the reactivity series
the greater the cell voltage
eg zinc-copper cell : cell voltage of 1.1V but magnesium and copper 2.7V because they are further apart in reactivity diagram so their voltage is larger
further apart=larger voltage
example of zinc electrode and copper electrode with aqueous CuSO4
example of zinc electrode and copper electrode with aqueous H2SO4