4.4 organic compounds containing oxygen
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
prefix of alcohols
hydroxy-
suffix of alcohols
-ol
suffix of aldehydes
-al
prefix of phenols
phen-
suffix of ketones
-one
suffix of carboxylic acids
-oic acid
suffix of esters
(alcohol)-yl (carboxylic acid)-oate
suffix of acid chlorides
-oyl chloride
suffix of acid anhydrides
-oic anhydride
suffix of nitriles
-nitrile
suffix of amines
-amine
suffix of amides
-amide
primary alcohol
1 alkyl group attached to C-OH
secondary alcohol
2 alkyl groups attached to C-OH
tertiary alcohol
3 alkyl groups attached to C-OH
carboxylic acid acidity
• weak acids
• negative charge of the carboxylate ion is delocalised, which stabilises the ion allowing H+ to form
alcohol acidity
• less acidic than carboxylic acids and phenols
• the charge on anion cannot be delocalised to another electronegative atom
alcohol solubility
as the carbon chain length increases the contribution of the polar -OH or the -COOH group decreases due to the larger non-polar section of the molecule
phenol acidity
• less acidic than carboxylic acids
• the negative charge of the phenoxide ion is stabilised by being delocalised around the ring
testing for carboxylic acids
• universal indicator; colour change from green to red
• sodium carbonate; effervescence passed through limewater turns milky
testing for alkenes
bromine water; colour change from orange brown to colourless
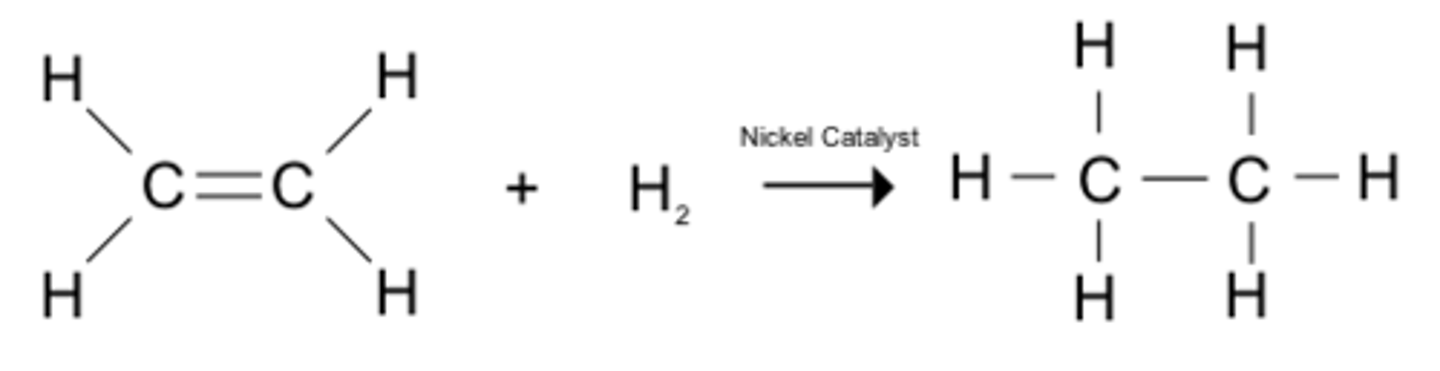
alkene → alkane
• electrophilic addition
• reagents; hydrogen gas (H₂), Ni catalyst
aldehyde → primary alcohol
• reduction
• reagents; sodium borohydride (NaBH₄)
• conditions; reflux
ketone → secondary alcohol
• reduction
• reagents; sodium borohydride (NaBH₄)
• conditions; reflux
carboxylic acid → aldehyde
• reduction
• reagents; lithium aluminium hydride (LiAlH₄), dry ether
• conditions; room temperature
carboxylic acid → primary alcohol
• reduction
• reagents; lithium aluminium hydride (LiAlH₄), dry ether
• conditions; room temperature
halogenoalkane → alcohol
• nucleophilic substitution
• reagents; NaOH (aq)
• conditions; reflux
halogenoalkane → alkene
• elimination
• reagents; NaOH in ethanol
• conditions; reflux
halogenoalkane → nitrile
• nucleophilic substitution
• reagents; KCN in ethanol
alcohol → bromoalkane
• bromination
• reagents; NaBr/H₂SO₄
• conditions; reflux
alcohol → chloroalkanes
• chlorination
• reagents; hydrogen chloride gas (HCl), anhydrous zinc chloride (ZnCl₂)
• conditions; heat
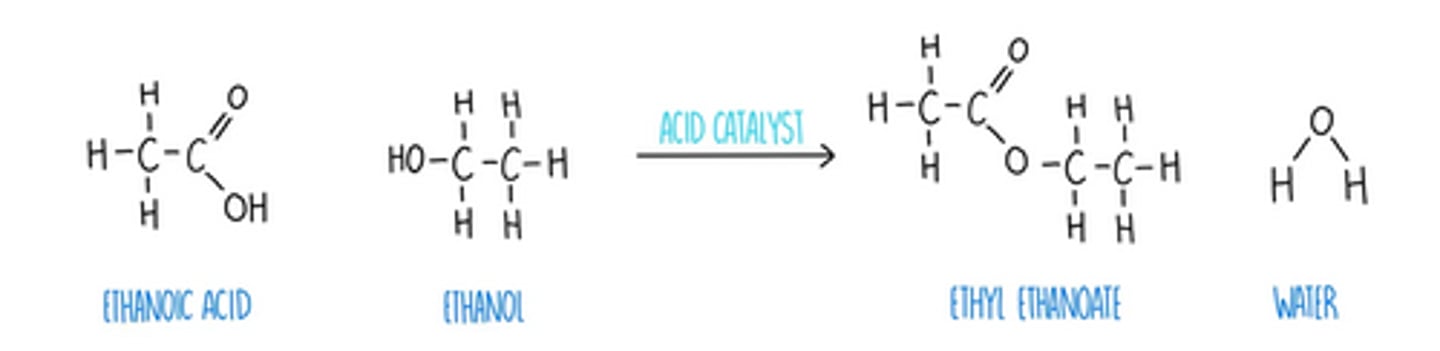
carboxylic acid + alcohol → ester
• esterification
• reagents; H⁺ catalyst
• conditions; reflux
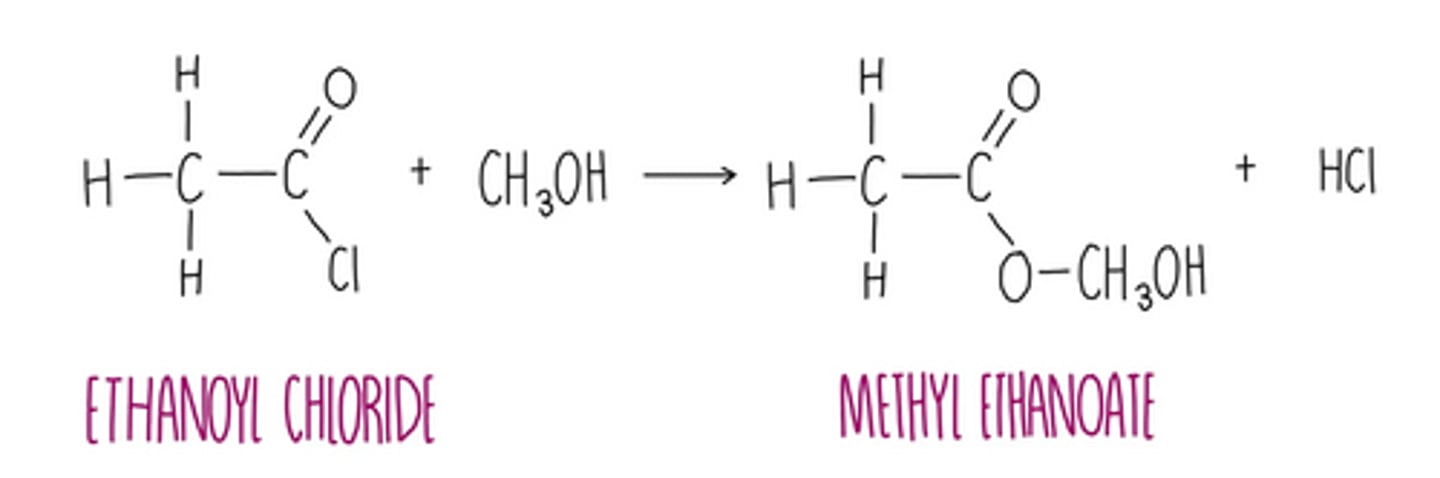
acyl chloride + alcohol → ester
• esterification
• conditions; room temperature, dropwise
primary alcohol → aldehyde
• oxidation
• reagents; H⁺/Cr₂O₇²⁻ or H⁺/MnO₄⁻
• conditions; gentle heating, immediate distillation
primary alcohol → carboxylic acid
• oxidation
• reagents; H⁺/Cr₂O₇²⁻ or H⁺/MnO₄⁻
• conditions; reflux
secondary alcohol → ketone
• oxidation
• reagents; H⁺/Cr₂O₇²⁻ or H⁺/MnO₄⁻
• conditions; heating
alcohol → alkene
• elimination
• reagents; concentrated H₂SO₄
• conditions; reflux
testing for primary and secondary alcohols
• acidified potassium dichromate (H⁺/Cr₂O₇²⁻); colour change from orange to green
• acidified potassium permanganate (H⁺/MnO₄⁻); colour change from pink to colourless
testing for secondary alcohols (iodoform)
warmed iodine in aqueous sodium hydroxide; pale yellow precipitate forms (CHI₃), antiseptic smell
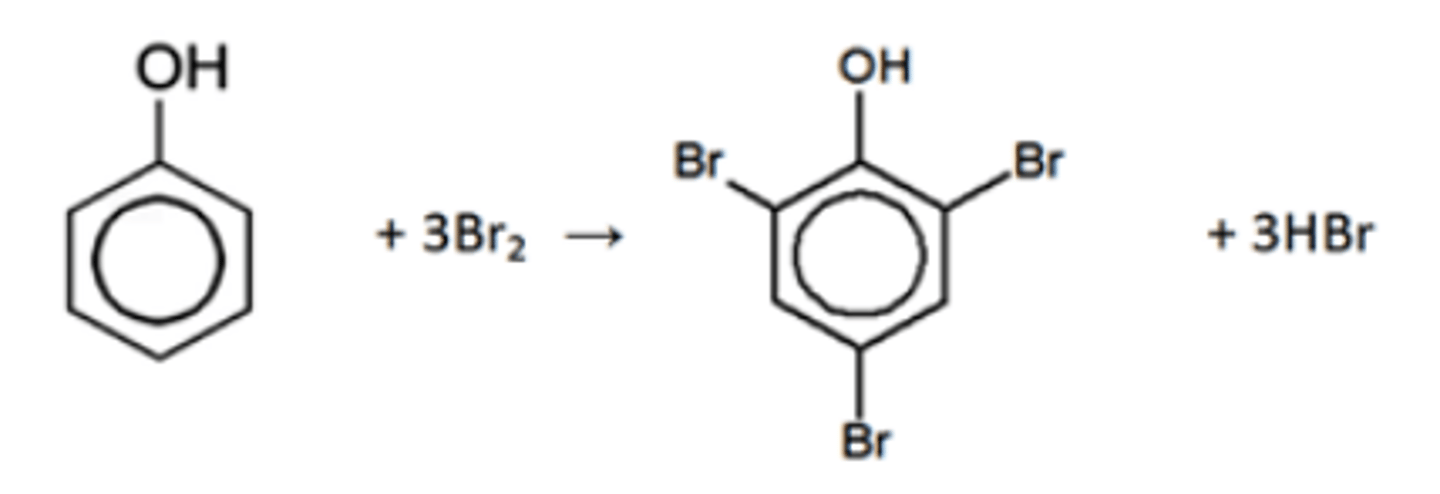
phenol → 2,4,6-tribromophenol (white solid)
• electrophilic substitution
• reagents; Br₂
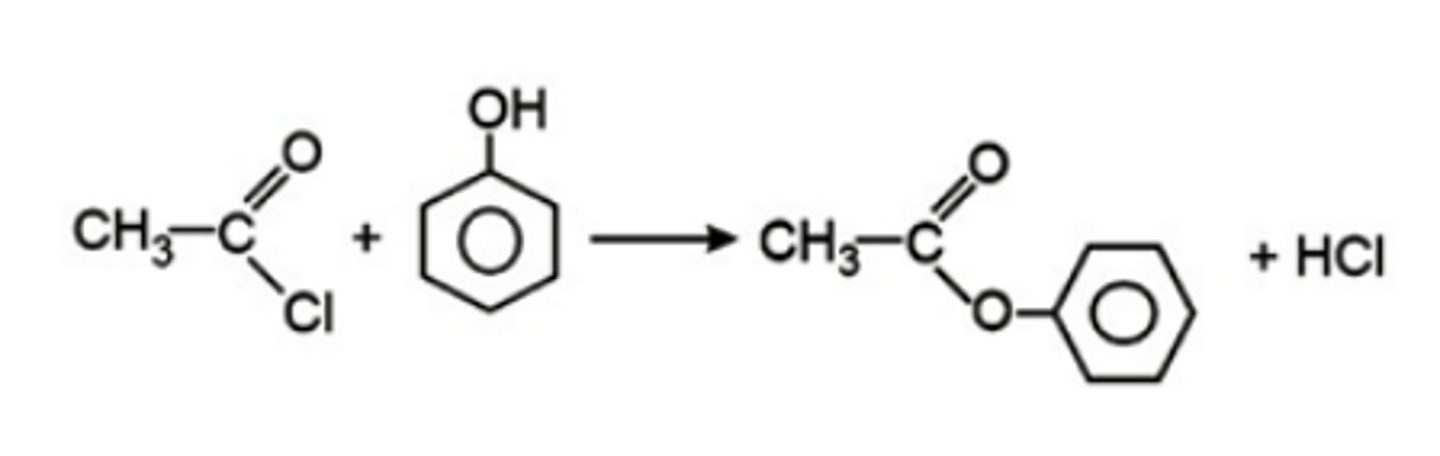
phenol → ester
• electrophilic substitution
• reagents; acyl chloride
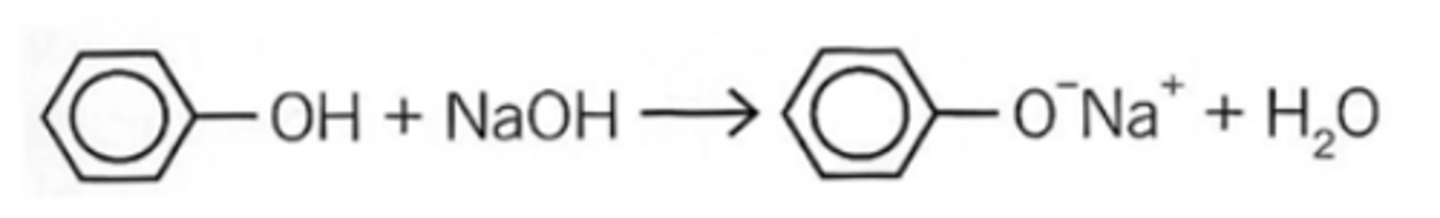
phenol → sodium phenoxide
reagents; sodium hydroxide
testing for phenols
neutral iron(III) chloride solution; purple/violet colour forms
aldehyde → carboxylic acid
• oxidation
• reagents; H⁺/Cr₂O₇²⁻
• conditions; heat
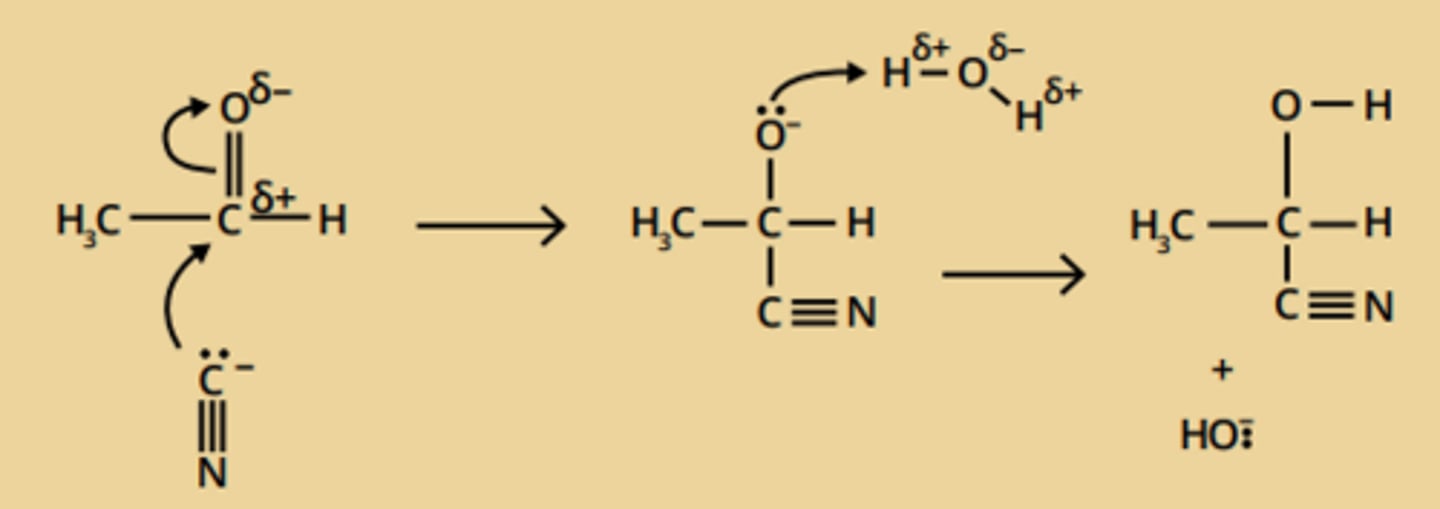
aldehyde/ ketone → nitrile
• nucleophilic addition
• reagents; potassium cyanide (KCN), dilute sulphuric acid (H₂SO₄)
testing for aldehydes
• 2,4-dinitrophenylhydrazine (2,4-DNPH); forms red crystals
• Tollens' reagent; forms silver mirror
• Fehling's reagent; colour change from blue solution to brick-red precipitate
• acidified dichromate (Cr₂O₇²⁻); colour change from orange to green
identifying an aldehyde with pure crystals
the crystals have very sharp melting points which can be compared with known values in a database
identifying an aldehyde with impure crystals
the melting point will be lower than the actual value and the crystals will melt over a temperature range
Tollen's reagent
• silver nitrate in aqueous ammonia
• Ag⁺(aq) + e⁻ → Ag(s)
Fehling's reagent
• Cu²⁺ ions dissolved in aqueous alkali
• 2Cu²⁺(aq) + 2e⁻ + 2OH⁻(aq) → Cu₂O(s) + H₂O(l)
testing for ketones (+ethanal)
warmed iodine in aqueous sodium hydroxide; pale yellow precipitate forms (CHI₃), antiseptic smell
ester → carboxylic acid + alcohol
• hydrolysis
• reagents; water, dilute hydrochloric acid
• conditions; reflux
ester → carboxyate + alcohol
• hydrolysis
• reagents; sodium hydroxide
• conditions; reflux
amides → carboxylic acid
• hydrolysis
• reagents; sodium hydroxide
• conditions; reflux, followed by acidification
acyl chloride → carboxylic acid
• hydrolysis
• reagents; water
• conditions; room temperature
nitrile → carboxylic acid
• hydrolysis
• reagents; water, dilute hydrochloric acid
• conditions; reflux, heat
benzene → carboxylic acid
• oxidation
• reagents; alkaline potassium manganate (VII) solution (MnO₄⁻)
• conditions; heat, followed by acidification
carboxylic acid → acyl chloride
• reagents; phosphorus pentachloride (PCl₅)
• conditions; anhydrous
carboxylic acid → alkane
• decarboxylation
• reagents; soda lime (NaOH)
• conditions; heat
carboxylic acid → amide
• oxidation
• reagents; ammonia (NH₃)
• conditions; heat/ reflux
amide → nitrile
• reagents; phosphorus(V) oxide (P₄O₁₀)
• conditions; heat