No lone pairs shapes and angles
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
how do pairs of electrons in the outer shell arrange themselves
as far apart as possible to minimise repulsion
what does planar mean
flat (all atoms in same plane)
which pairs of electrons have the greatest repulsion
1. lone pair - lone pair
2. lone pair - bond pair
3. bond pair - bond pair
How does a lone pair affect bond shape
It affects bond angle by 2.5º
what does a wedge tell us
coming out of the page
what does a dotted line tell us
behind plane of page
what does a solid line tell us
on plane of page
what is shape of a molecule determined by
the electron pairs surrounding the CENTRAL atom (as electron pairs repel other pairs)
how do we treat a lone pair
as another bond (e.g if there are 3 atoms and one lone pair then has the tetrahedral shape)
what if the lone pair has dative covalent
then the bond angle is that of a normal molecule, despite being a lone pair (no extra repulsion)
what is a covalent bond
a pair of electrons
2 bonding pairs 0 lone pairs
linear, 180°
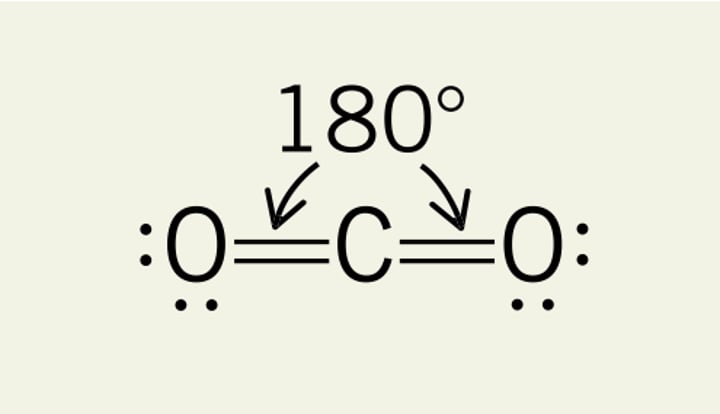
3 bonding pairs
trigonal planar, 120
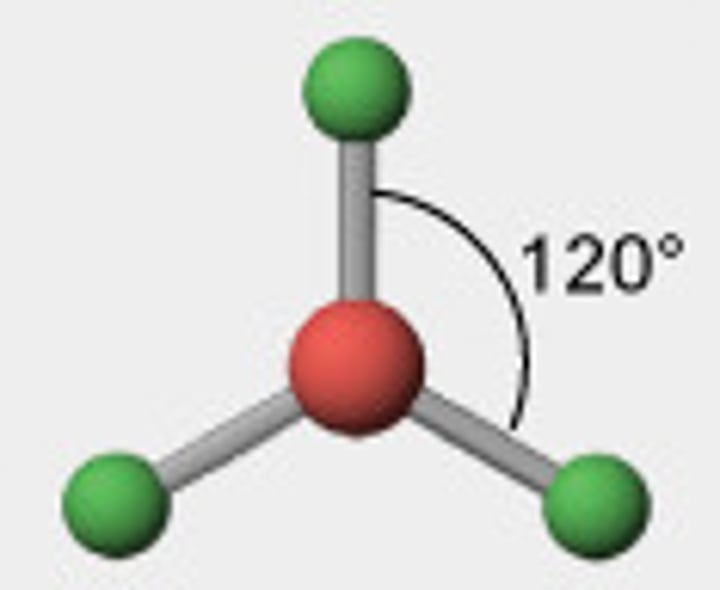
4 bonding pairs
Tetrahedral 109.5
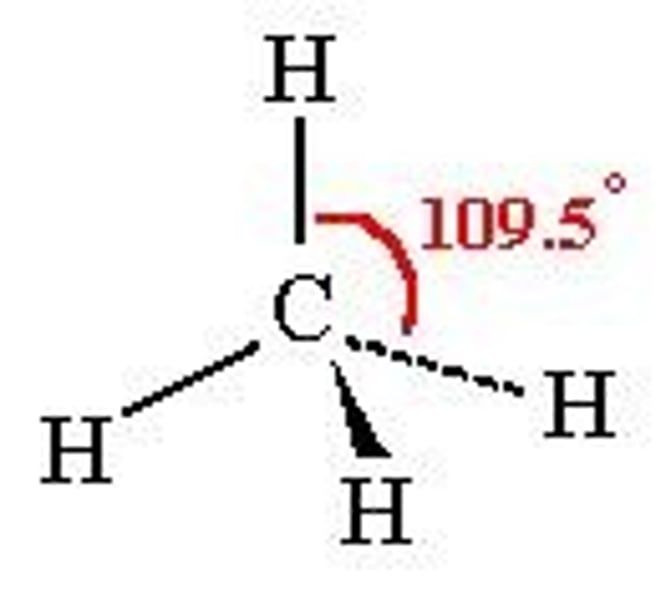
5 bonding pairs
trigonal bipyramidal, 90 120
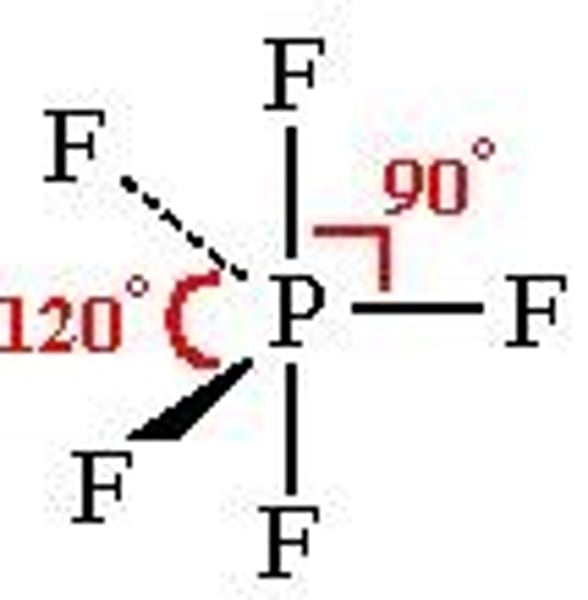
6 bonding pairs
Octahedral 90
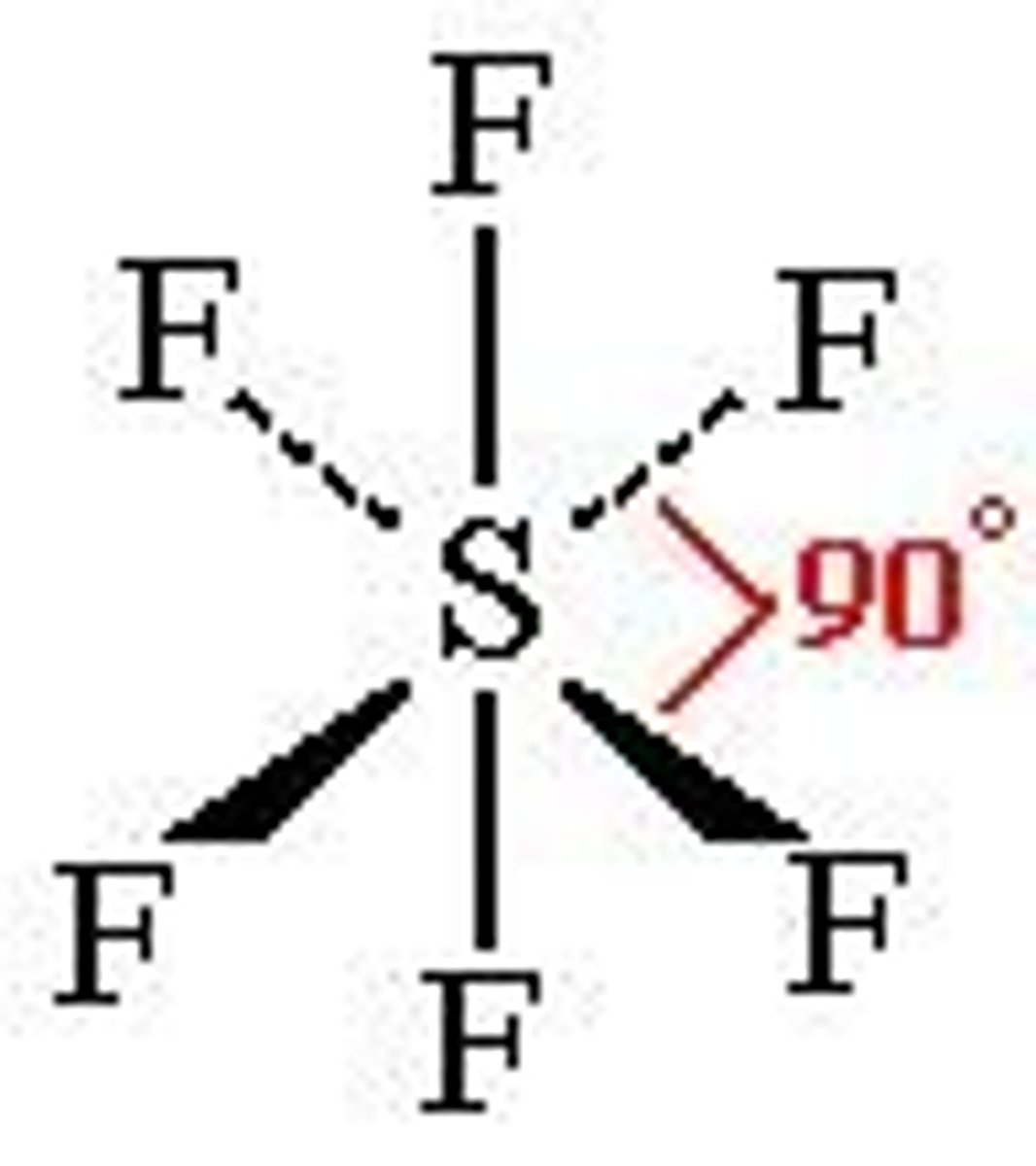
explain why x has bond angle x
there are x bonding pairs of electrons, therefore they repel equally and spread out evenly as far apart as possible.
when is it obvious there is a dative covalent bond
when something bonds to a H+ (pair of electrons donated to H+)
type of bond how it is formed question
this is always a dative/coordinate bond question
1. coordinate bond
2. the H+ gains 2e- from other atom/molecule