Respiratory Physiology and Control Mechanisms PHGY 355
1/329
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
330 Terms
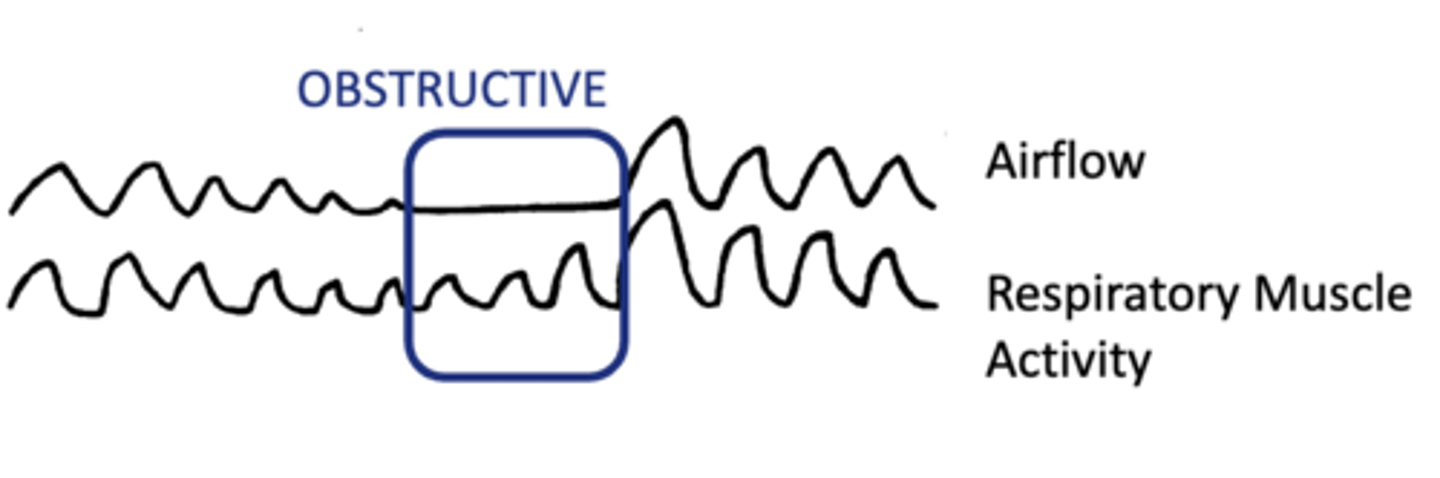
Obstructive Apnea
No airflow due to upper airway obstruction.
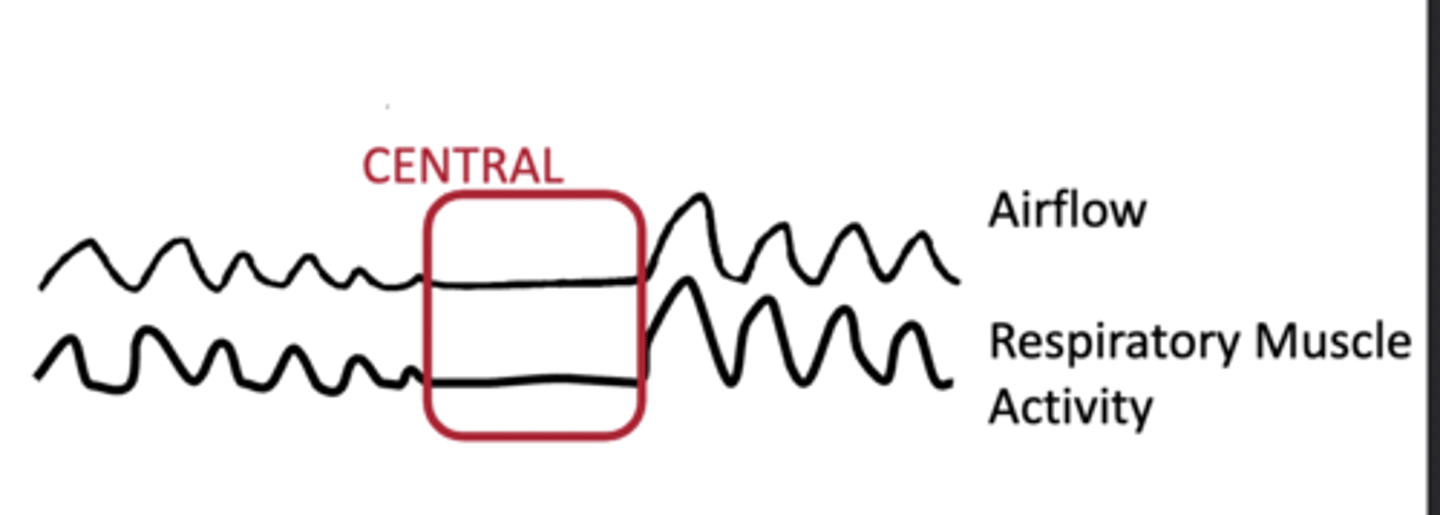
Central Apnea
No breathing activation; respiratory control failure.
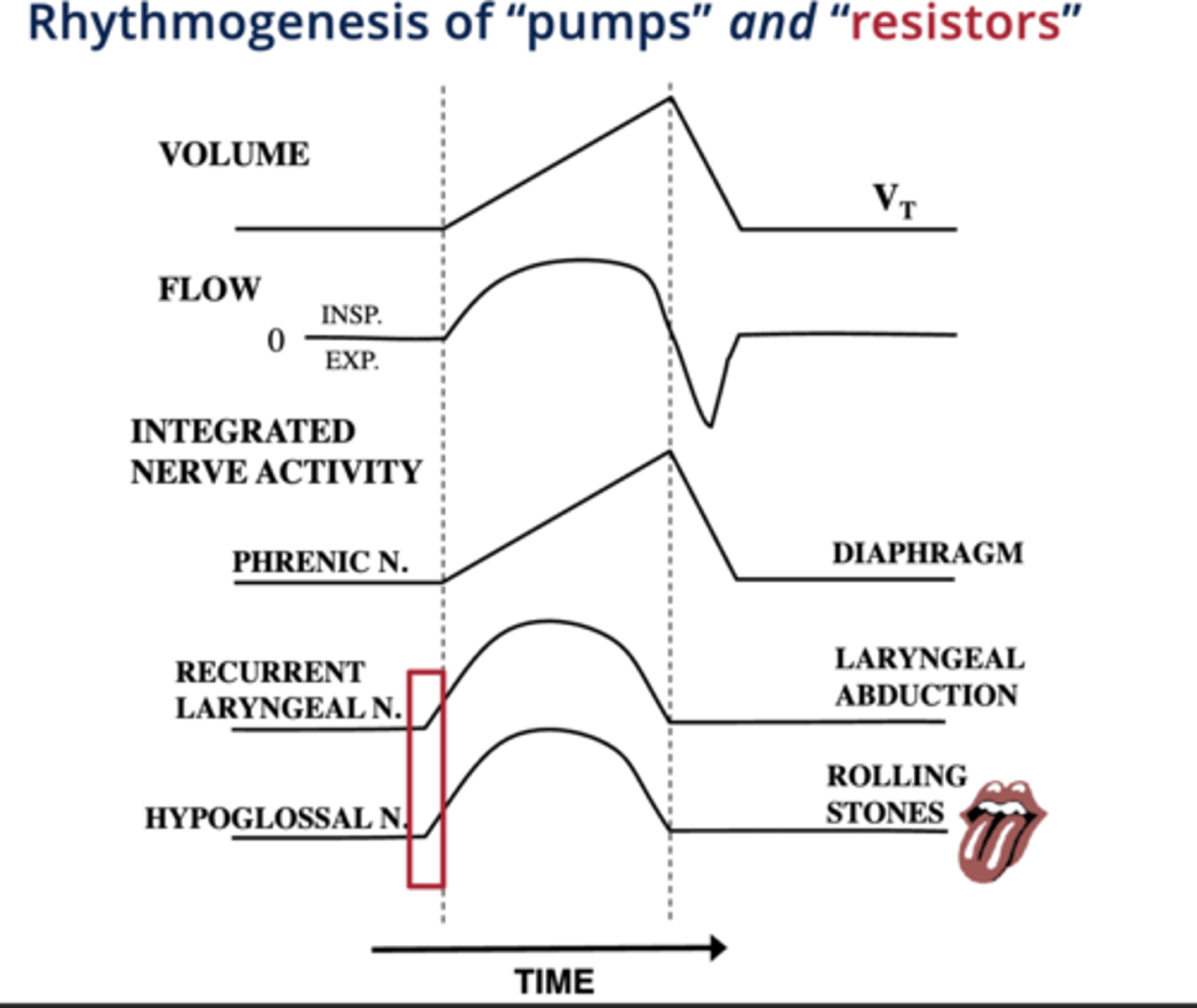
Rhythmogenesis
Generation of rhythmic breathing patterns via encoded neural signals
Bötzinger/Pre-Botzinger Complex
Site in brain for rhythmogenesis of breathing.
Neural Control of breathing
Medulla and pontine respiratory groups in the brainstem manage breathing patterns.
Chemoreception
Sensory feedback from chemical changes in blood (O₂, CO₂, H+)
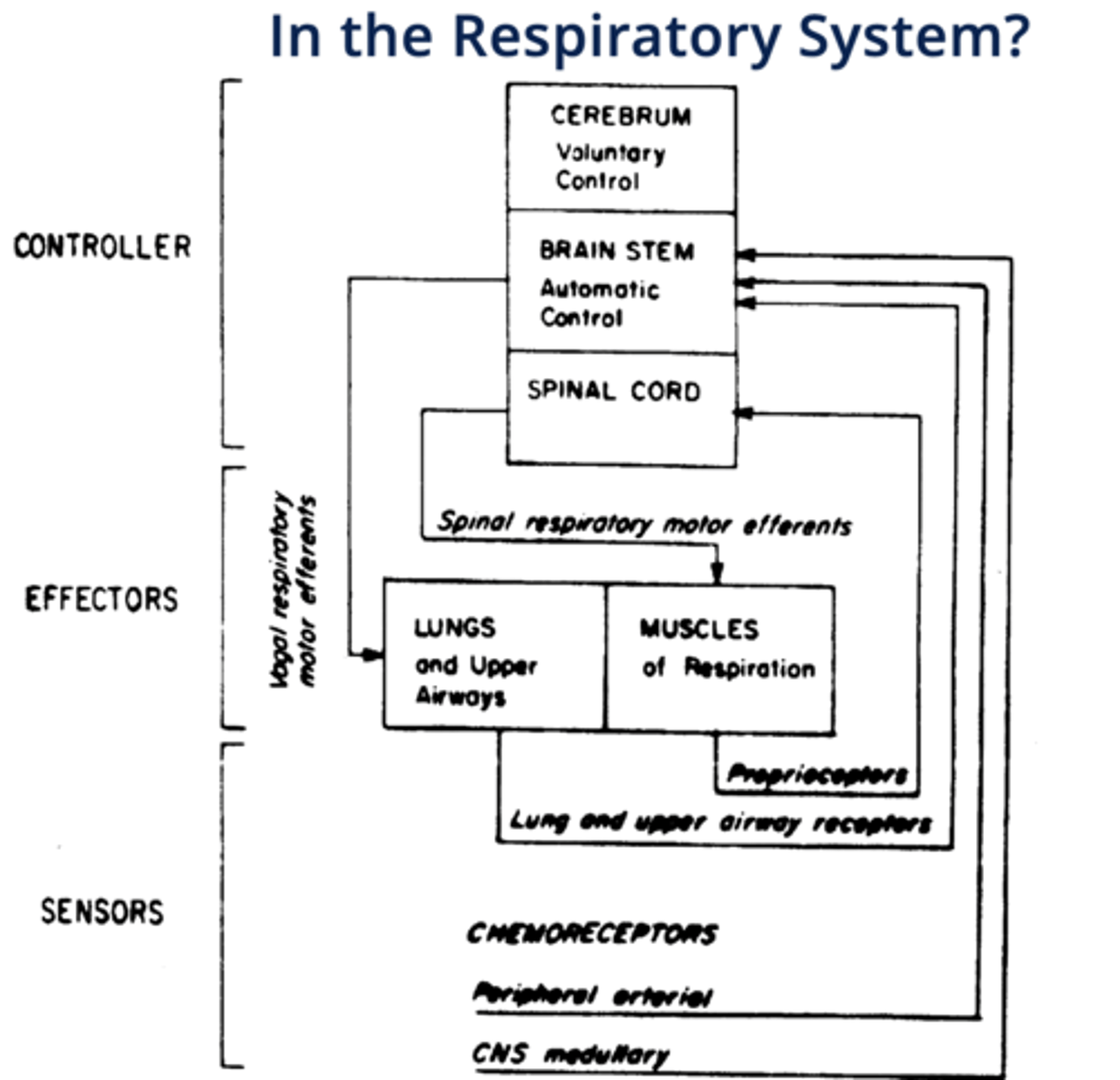
"Controllers" of respiration
cerebrum (voluntary control), brainstem (automatic control), spinal cord
(CBS)
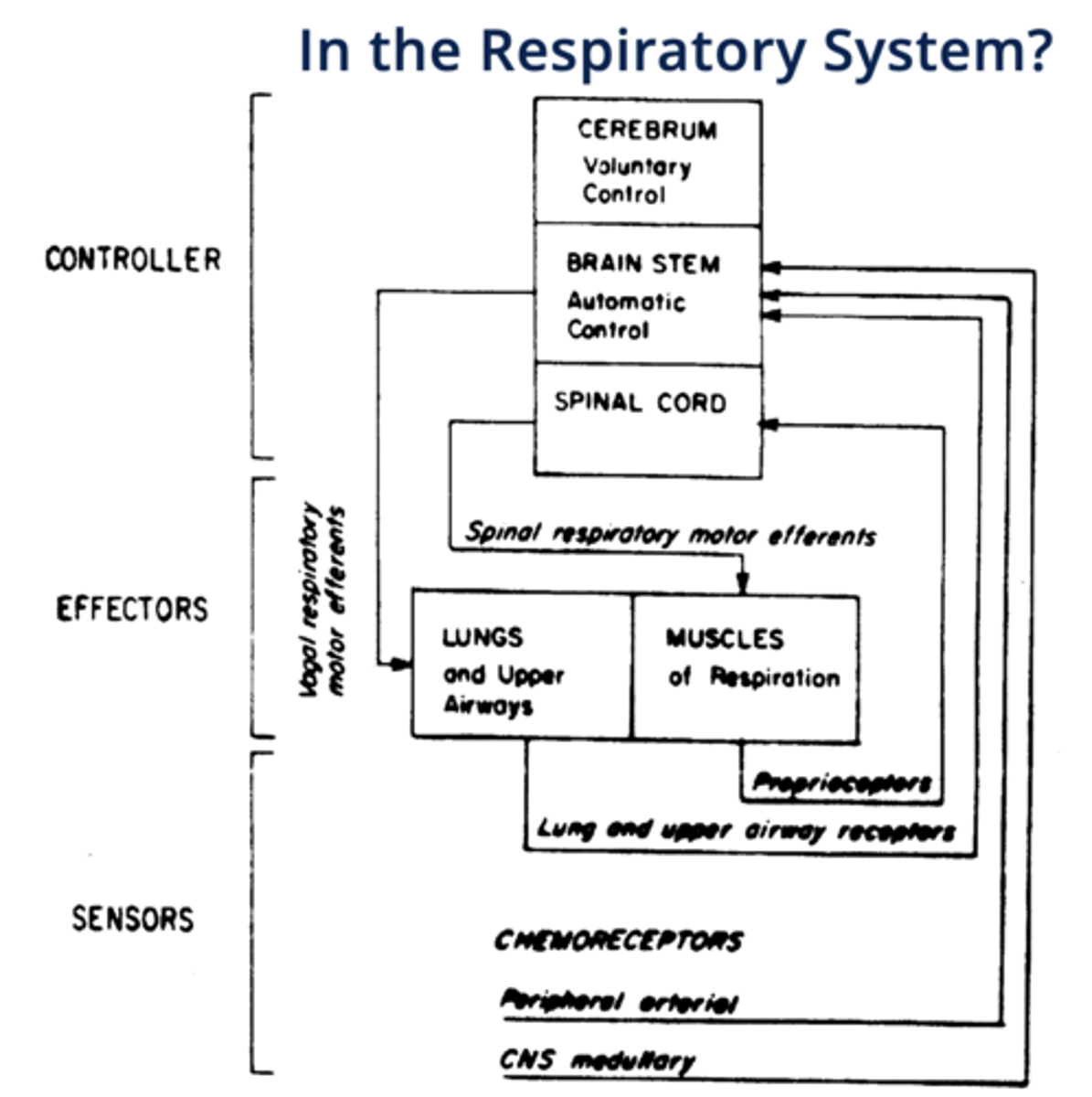
"Effectors" of respiration
lungs, upper airways and respiratory muscles
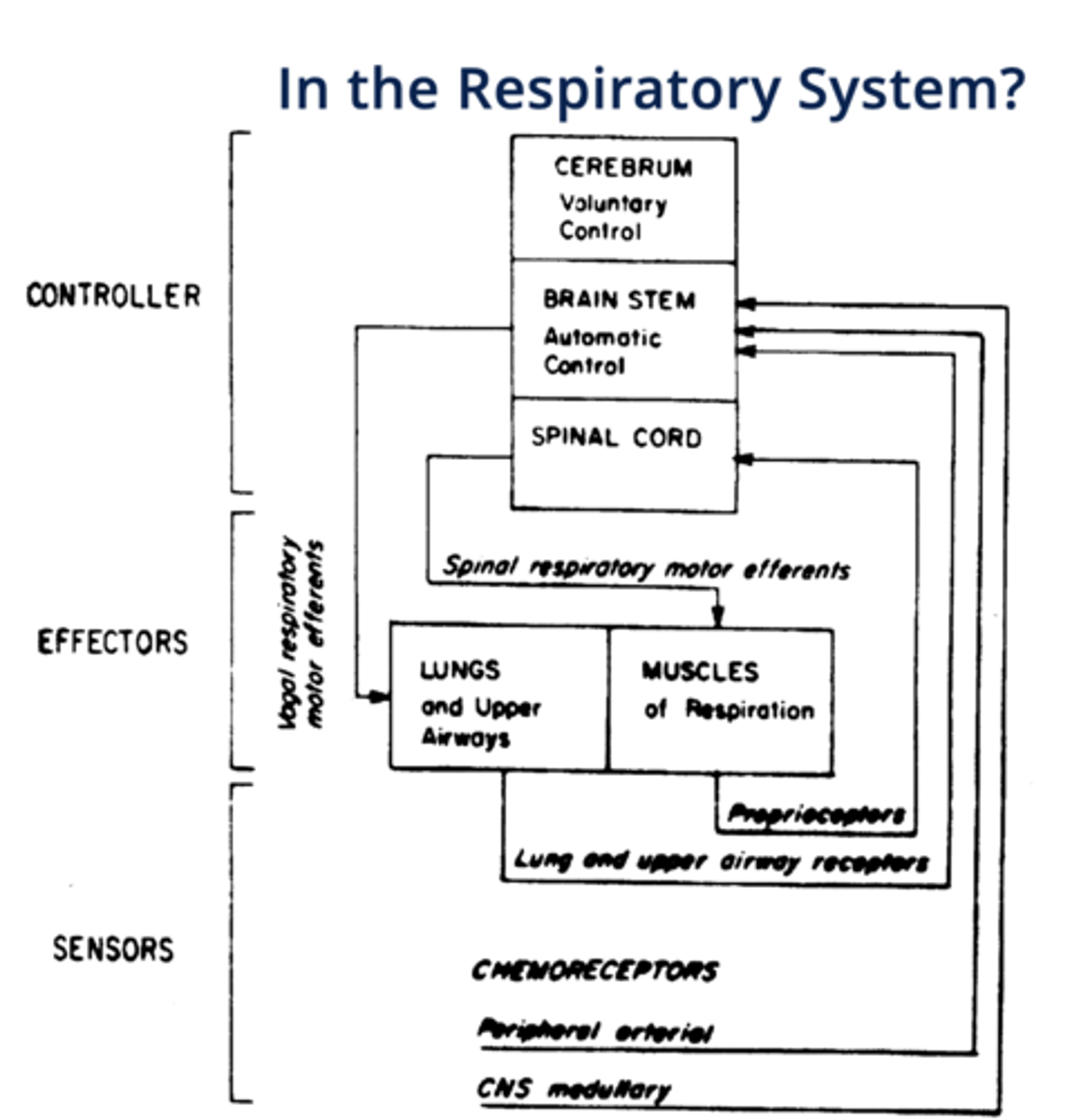
"Sensors" of respiration
chemoreceptors, mechanoreceptors
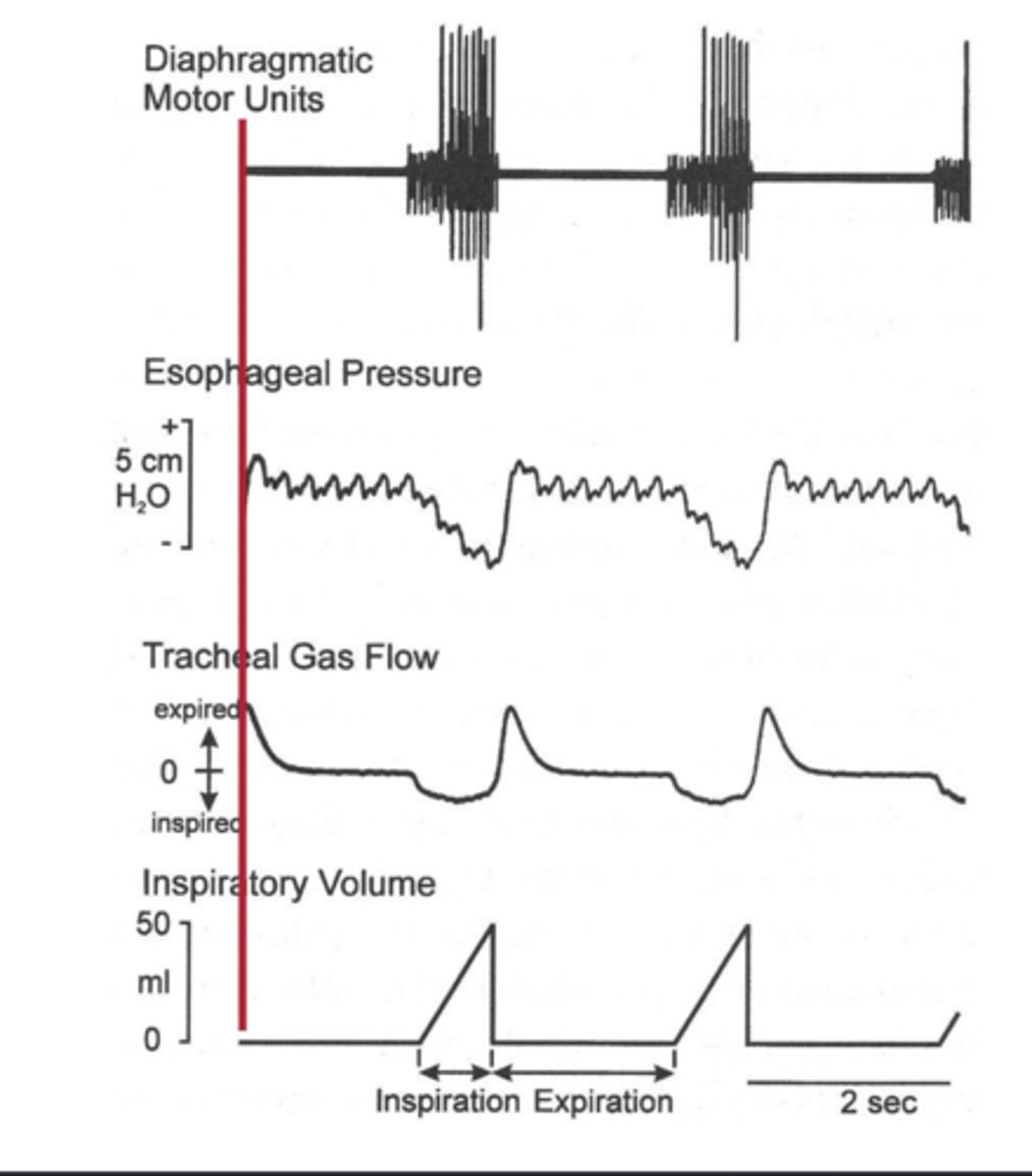
Rate Coding
Frequency modulation of motor unit activation.
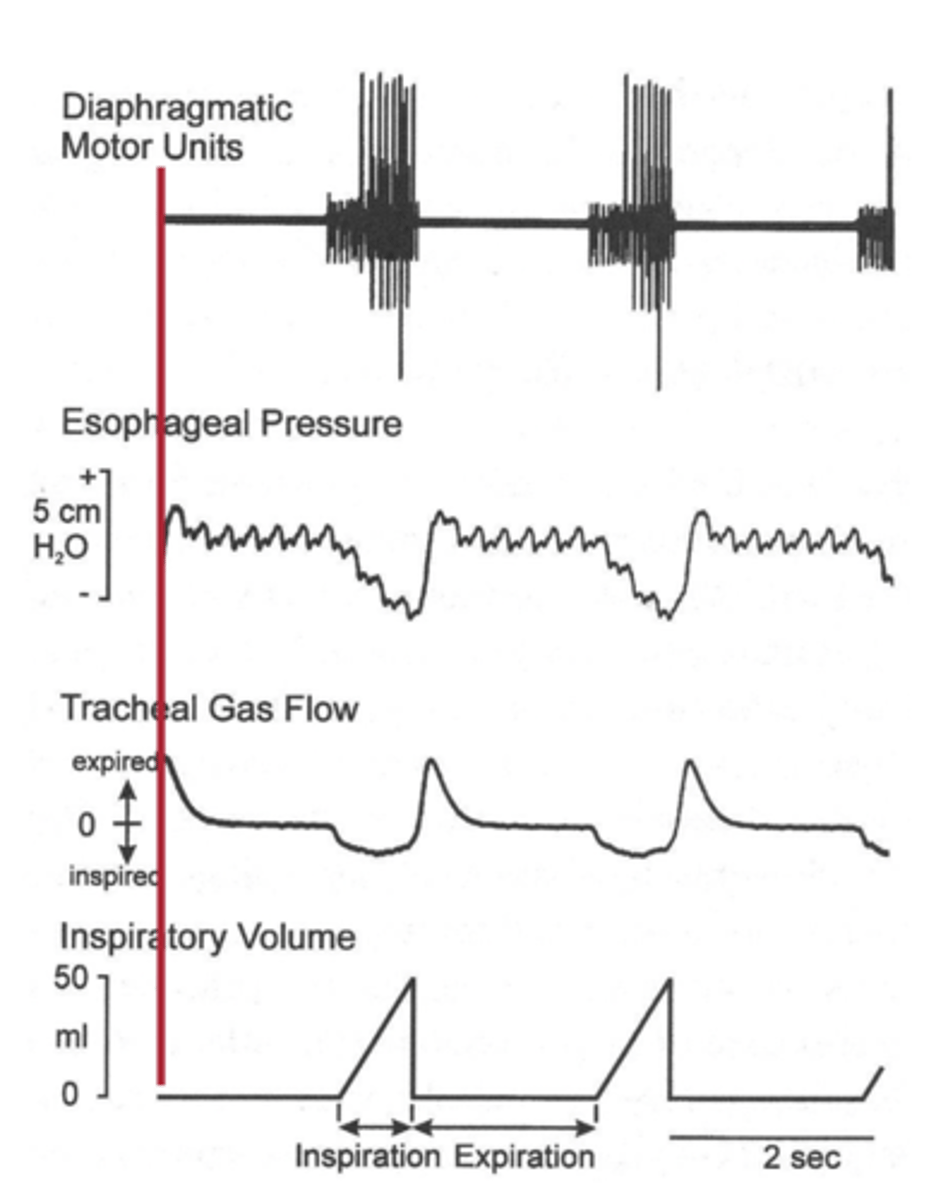
Recruitment of Motor Units
Number of motor units activated for muscle contraction.
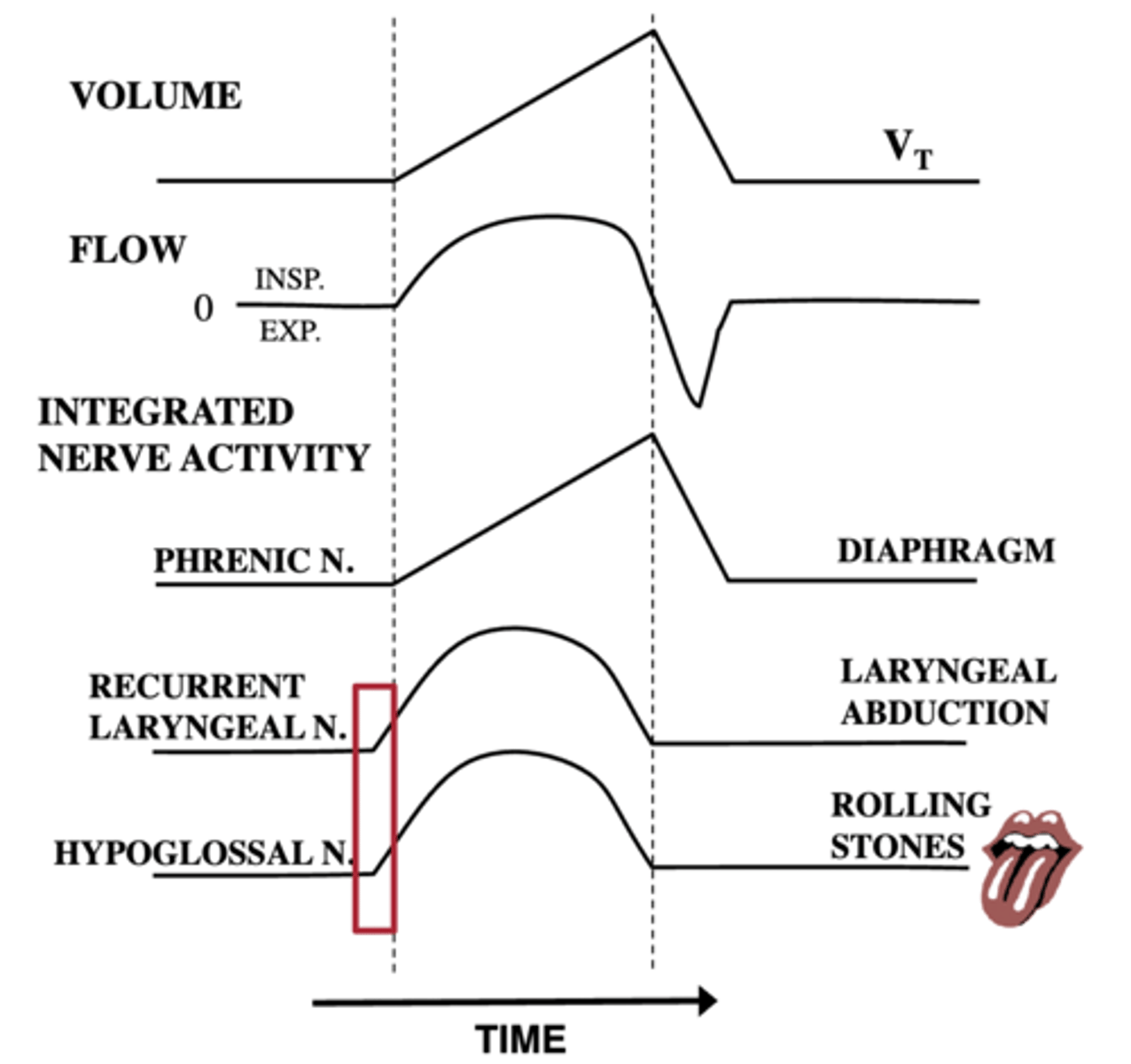
why do the smaller muscles (laryngeal and
hypoglossal) contract first
because they create a permissive area to decrease the resistance to make it easier for the bigger muscles, such as the diaphragm to contract
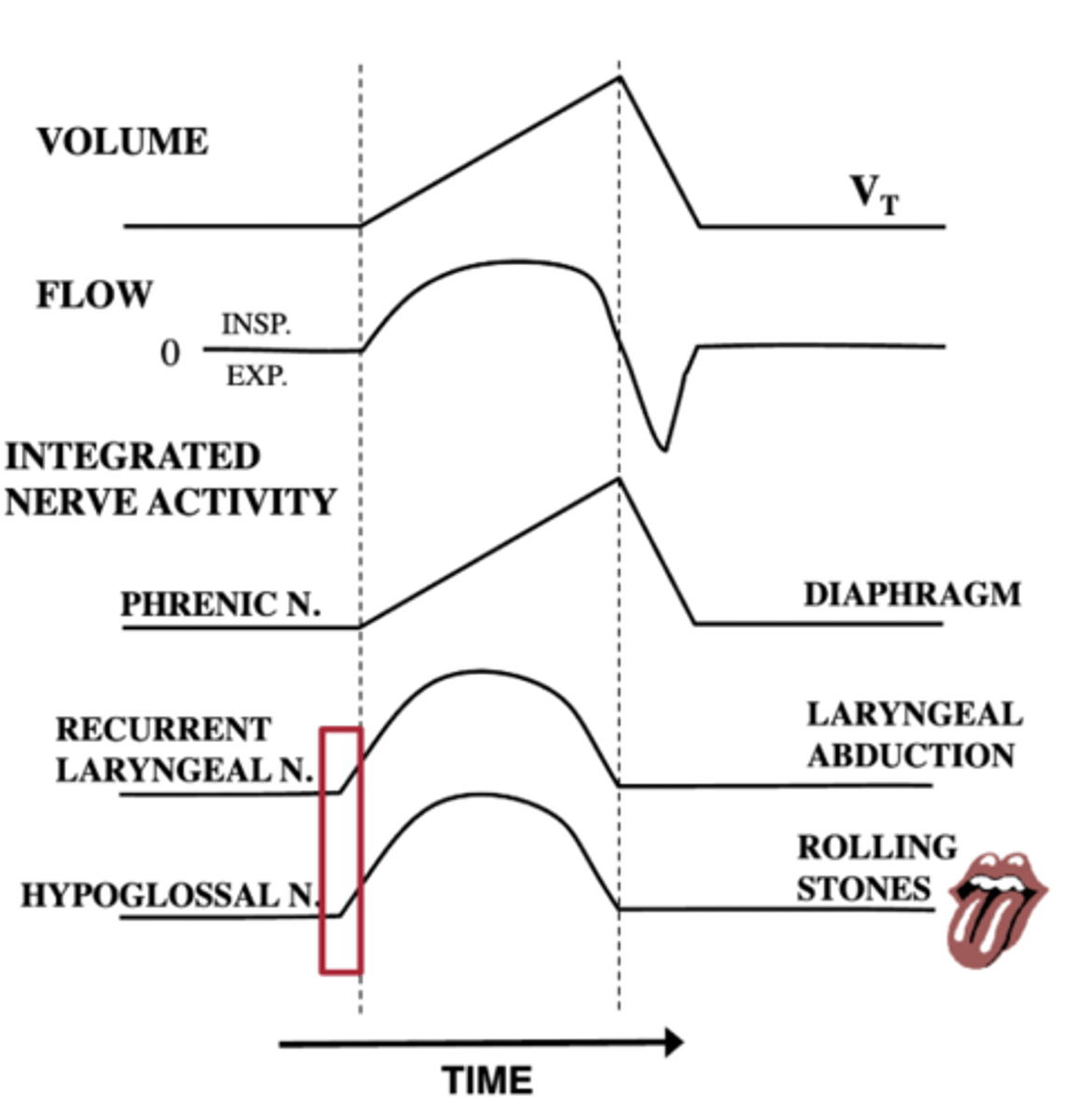
what does the phrenic nerve innervate
diaphragm
what does the hypoglossal nerve innervate
tongue
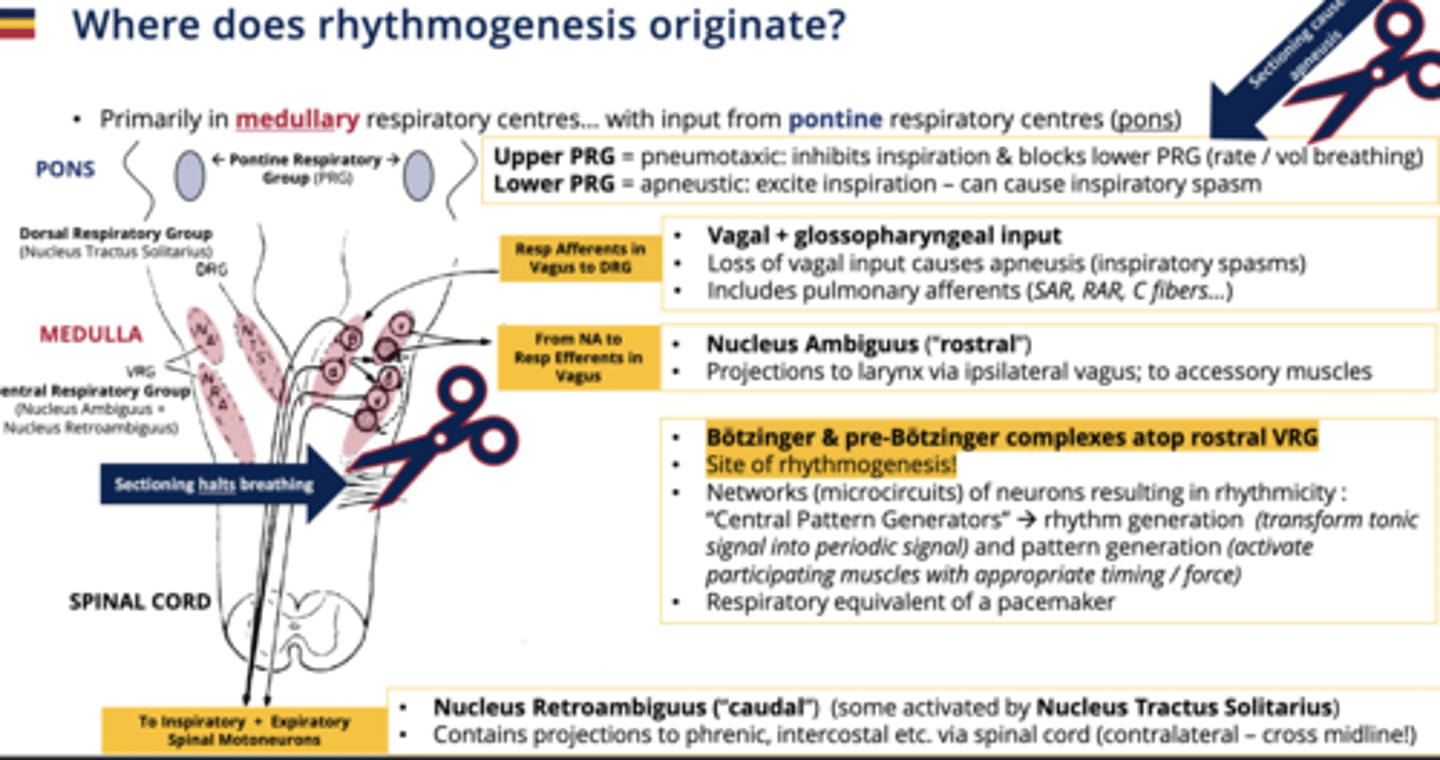
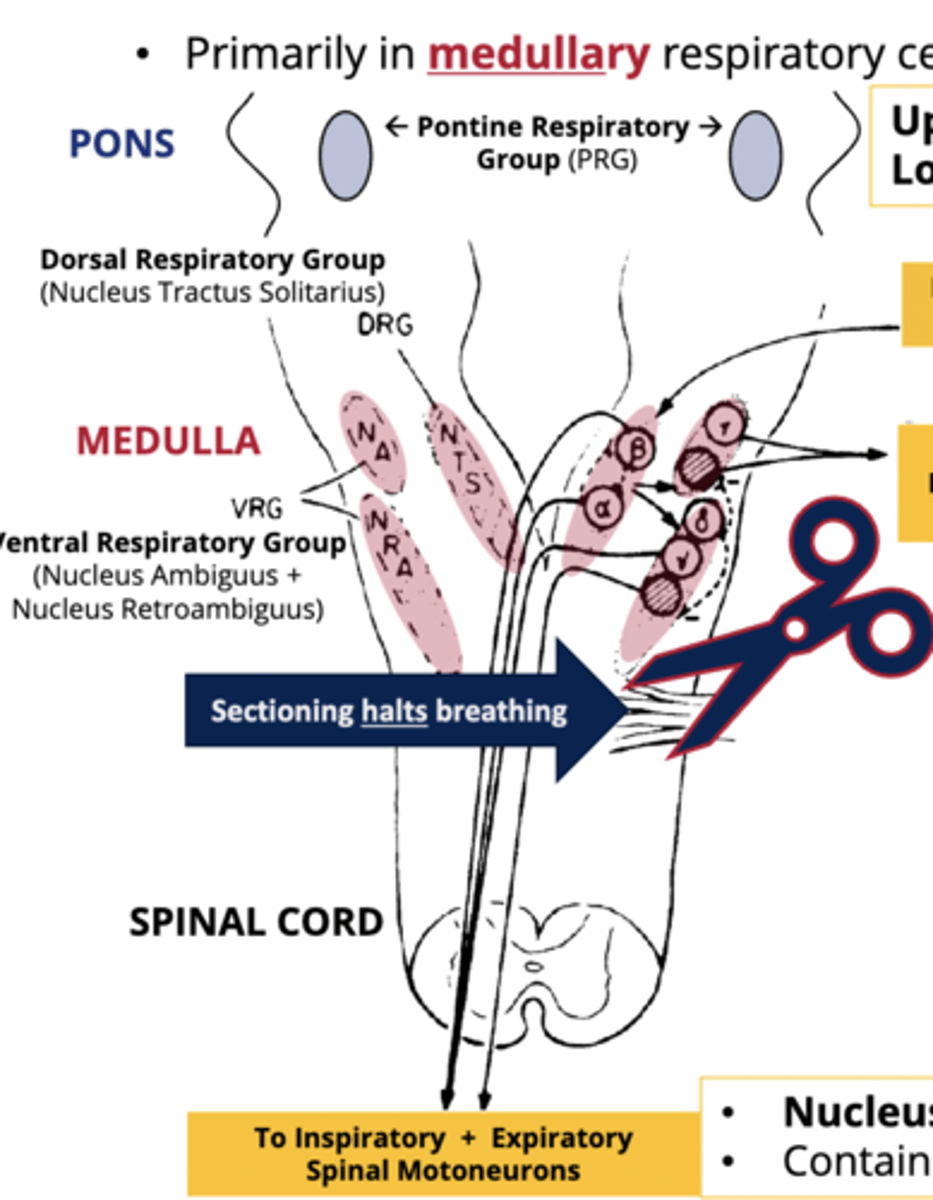
What does the pontine respiratory group consist of and what is it responsible for
upper (pneumotaxic) and lower (apneustic) groups: controls depth/rate of breathing
Pneumotaxic Center (Upper PRG)
Inhibits inspiration; regulates breathing rate, blocks lower PRG
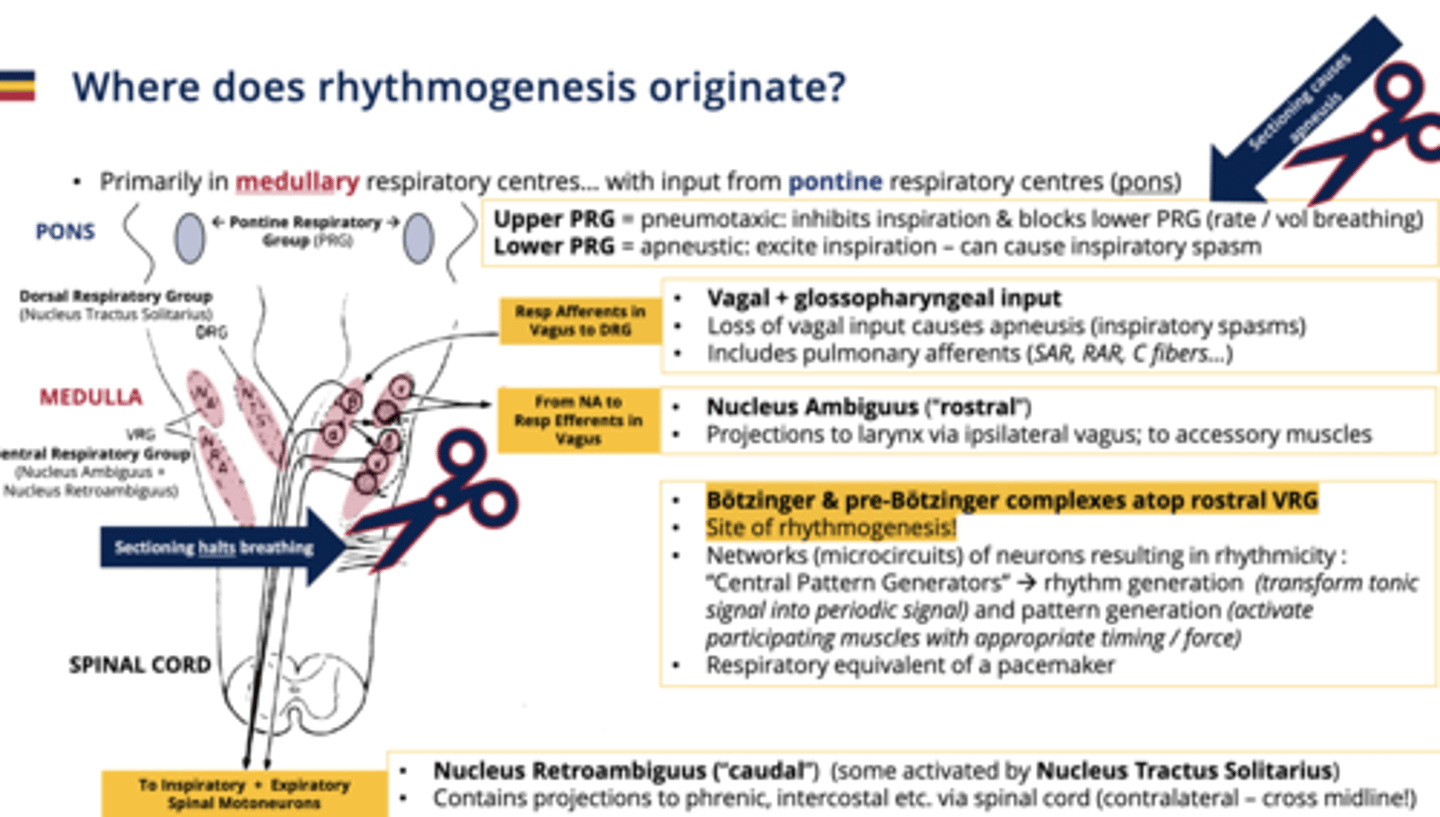
Apneustic Center (Lower PRG)
Promotes inspiration; can induce spasms.
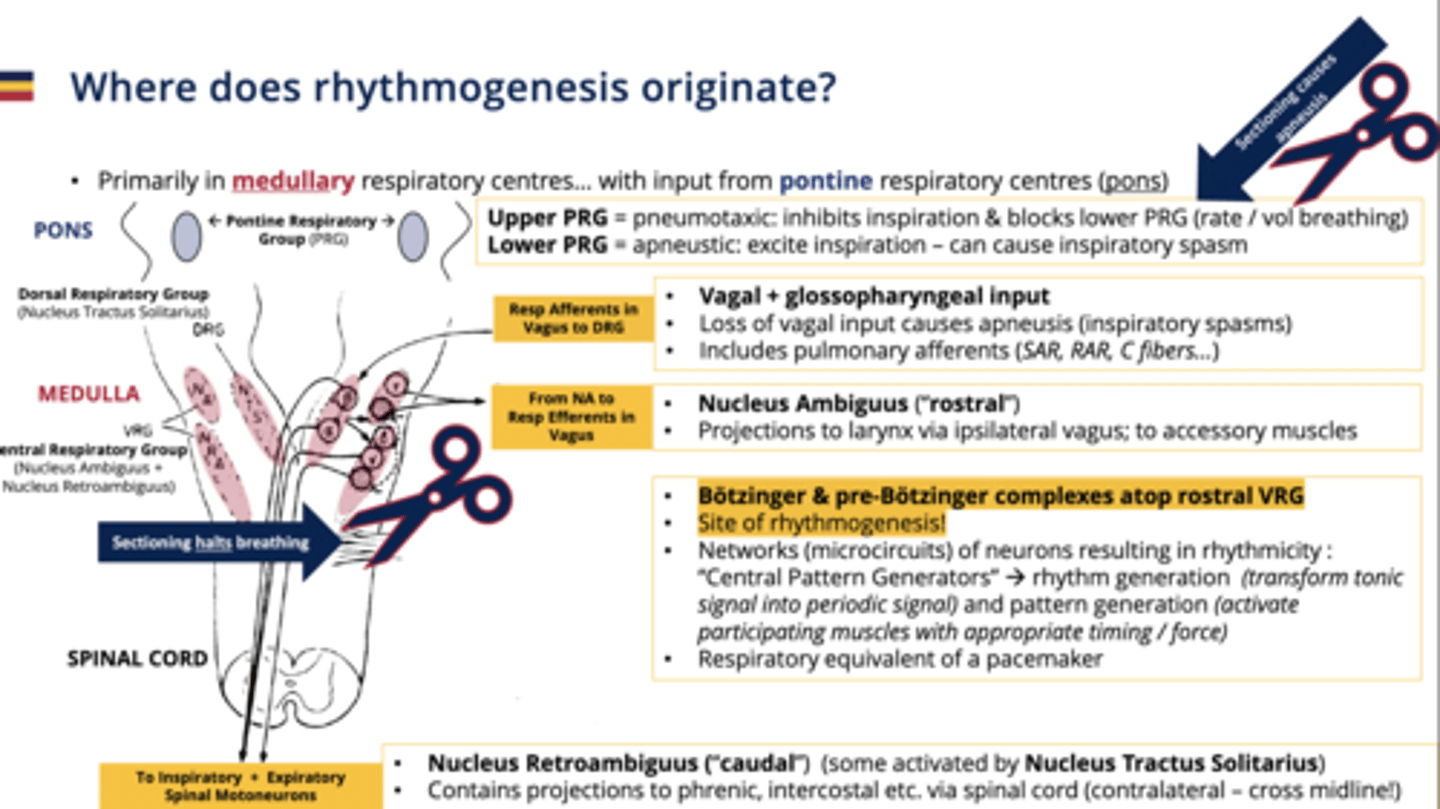
Dorsal Respiratory Group (DRG)
Initiates inspiration
-Receives input from chemoreceptors and pulmonary stretch receptors.
- primarily comprises the nucleus tractus solitarius.
- Vagal + glossopharyngeal input (includes pulmonary afferents)
nucleus tractus solitaris
PART OF THE DRG
has a series of different inputs that are coming from the rest of the body, primarily carried through the vagus nerve that can give input on things like breathing, depth and control.
- includes a number of pulmonary afferent fibers
how does the DRG communicate with other nerves
-resp afferents in the vagus nerve are sent to the DRG
- also pulmonary afferents communicate here (SAR, RAR, C fibres)
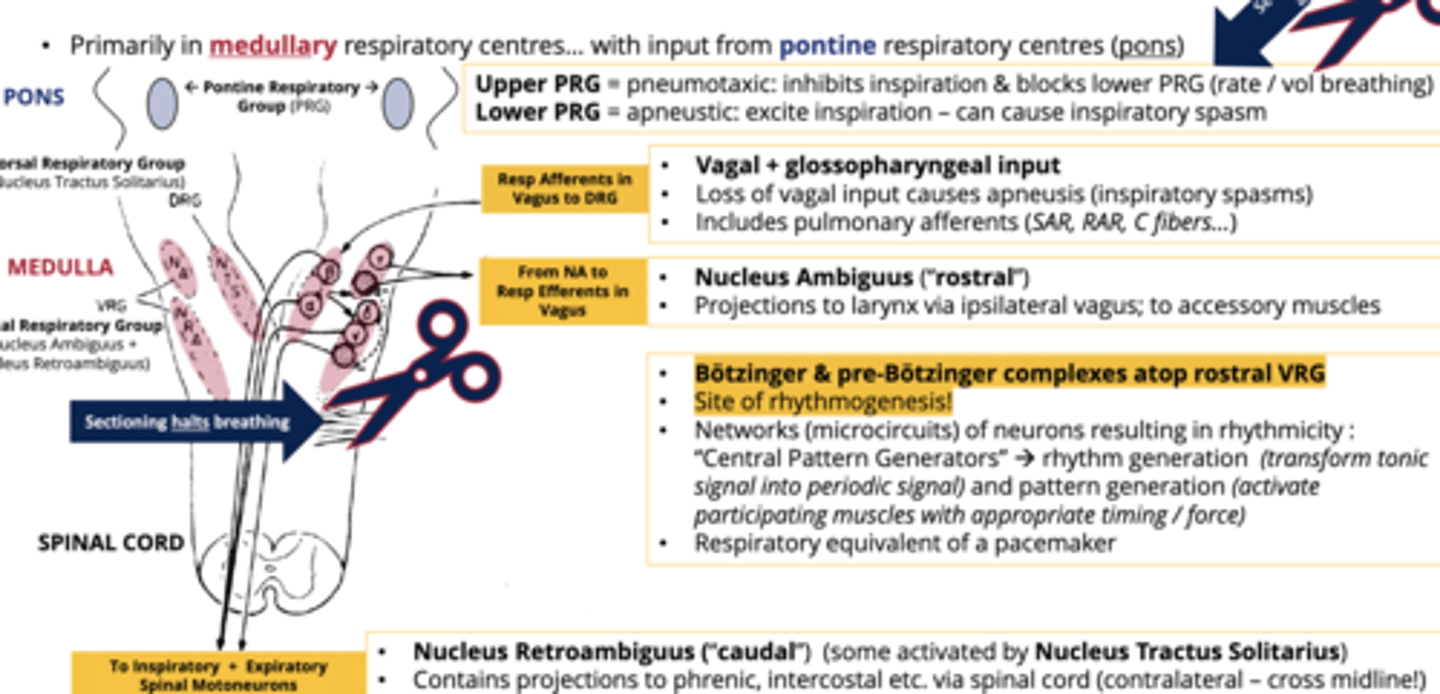
What are the two ventral respiratory groups
1. Nucleus ambiguus (rostral/front)
2. Nucleus retroambiguus (caudal/back)
what does sectioning the upper PRG and lower PRG cause
apneusis

what does a loss of vagul input to the DRG cause
apneusis (inspiratory spasms)

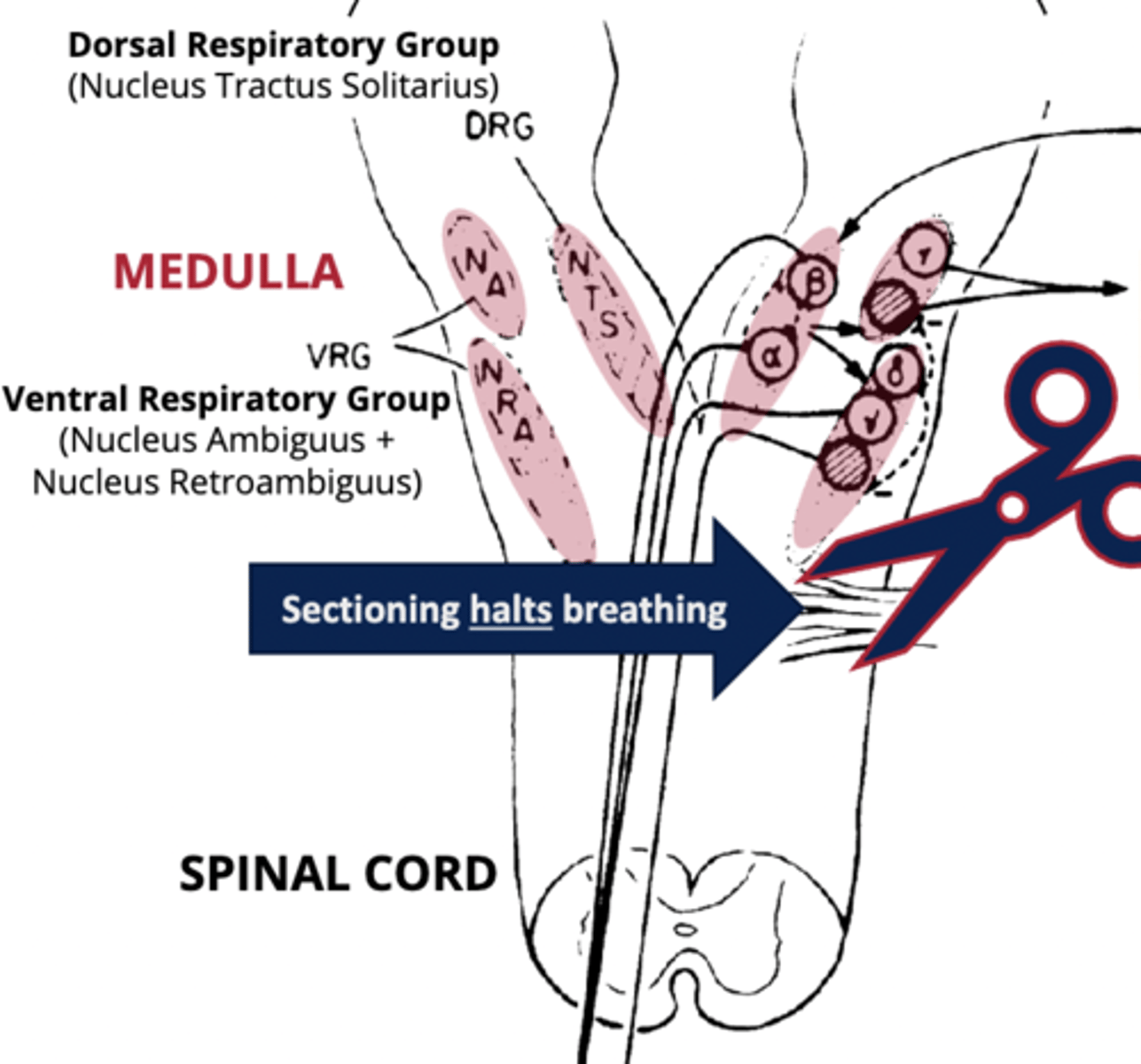
Ventral Respiratory Group (VRG)
Controls inspiratory and expiratory motor neurons.
- primarily comprises the Nucleus Ambiguus & Nucleus Retroambiguus.
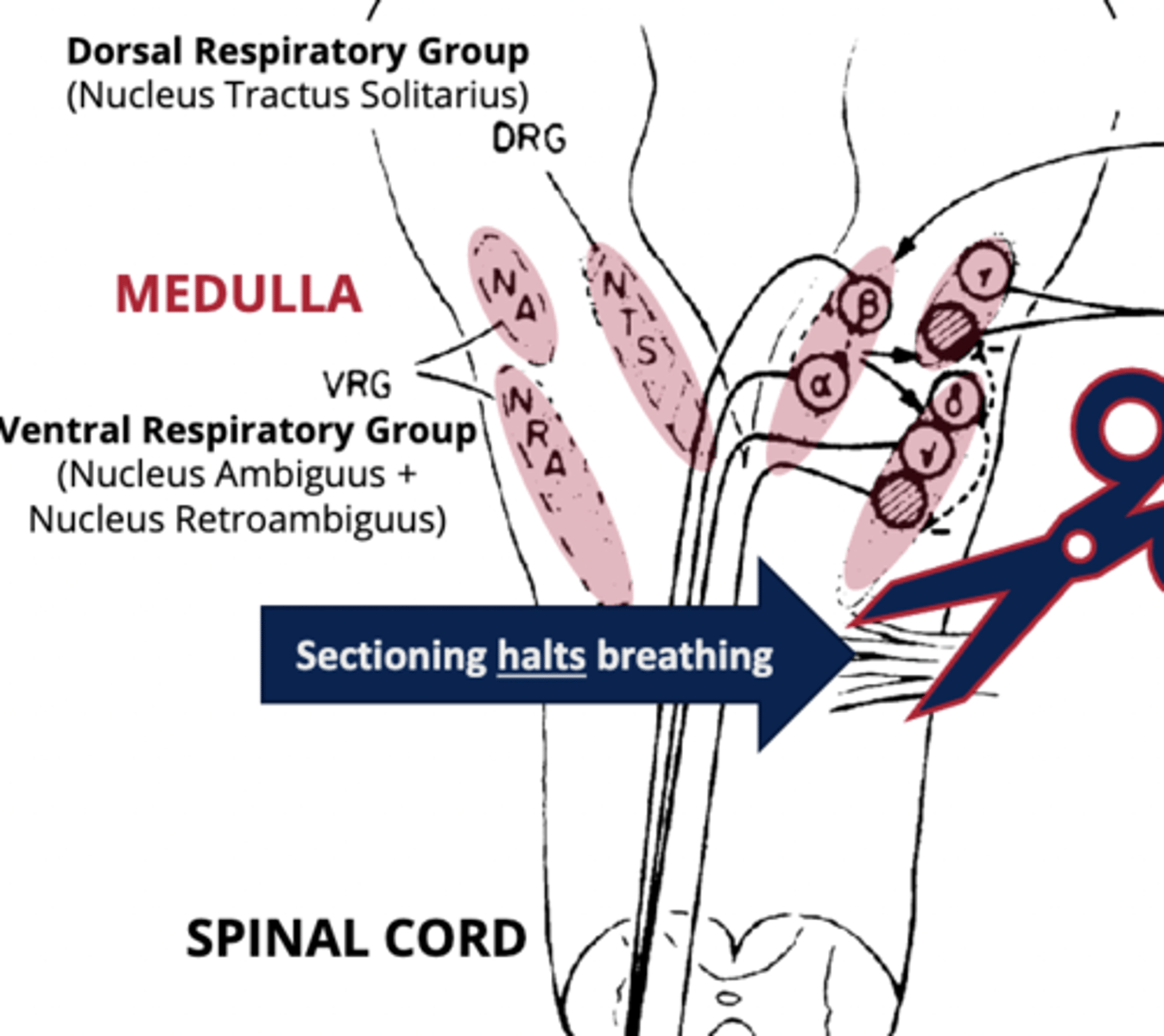
nucleus ambiguus
part of the VRG that contains neurons that project through the vagus nerve to larynx and accessory muscles
- primarily dealing with things that are upper airway controllers.

nucleus retroambiguous
part of the VRG that houses the projections that are going, via the phrenic nerve to the diaphragm, or going through other neurons to the intercostal muscles
- most caudal part of VRG
Botzinger/ Pre-Bötzinger Complex
- located atop the ventral respiratory group
- sites of primary rhythmogenesis
- take tonic signals from higher pontine group and introduce rhythm/modulation
- determine tidal volume/breathing frequency
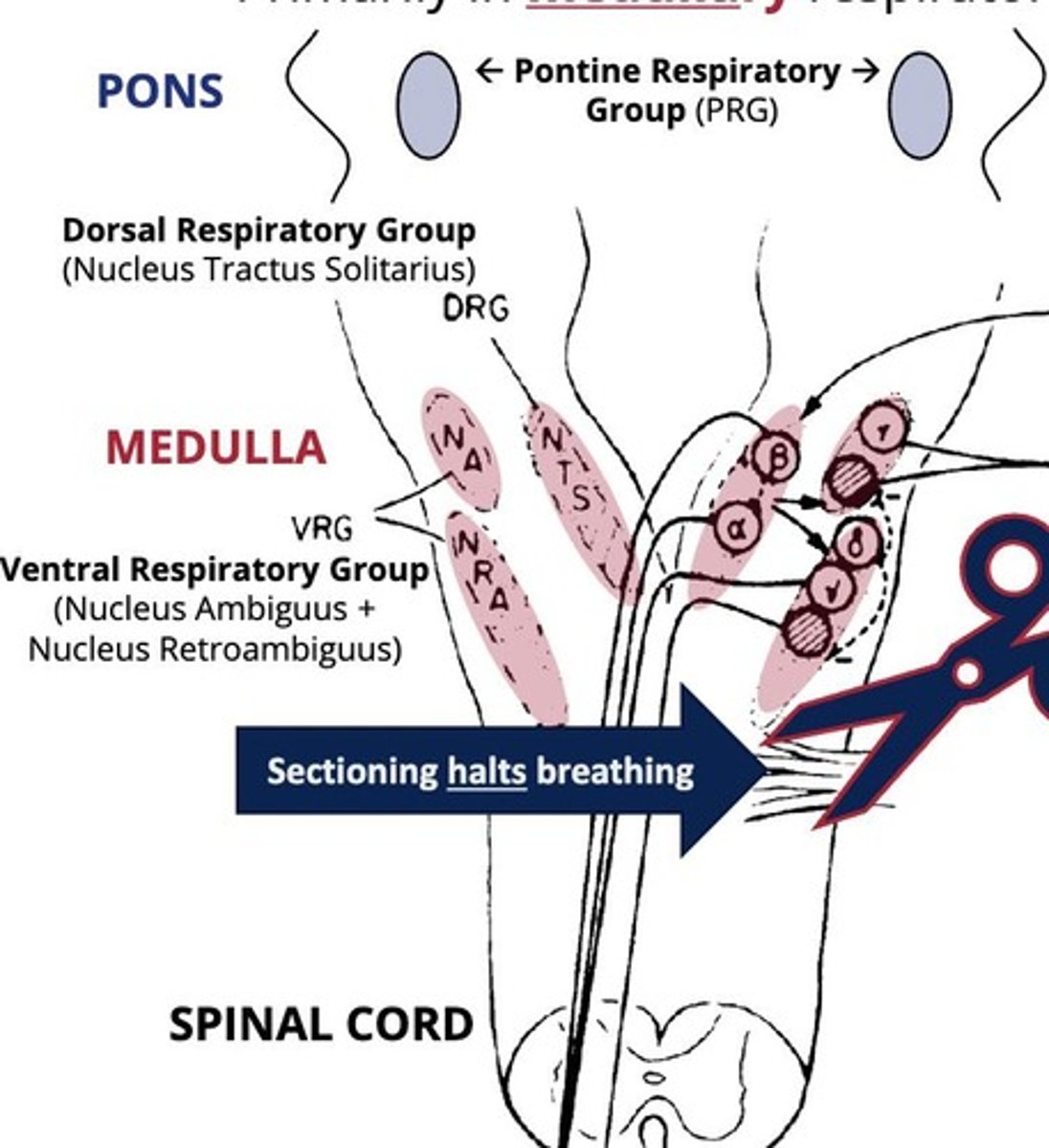
Central Pattern Generators
Transform tonic signals into rhythmic breathing.
- in the Pre-Bötzinger Complex
- also influenced by vagal inputs

expiratory pattern generation towards what part of the spine
medial
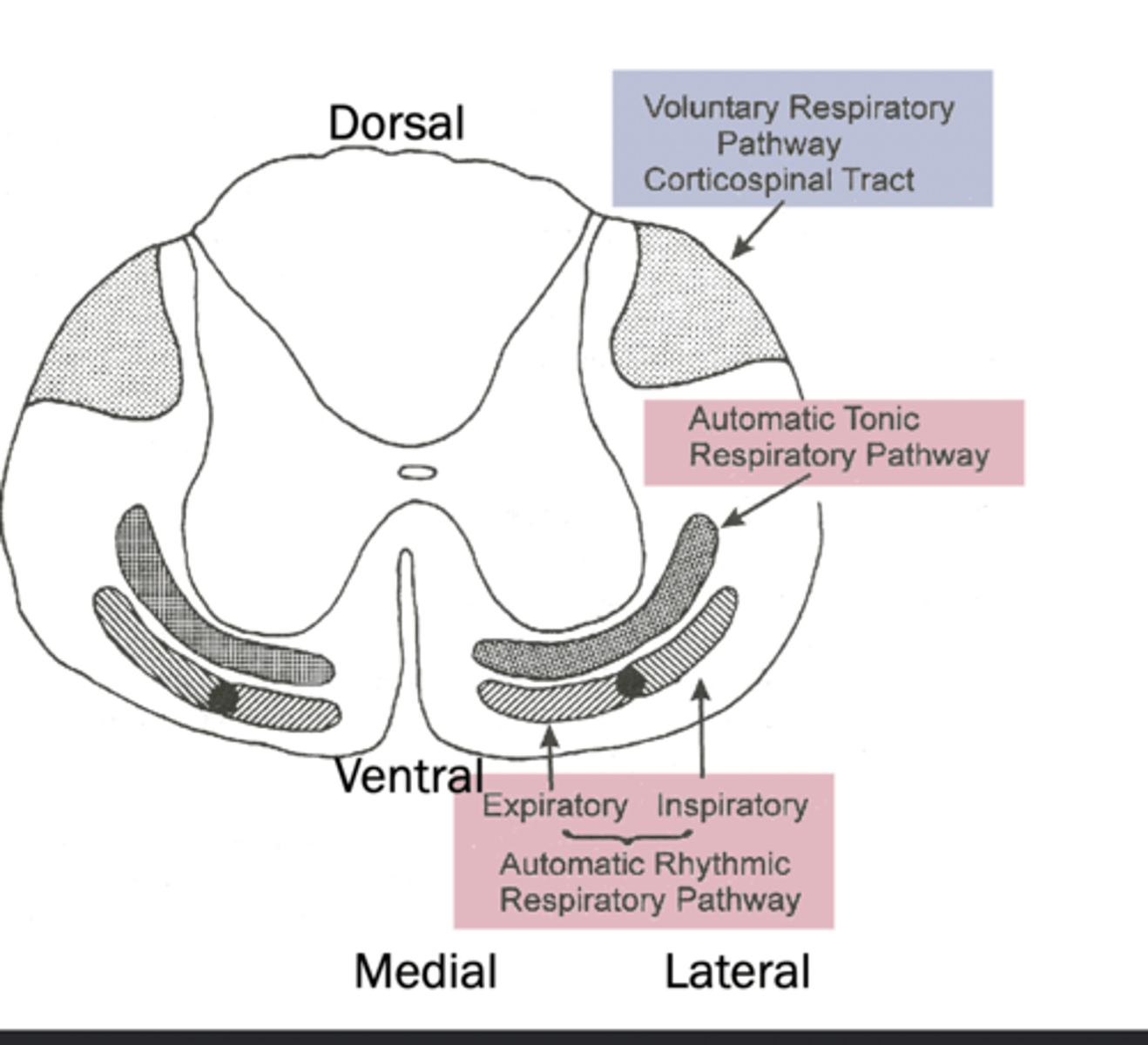
inspiratory pattern generation towards what part of the spine
lateral
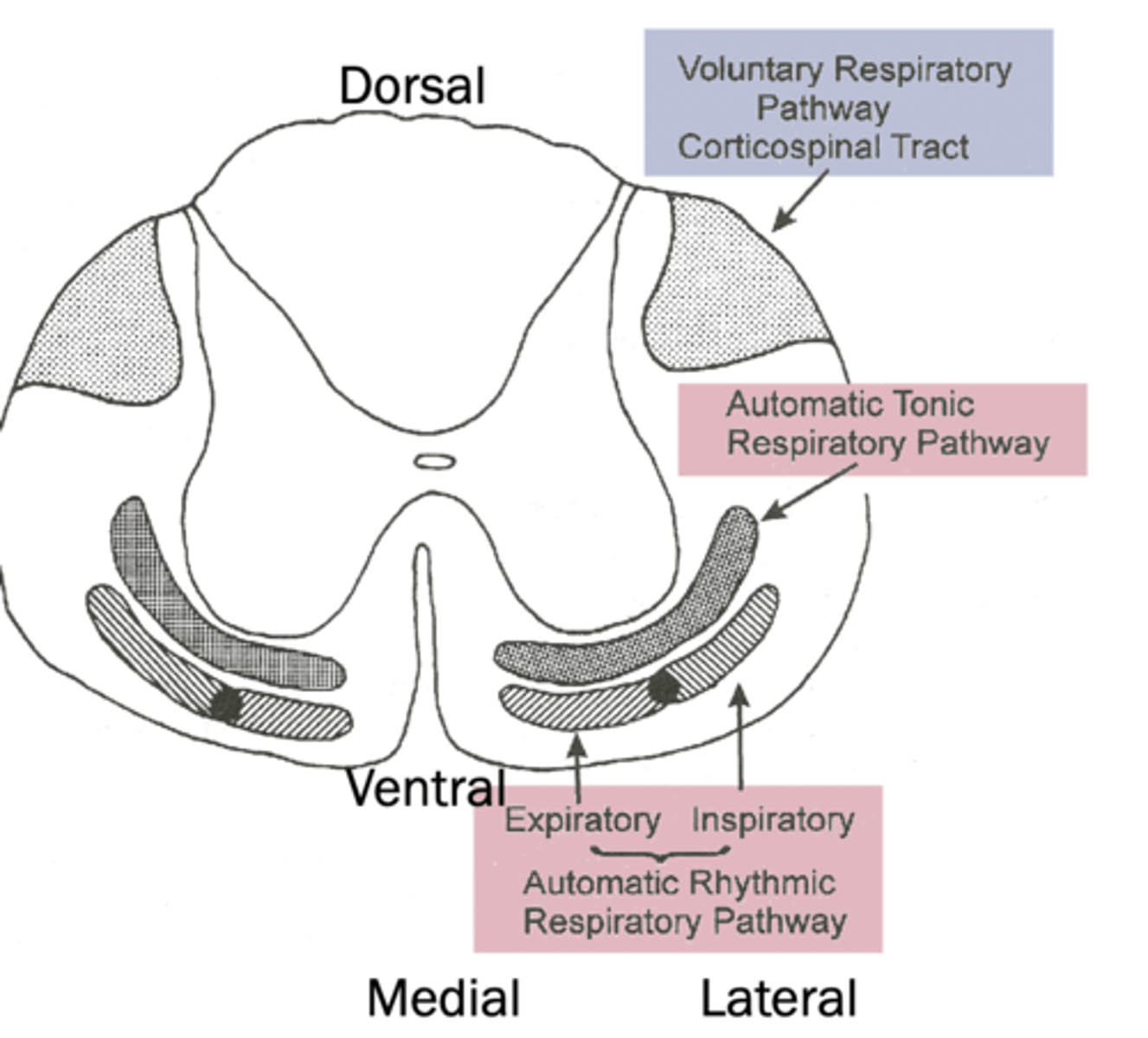
Motor Cortex effect on breathing
Controls voluntary breathing actions.
Phrenic Nerve role in breathing
Innervates diaphragm for breathing control.
voluntary control of breathing pathway location
dorsal, comes from cerebrum
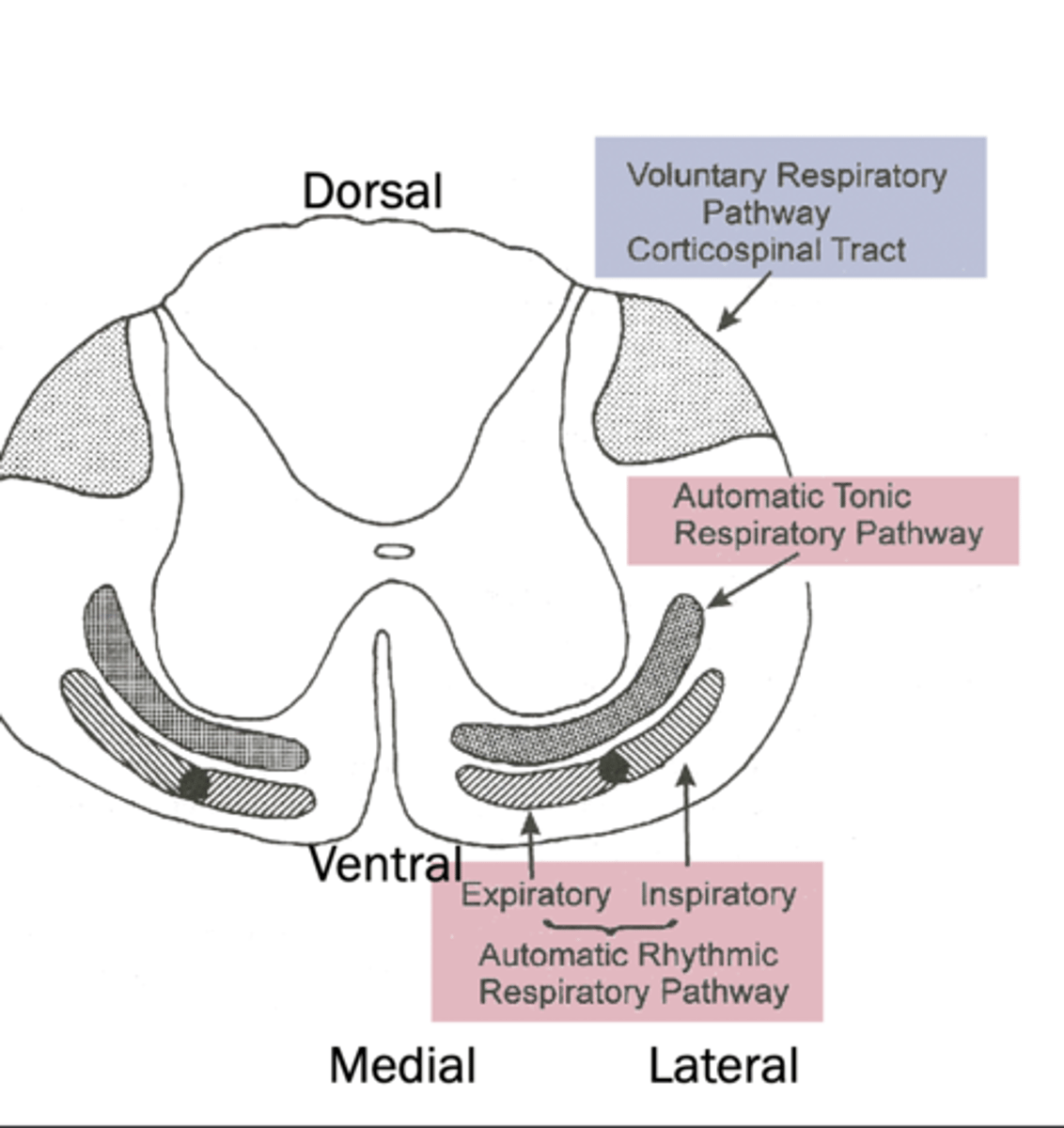
automatic tonic respiratory pathway location
ventral
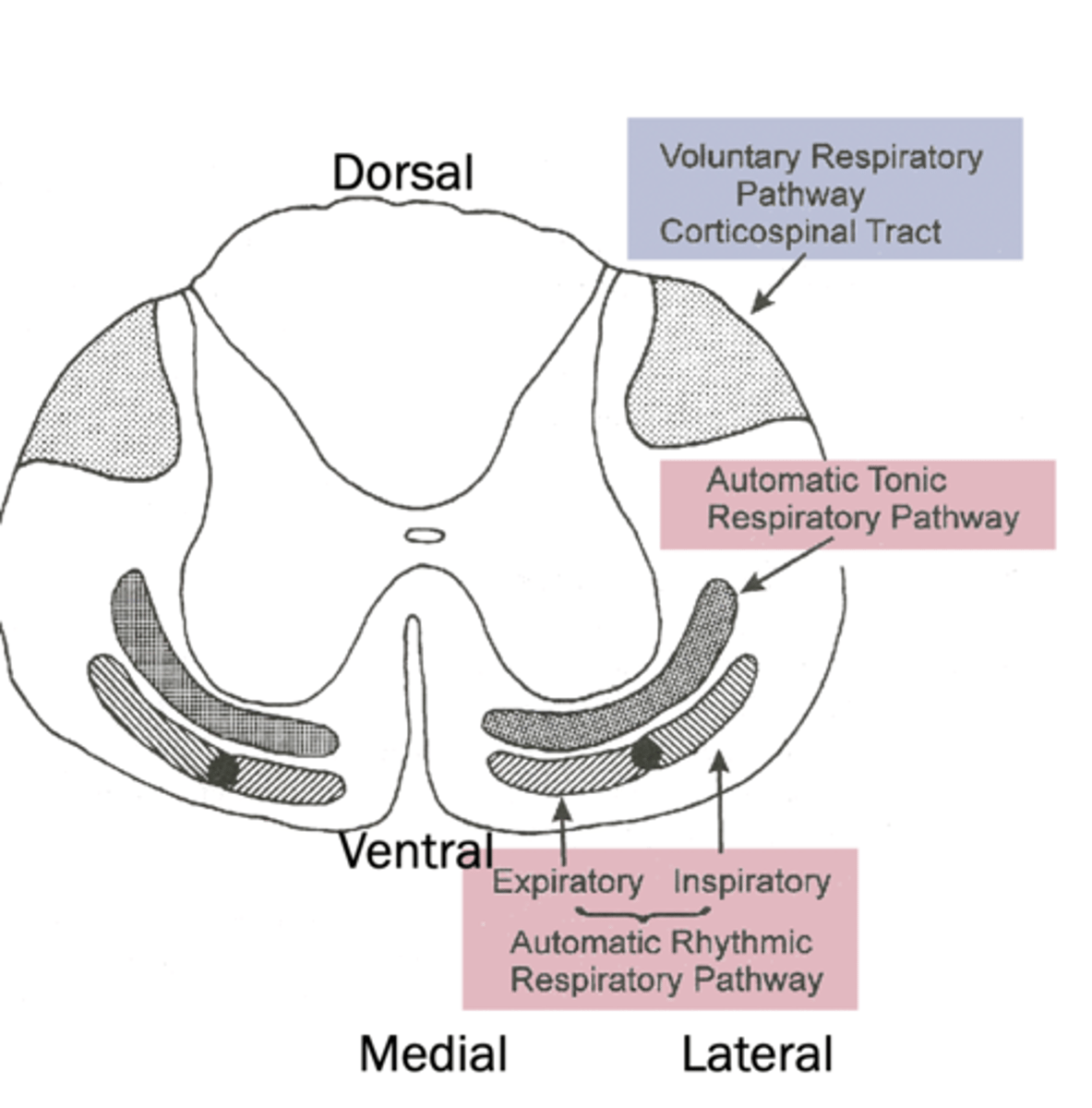
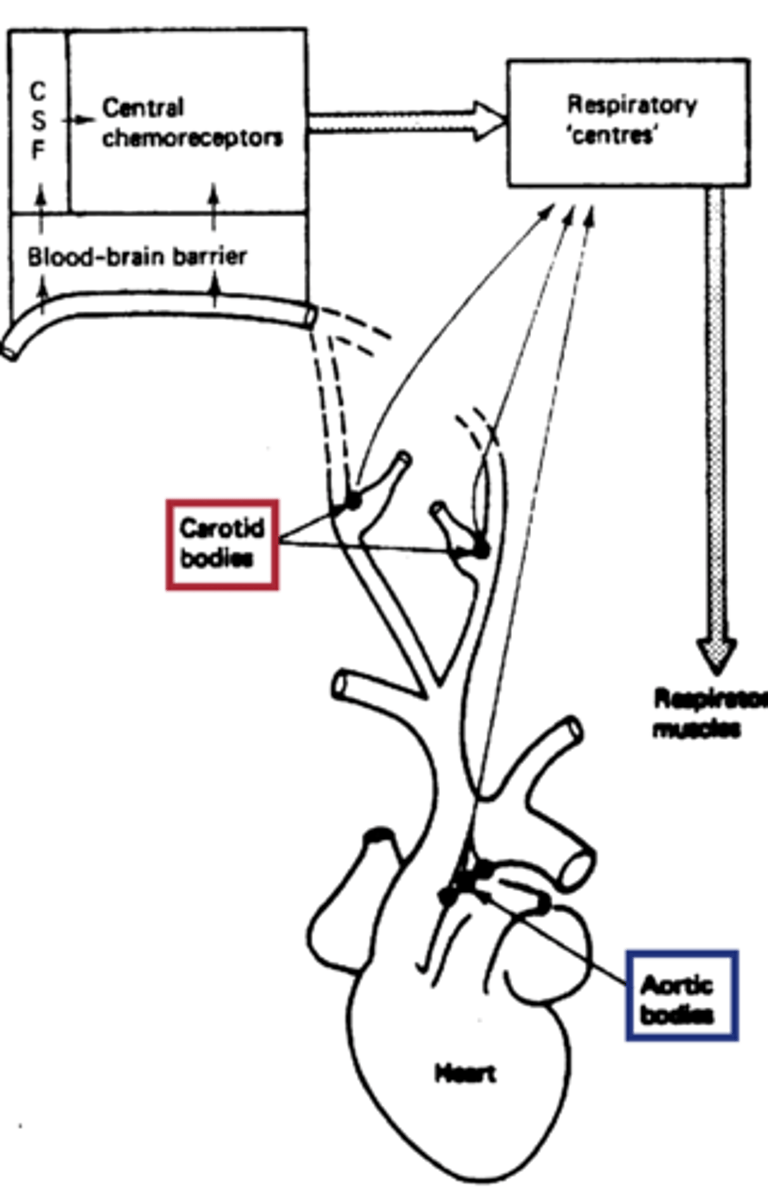
Peripheral Chemoreceptors
- Monitor blood gases in the periphery
- include carotid and aortic bodies.
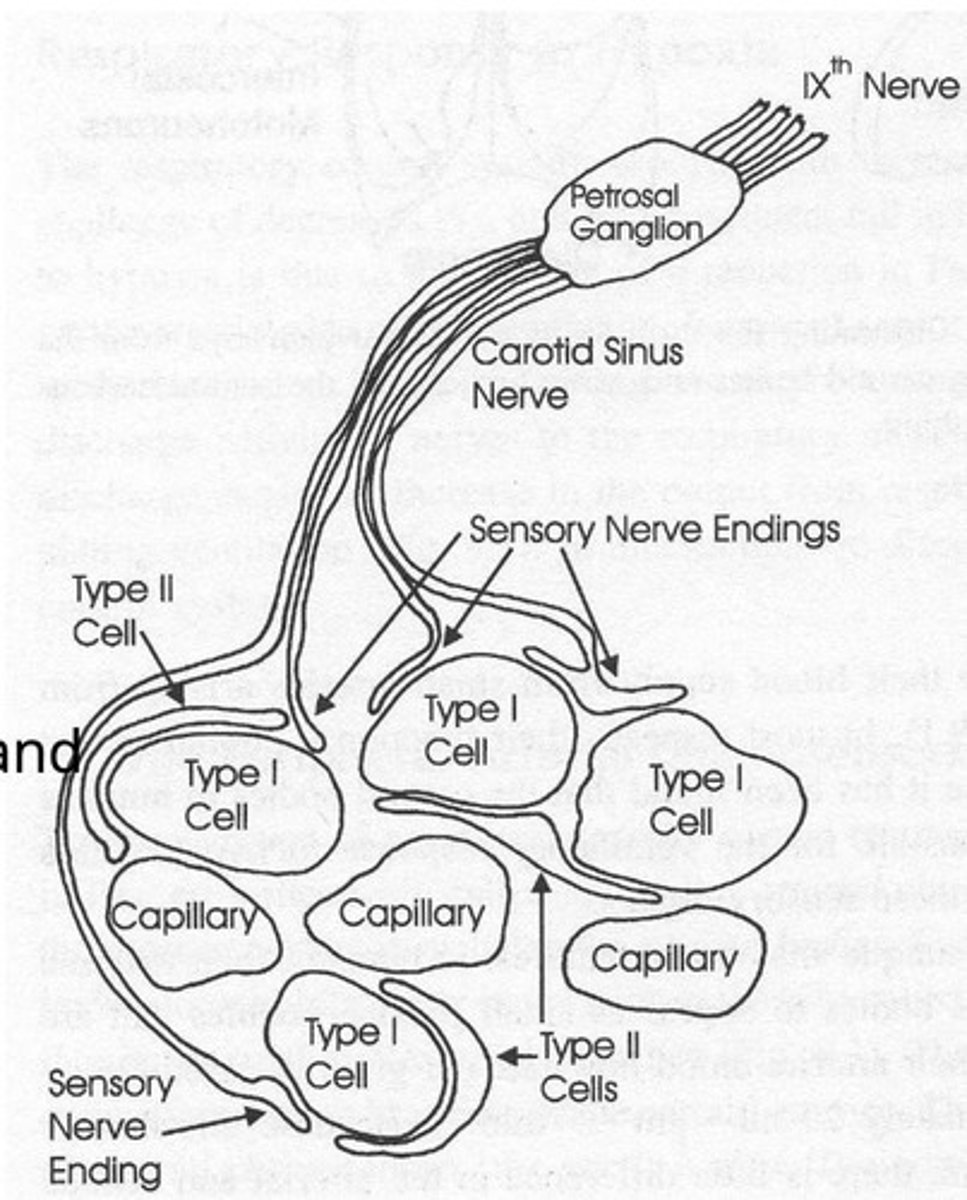
Carotid Bodies
Primary peripheral chemoreceptors; detect blood gas changes.
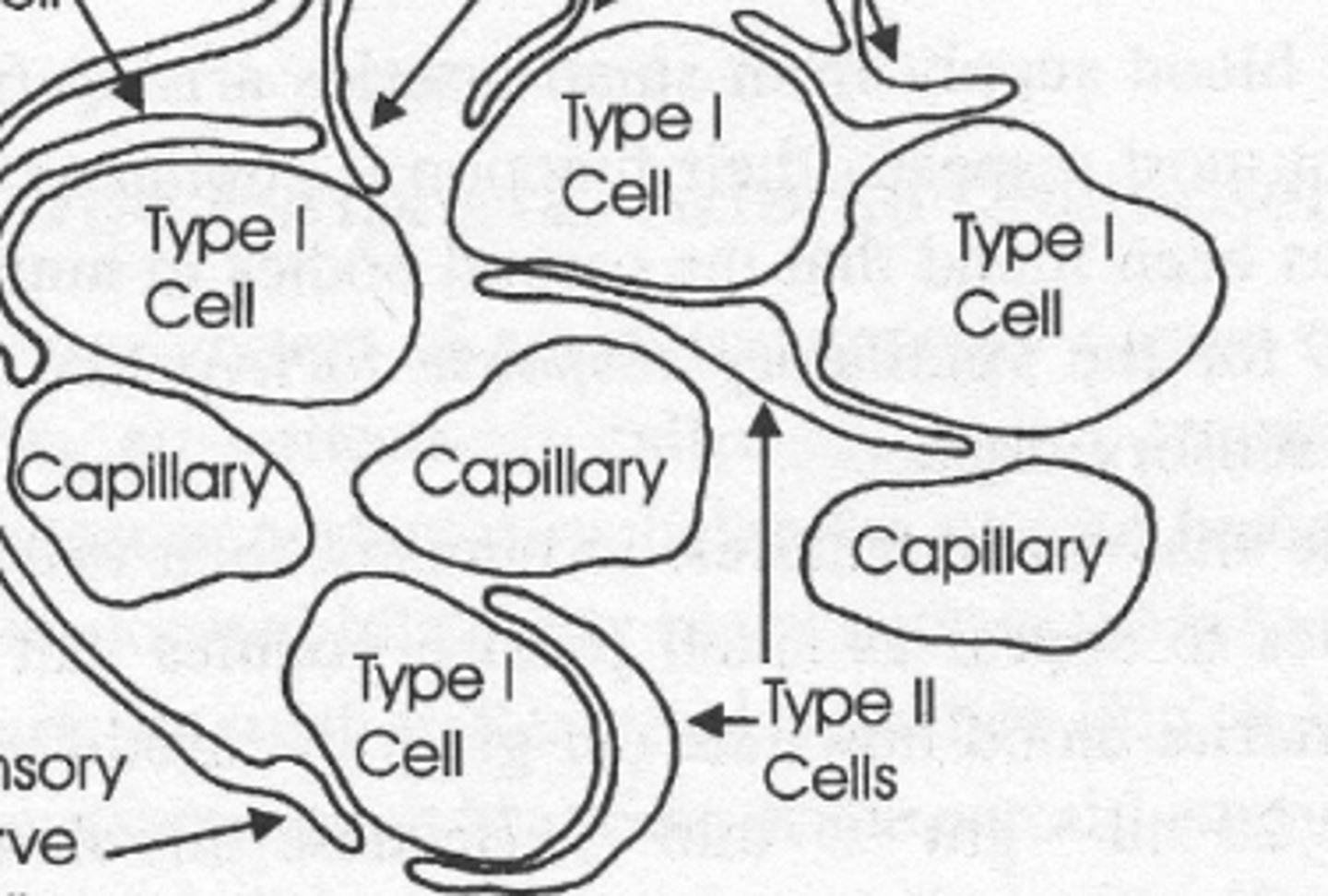
- composed of type 1 and type 2 cells
- projects via carotid sinus nerve branch of glossopharyngeal nerve to NTS
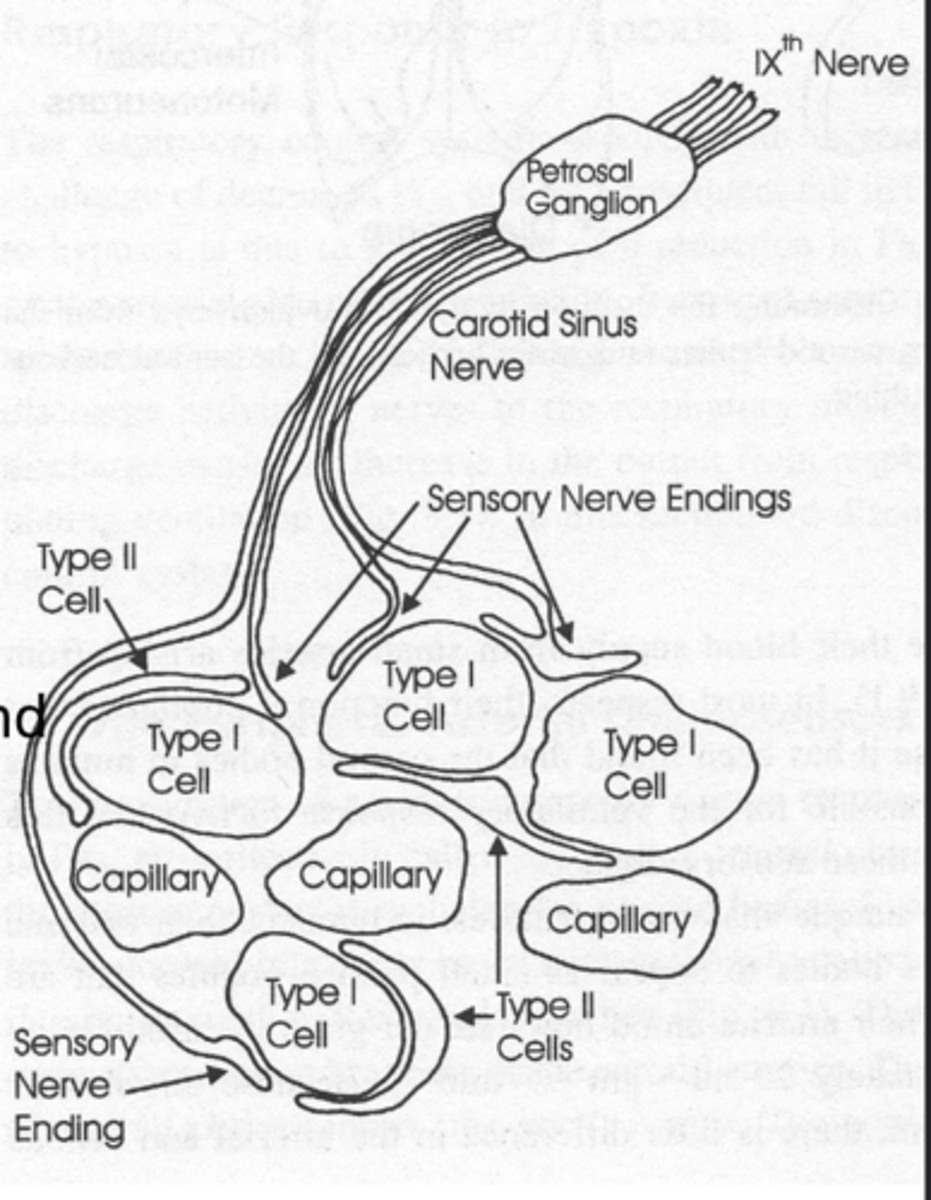
Type I Glomus Cells
Detect ↓PaO₂, ↑CO₂, ↑H⁺ and synapse with sensory endings
- projects via CSN branch of IX cranial nerve to NTS of medulla
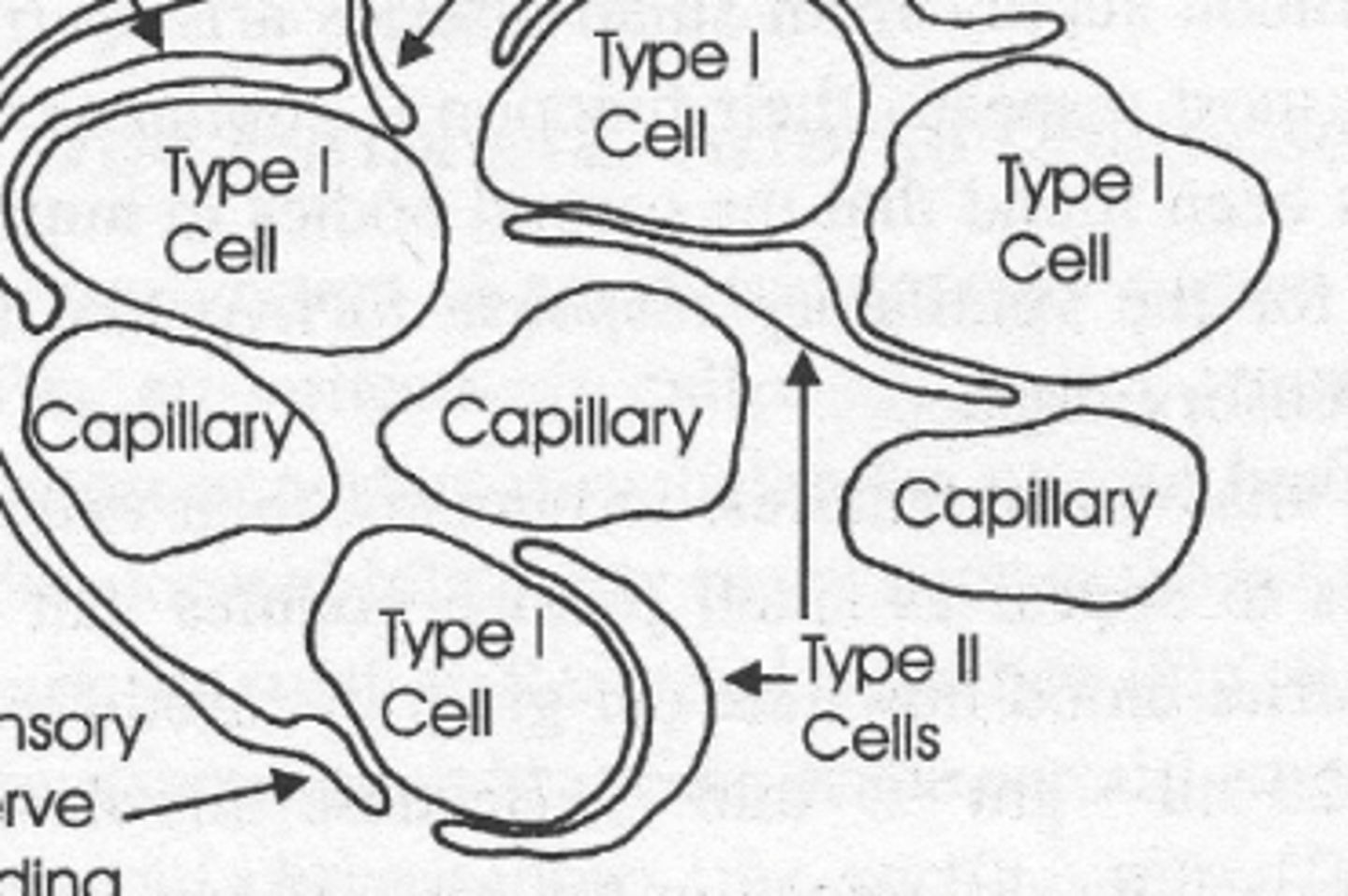
Type 2 Sustentacular Cells
Provide structural support to Type 1.
Aortic Bodies
Detect O₂ and CO₂; signals via cranial nerve.
- More involved in cardiovascular regulation.
- via X cranial nerve
Carotid Body Response to Hypoxia
senses arterial PaO₂ (arterial partial pressure) and NOT O2 content
- Responsible for 100% of ventilatory response to hypoxia in humans
what is Oxygen content largely driven by
by how much oxygen is bound to hemoglobin and also how much hemoglobin do you have
PaO2
how much soluble dissolved oxygen is in your blood based on the partial pressures in the environments
why is the carotid body not good at sensing when it comes to anemia
because their PaO2 is perfect but they might have a very low O2 content and you carotid body cant sense this and thinks everything is fine because of the PaO2 levels
- same with carbon monoxide poisoning
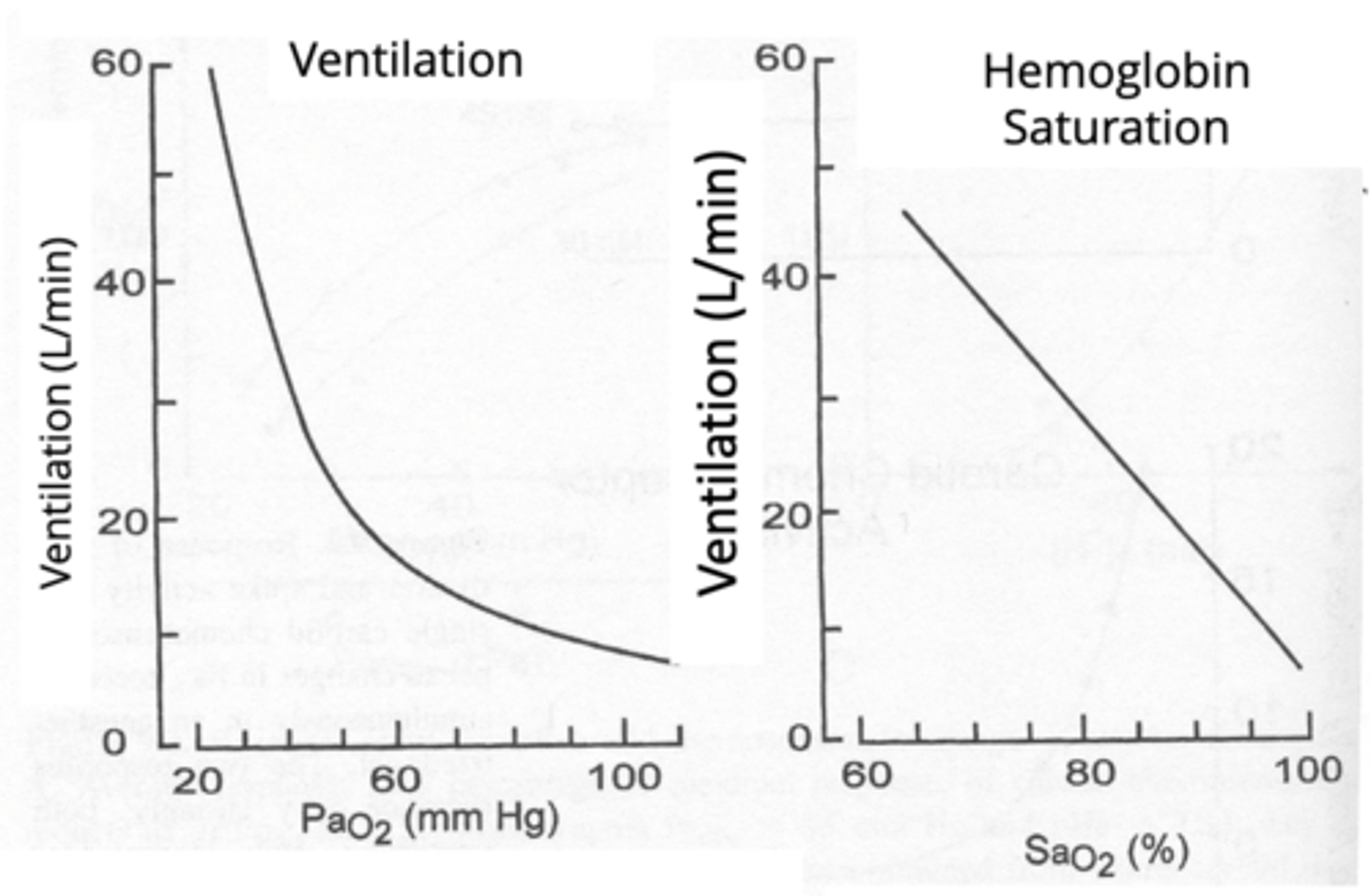
Nonlinear vs linear Response of Carotid Body Ventilation Response to Hypoxia
Nonlinear: response to PaO2
- Ventilation increases significantly when PaO₂ < 60 mmHg.
Linear: with SaO2 due to the hemoglobin-oxygen dissociation curve.
why are the carotid bodies so good at sensing PaO2
because they are located in the closest place to the heart where the fresh blood that's just come from the lungs is coming out
Anything that's going to increase the level of hydrogen ion in your blood such as acidosis or from exercise, is also going to be sensed by what
the carotid body and will contribute to a similar ventilation-stimulating mechanism as well.
How can the carotid body also sense changes in H+ and CO2?
It is multimodal meaning it responds to a variety of stimuli
Factors affecting carotid body response:
- Hypercapnia (high CO2) and acidosis (low pH) enhance response. = increased ventilation!!
- Hypocapnia dampens response.
- Opioids (e.g., morphine) can suppress carotid body function → risk of respiratory depression
When there is a change in arterial O2, if you see a change in ventilation as a result of that what is responsible
all due to the carotid body 100% of PaO2 sensing in humans
how much of the CO2 response is attributed to the carotid body
30%
SpO2
oxygen saturation
why does the carotid body respond to CO2
fast, well-adapted to respond to ABRUPT changes in PCO2

if the carotid bodies are only responsible for the response to 30% of the CO2 what detects the rest?
central chemoreceptors
ventilation response at a given PaO2 is enhanced by what 2 things
by increased PCO2 or H+
(breath hold, acidosis, increased metabolism)
so your carotid bodies will be more sesnitive to the partial pressure of oxygen if the partial pressure of co2 or H+ is high because that would be bad
Hypocapnia
Low CO2 levels decrease carotid body mediated ventilation response.
Neuronal Response Hypothesis (carotid body O2 sensing)
Glomus cells release neurotransmitters like neurons.

Membrane Hypothesis (carotid body O2 sensing)
Closure of O2-sensitive K⁺ channels causes depolarization.
- not maintained between species

Mitochondrial Hypothesis (carotid body O2 sensing)
Mitochondria function as oxygen sensors.

ATP/AMPK Hypothesis (carotid body O2 sensing)
Links oxygen sensing to cellular metabolism.

Carotid Bodies account for what percent of CO2 response
Account for less than 30% of CO2 response.
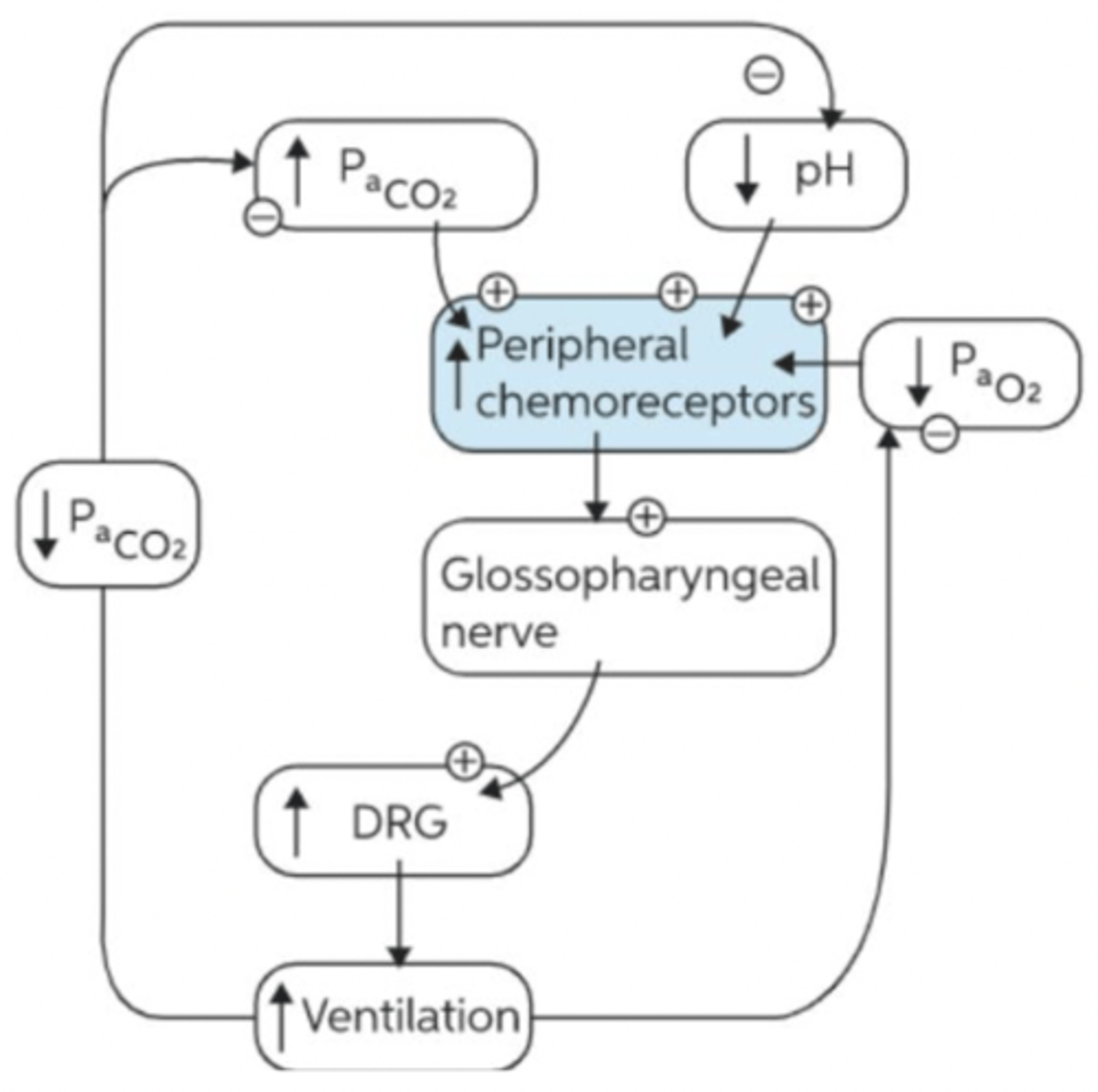
Central Chemoreceptors
Located in brainstem (medulla), account for ~70% CO2 response.
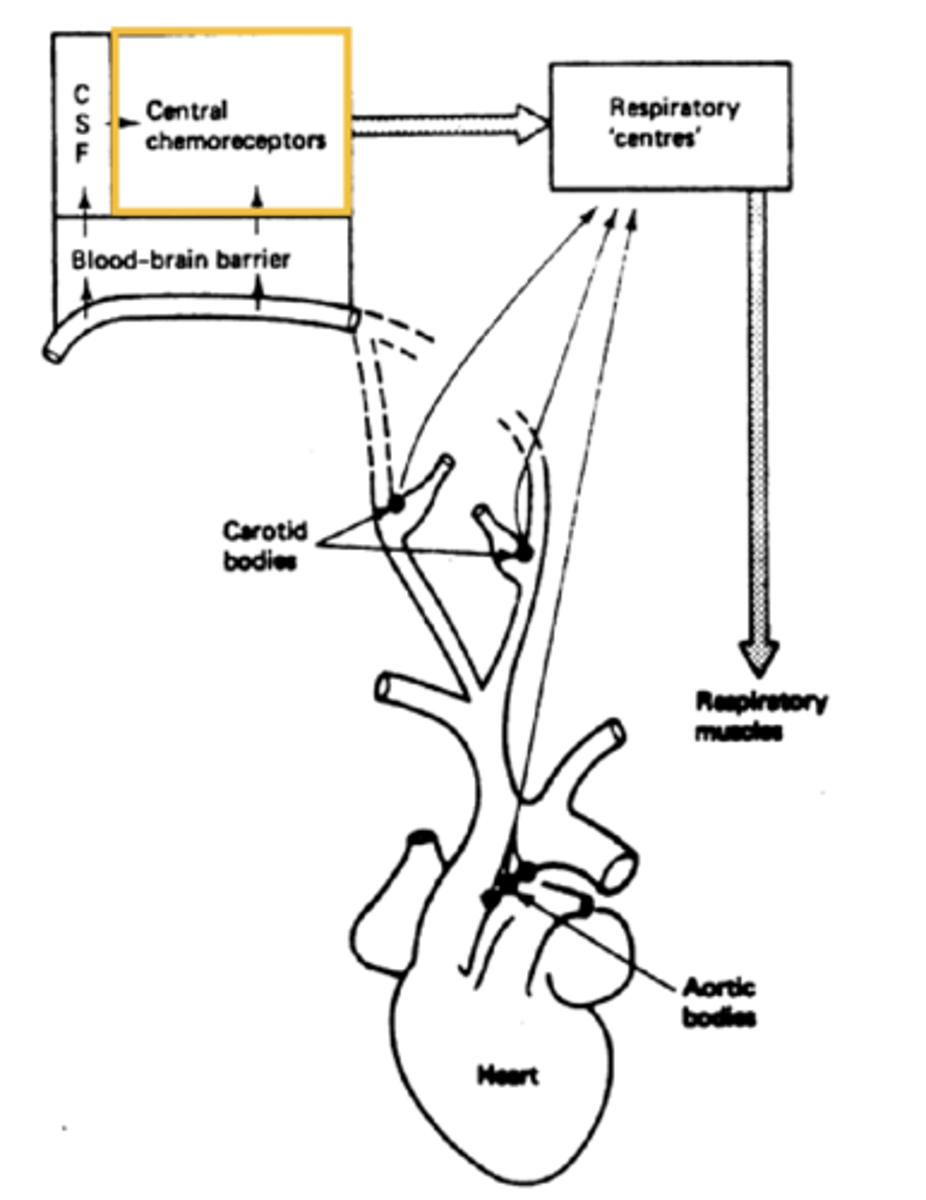
why can't the medullas chemoreceptors directly sample blood
cause of the blood brain barrier between the two
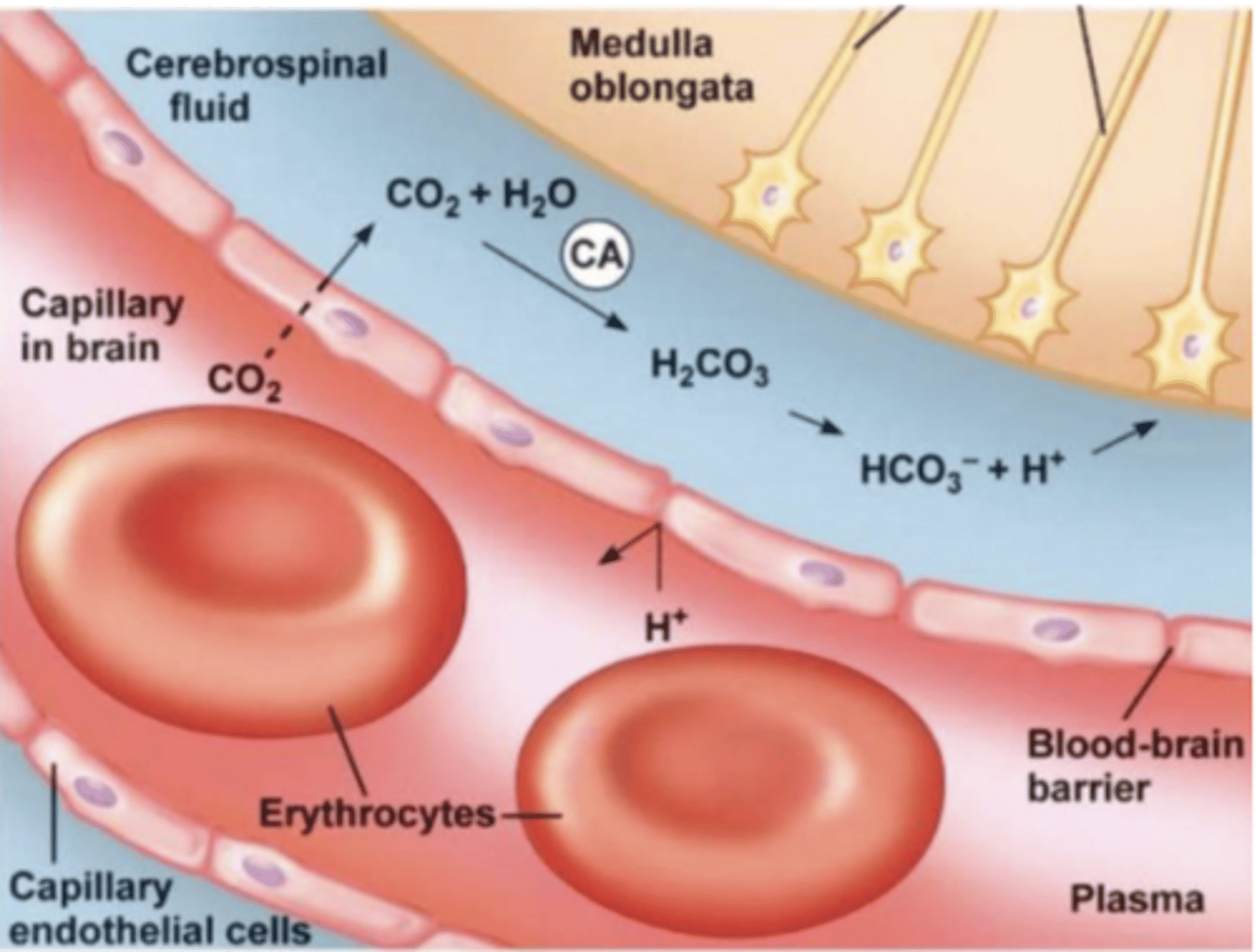
what is the primary driver of ventillation
CO2 is the primary driver of ventilation (not O2)!!
Central chemo receptors respond to what
H⁺ concentration, but H⁺ cannot cross the blood-brain barrier because its charged → so CO₂ crosses and binds with H20 and generates H⁺ via carbonic anhydrase.
Cerebrospinal Fluid (CSF)
Medium for central chemoreceptor exposure to CO2.
what is another important characteristic of CO2
it is a very potent vasodialator
Carbonic Anhydrase
Enzyme converting CO2 + H2O into HCO3- and H⁺.
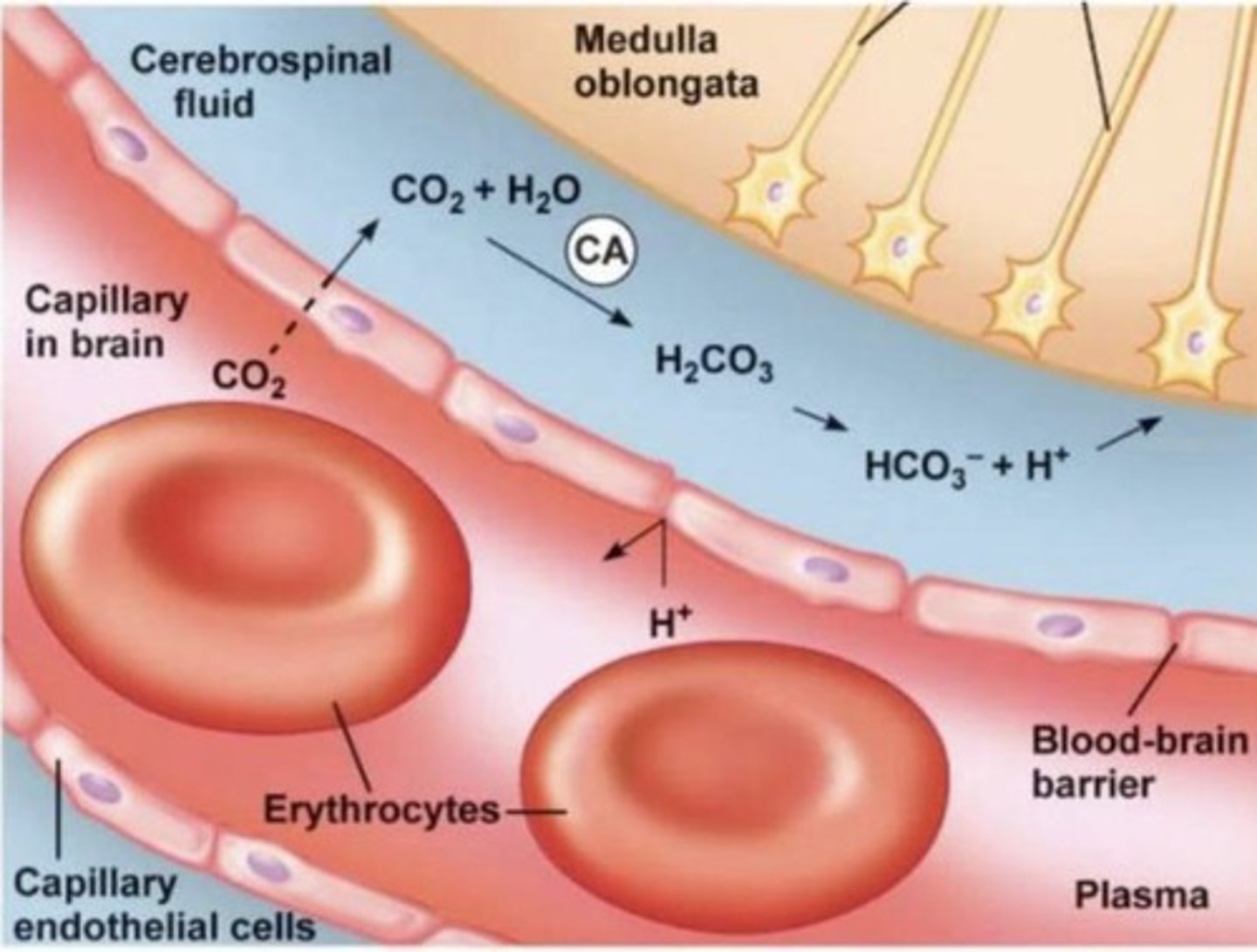
mechanism of H+ reaching chemoreceptors
1. CO2 diffuses across the blood-brain barrier (unlike H⁺, which cannot cross)
2. In CSF: CO2 + H2O → H2CO3 → H⁺ + HCO3⁻ (via carbonic anhydrase).
3. It's the H⁺ in CSF that stimulates central chemoreceptors.
What do upper airway afferents sense
flow via temp, pressure, upper airway muscle contraction
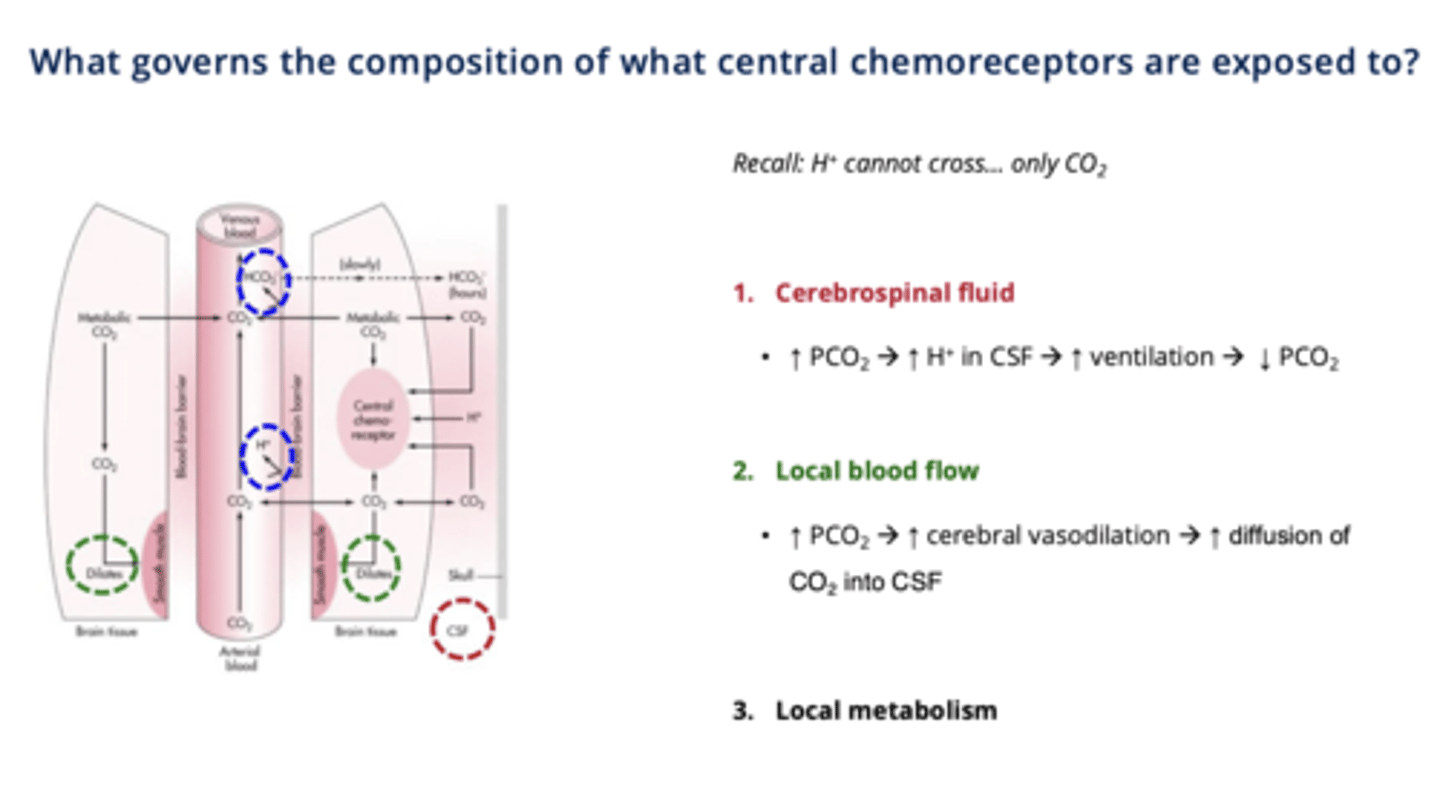
3 Key Factors Governing Central Chemoreceptor Exposure:
1. Cerebrospinal Fluid (CSF) Feedback loop:
↑ PCO2 → ↑ H⁺ in CSF → ↑ ventilation → ↓ PCO2 → restores balance.
- so CO2 would increase if the person was exercising or holding their breath
2. Local Blood Flow:
↑ PCO2→ ↑ cerebral vasodilation → ↑ blood flow → ↑ CO2 diffusion → ↑ H⁺ in CSF → ↑ ventilation.
- again will happen with breath hold or exercise
-Allows for more SA for diffusion to occur
3. Local Metabolism:
Influences acid-base balance in the CSF.
- Very minor
Alveolar Ventilation Equation
PaCO2 (arterial partial pressure of CO2) is proportional to CO2 production over ventilation.
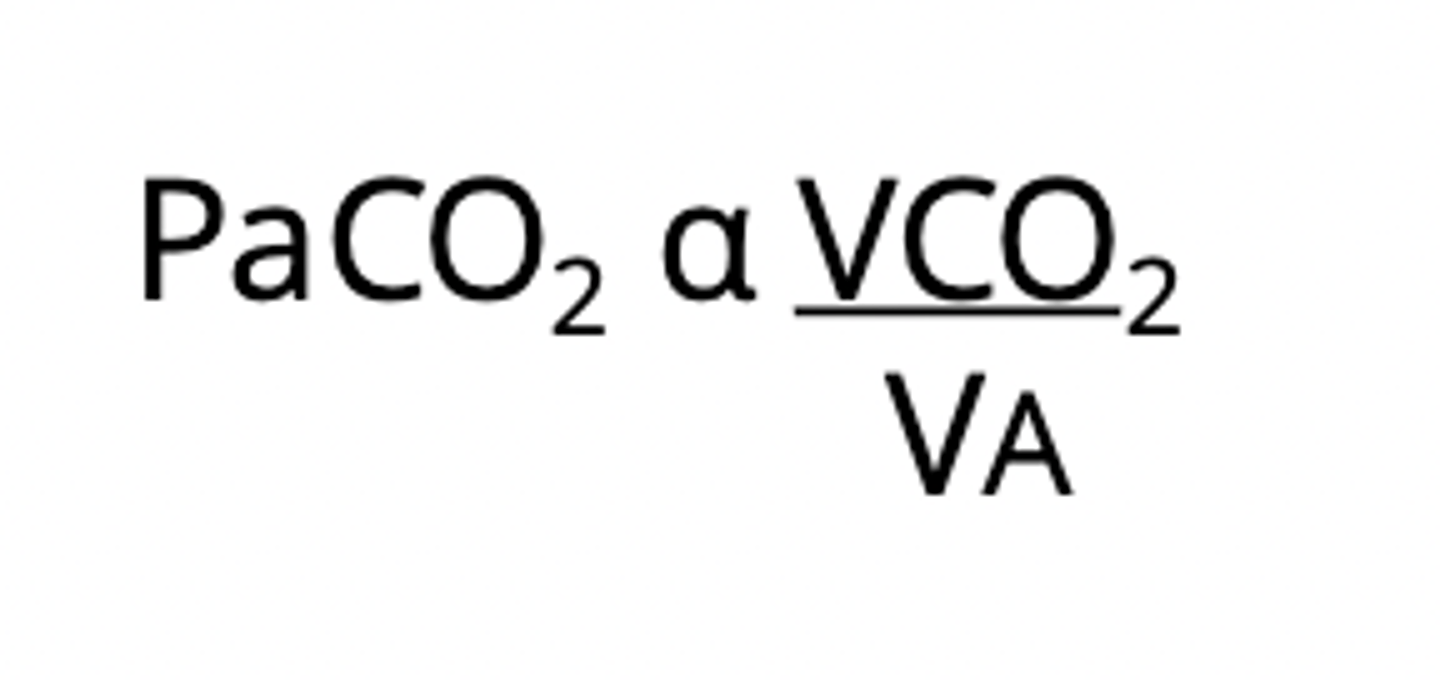
what is responsible for 100% of ventilatory response to PaO2 in humans
carotid bodies
If Vₐ increases what happens to PaCO2
it decreases since they are inversely proportional
If VCO2 increases what happens to PaCO2?
it increases since they are directly proportional
Threshold of CO2 Response
Minimum CO2 level for increased breathing (~40 mmHg).
- remember everyone is slightly different
- threshold decreases in a hypoxic enviroment
- threshold increases in a hyperoxic enviroment
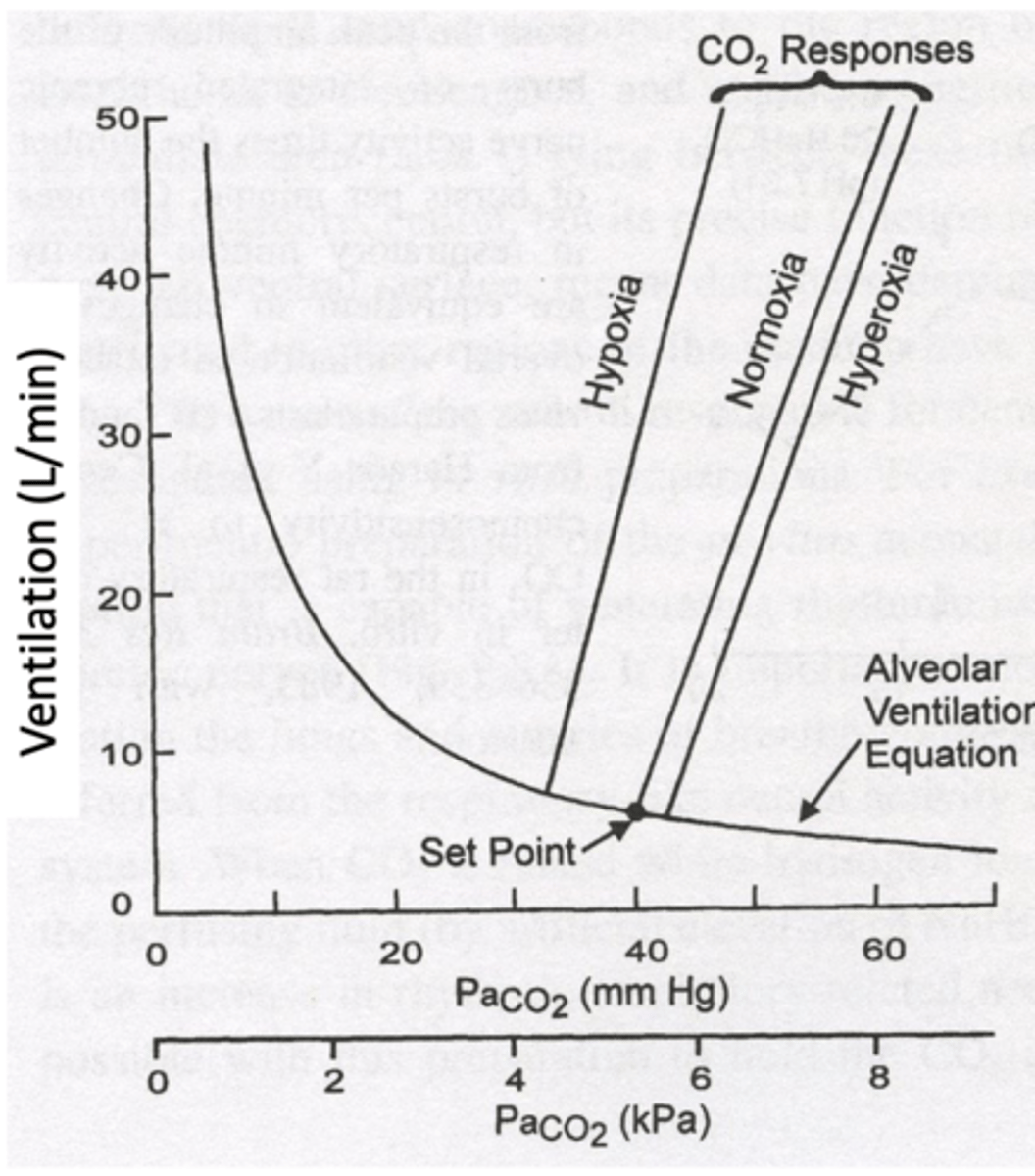
Sensitivity (slope) of CO2 Response
Ventilation increase per mmHg rise in CO2.
- more sensitive in hypoxic conditions
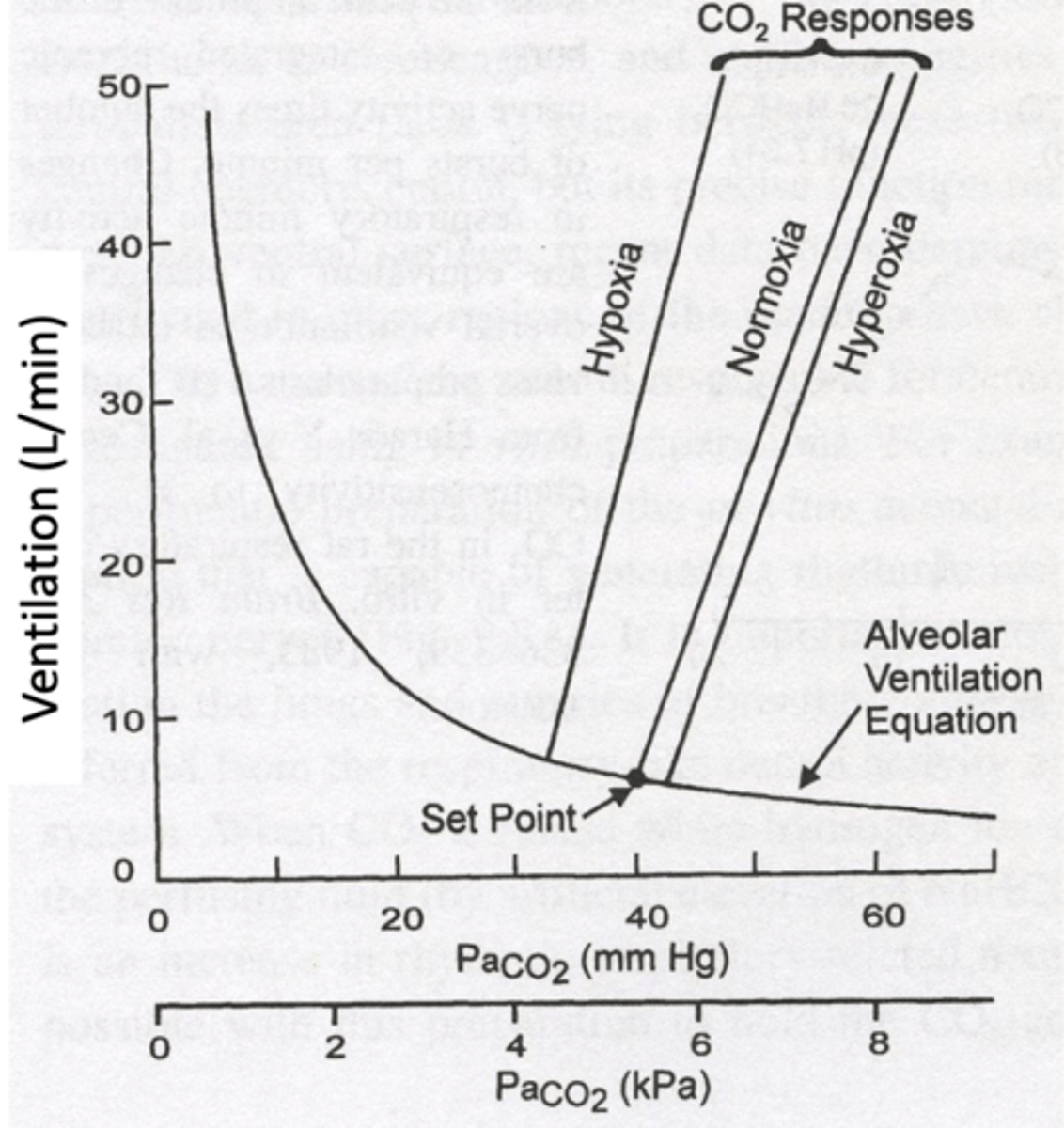
Hypoxia effects on CO2 sensing
Low oxygen decreases CO2 threshold and increases sensitivity (slope of response).
Hyperoxia on CO2 sensing
High oxygen levels blunt CO2 response. Higher threshold and less sensitive
Sleep stages affect on CO2 sensing
- Wakefulness: Highest sensitivity to CO2 changes. Lower threshold.
- Non-REM Sleep (3,4,2): Reduced ventilatory response (flattened sensitivity). Higher threshold.
- REM Sleep: Lowest sensitivity, highest threshold.
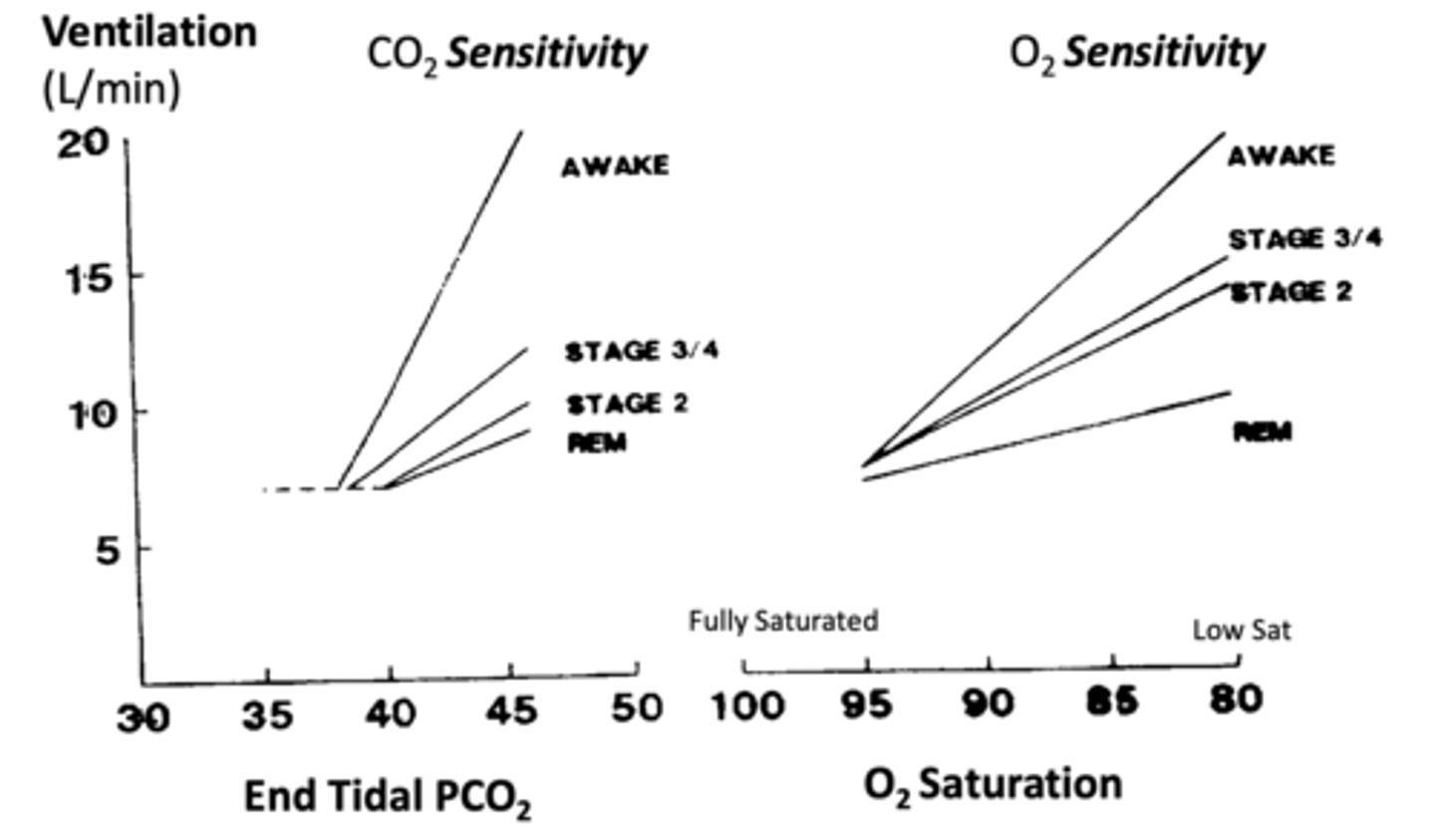
Sleep stages affect on O2 sensing
- Wakefulness: Highest sensitivity to O2 changes.
- Non-REM Sleep (3,4,2): Reduced ventilatory response (flattened sensitivity).
- REM Sleep: Lowest sensitivity.
how do opioids effect O2 and CO2 sensing
they actively suppress or dampen the sensitivity for both CO2 and O2.
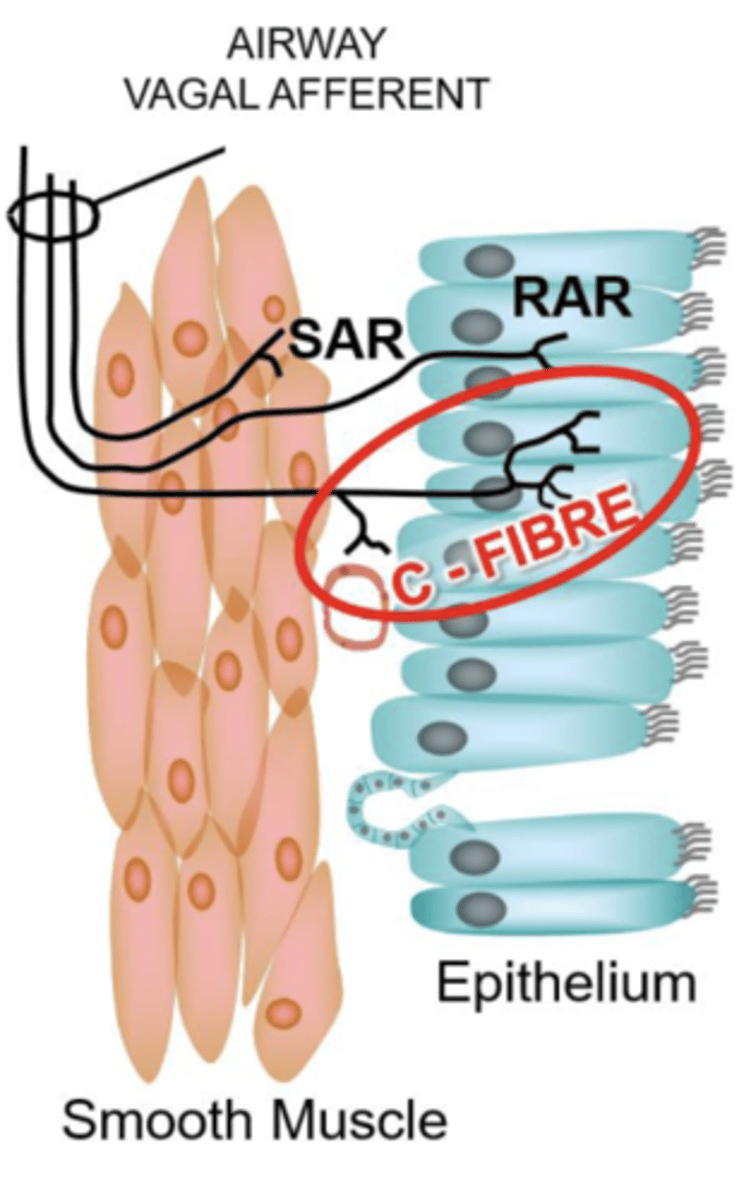
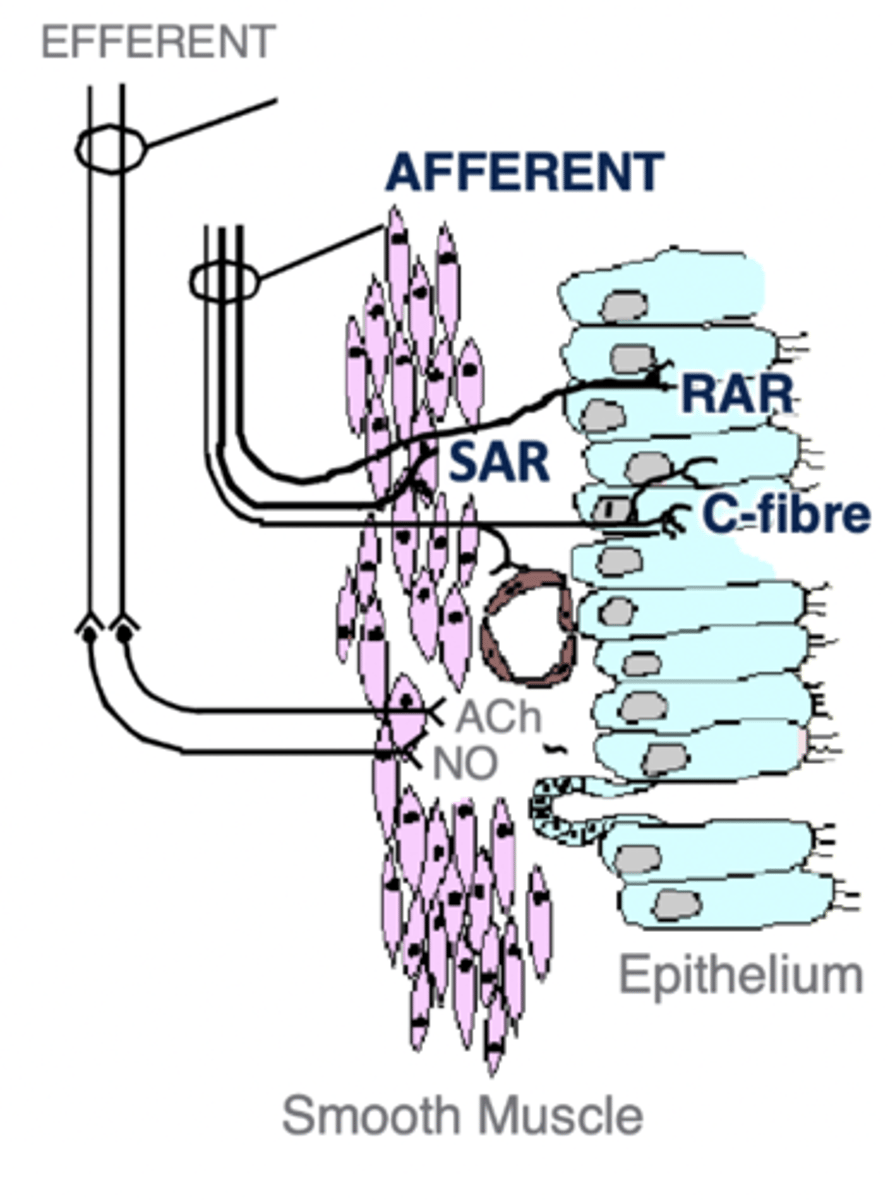
Intrapulmonary Afferents
Sensors detecting mechanical and chemical stimuli in airways.
- SAR
- RAR
- C-fibres
Intrapulmonary Afferents are classified by what 5 things
1. Myelination: Fast (myelinated) vs. slow (unmyelinated).
2. Location: Airway smooth muscle vs. airway epithelium.
3. Modulation by breathing: Active during inspiration, expiration, or both?
4. Adaptation: Do they fire continuously or adapt quickly?
5. Stimuli sensitivity: Physical stretch, irritants, histamine, osmolality, etc.
Slowly-Adapting Receptors (SARs)
Respond to lung inflation; do not adapt quickly.
- Myelinated
- located in airway smooth muscle
- Quiescent during expiration (doesn't do anything)
- Hering-breuer reflex
2 components to what it senses:
1. Static- senses Ptp (absolute volumes)
2. Dynamic - senses rate of inflation change
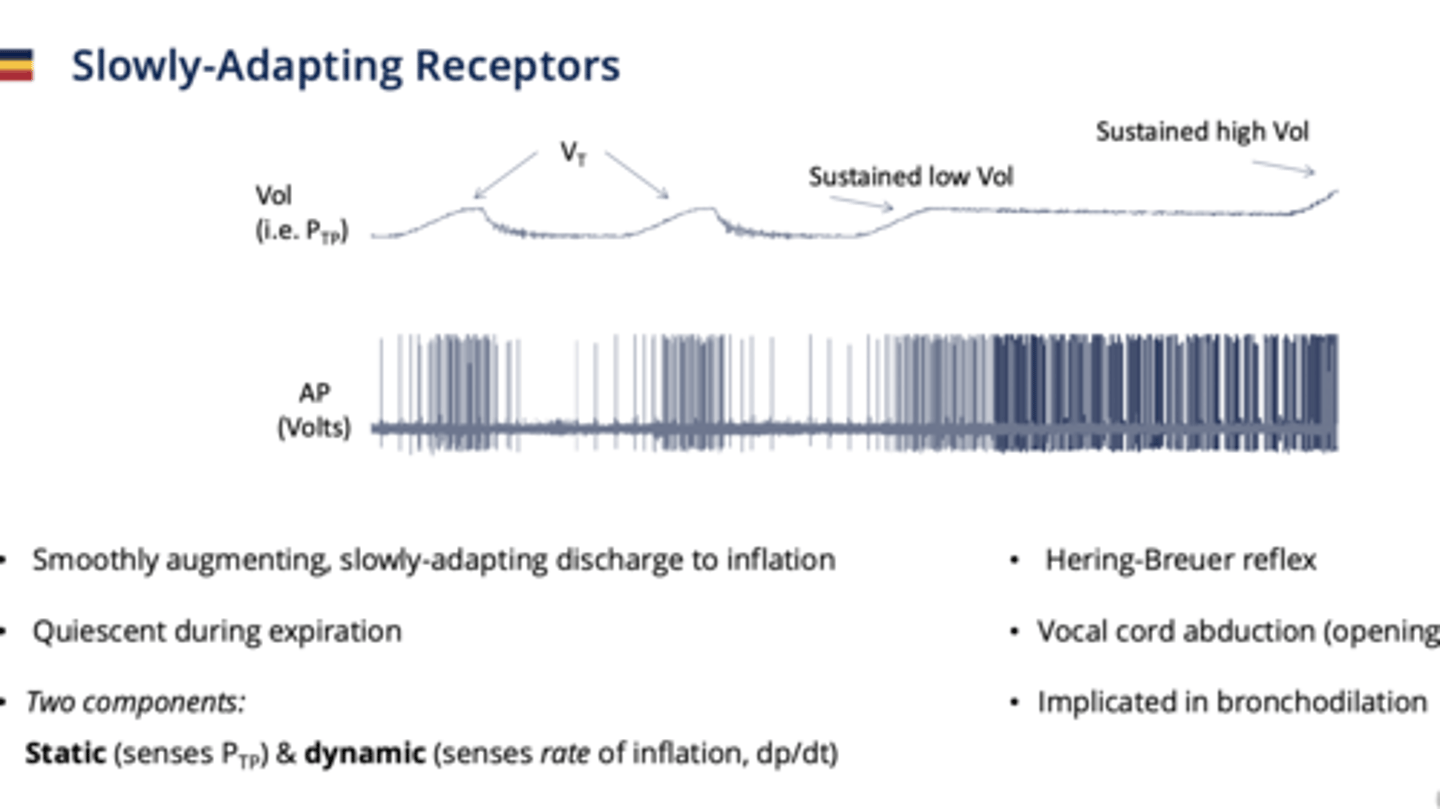
where do Slowly-Adapting Receptors (SARs) send their info
they send afferent info to the Nucleus Tractus Solitarius in the DRG
Hering-Breuer reflex
A reflex from the SARs present in babies which prevents them from taking too big of a breath
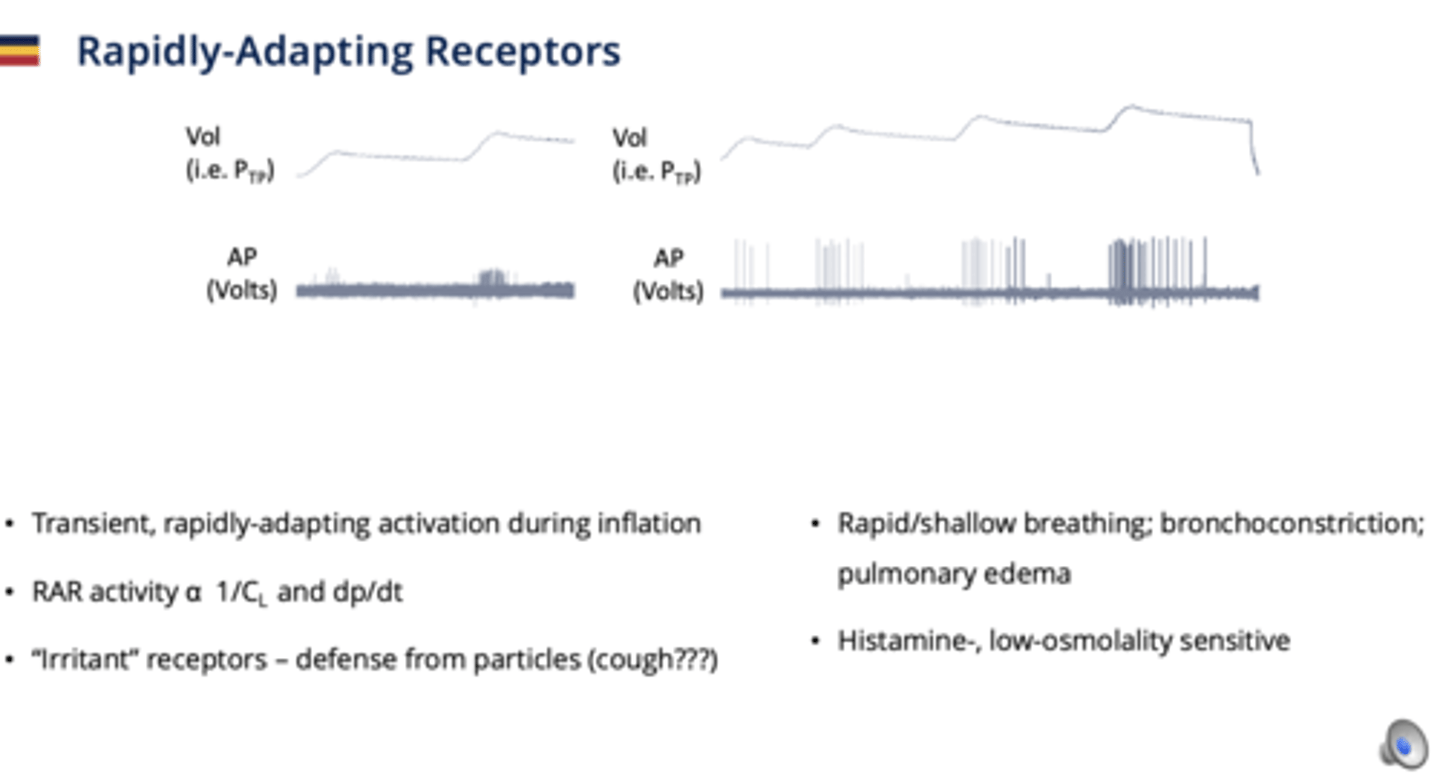
Rapidly-Adapting Receptors (RARs)
Respond to inflation changes.
- Myelinated
- Located in airway epithelium
- monitor 1/compliance and rate of change of transpulmonary pressure
- respond to: Mechanical stimuli (stretch, pressure), Chemical irritants, histamine, low osmolality, cold air.
- transient activation during inflation
**important in cough reflex**
what percentage of intrapulmonary afferents are myelinated
15% (only SAR and RAR)
85% are unmyelinated (C-fibres) !
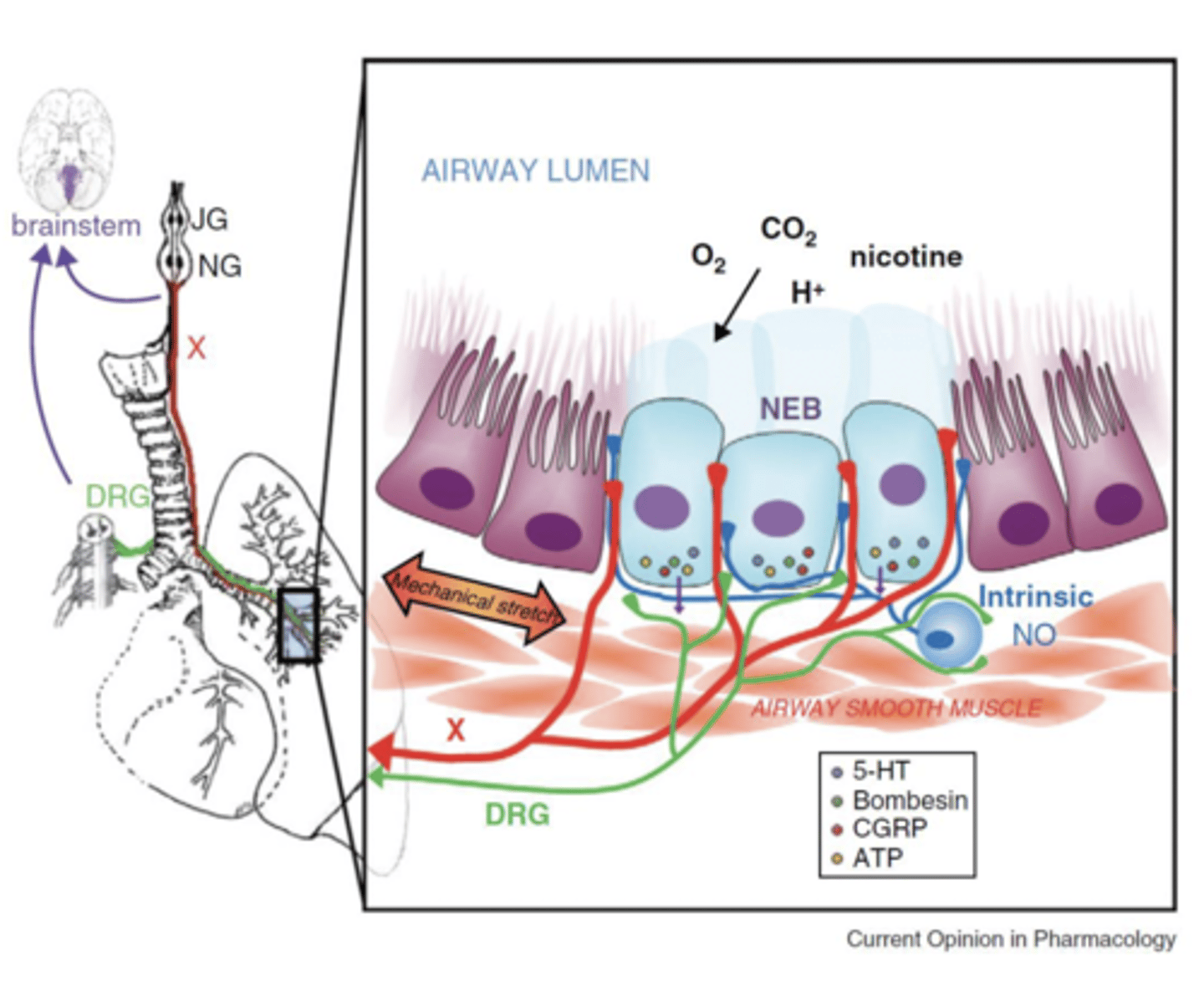
Neuroepithelial Bodies (NEBs)
Clusters of 5-15 Pulmonary Neuroendocrine Cells (PNECs), respond to stretch and potentially have a major O2 sensing role.
- located in airway bifurcations (branching points).
- Highly innervated, polymodal, respond to: MANY THINGS! hypoxia, mechanical stretch, nicotine and many other volatile compounds
** they are the first cell type in the lung to develop so thought of as a potential O2 sensor at birth **
what do the upper airways function as
a checkpoint system since they are densely innervated and come before the lower airways
where are the upper airway afferent cell bodies found
jugular ganglion and nodose ganglion
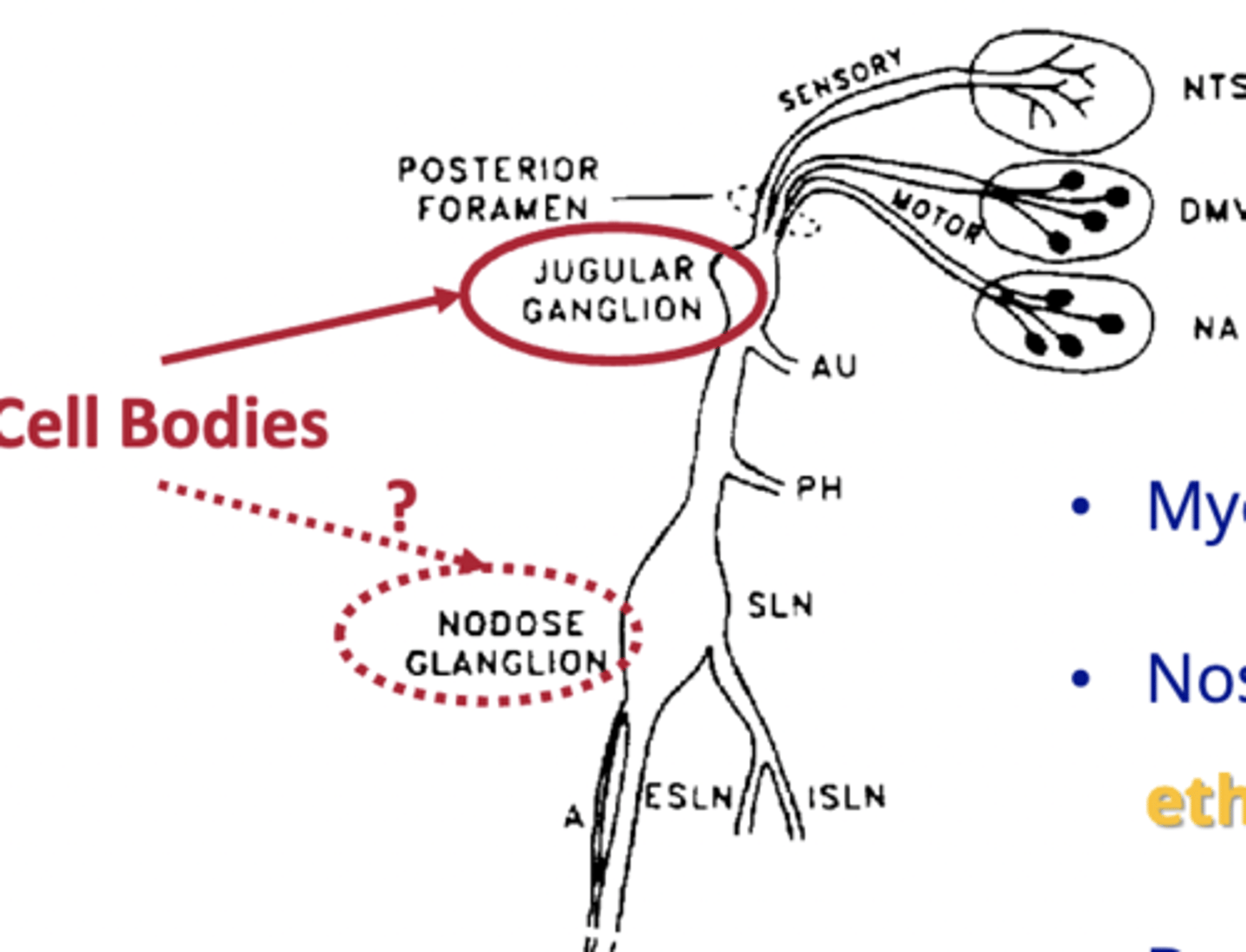
are the Upper Airway Afferents myelinated or no
they have myelinated & unmyelinated regions
Nose & larynx project to the medulla via what
ethmoidal nerve and the vagus nerve ( (SLN) & (RLN) from larynx.
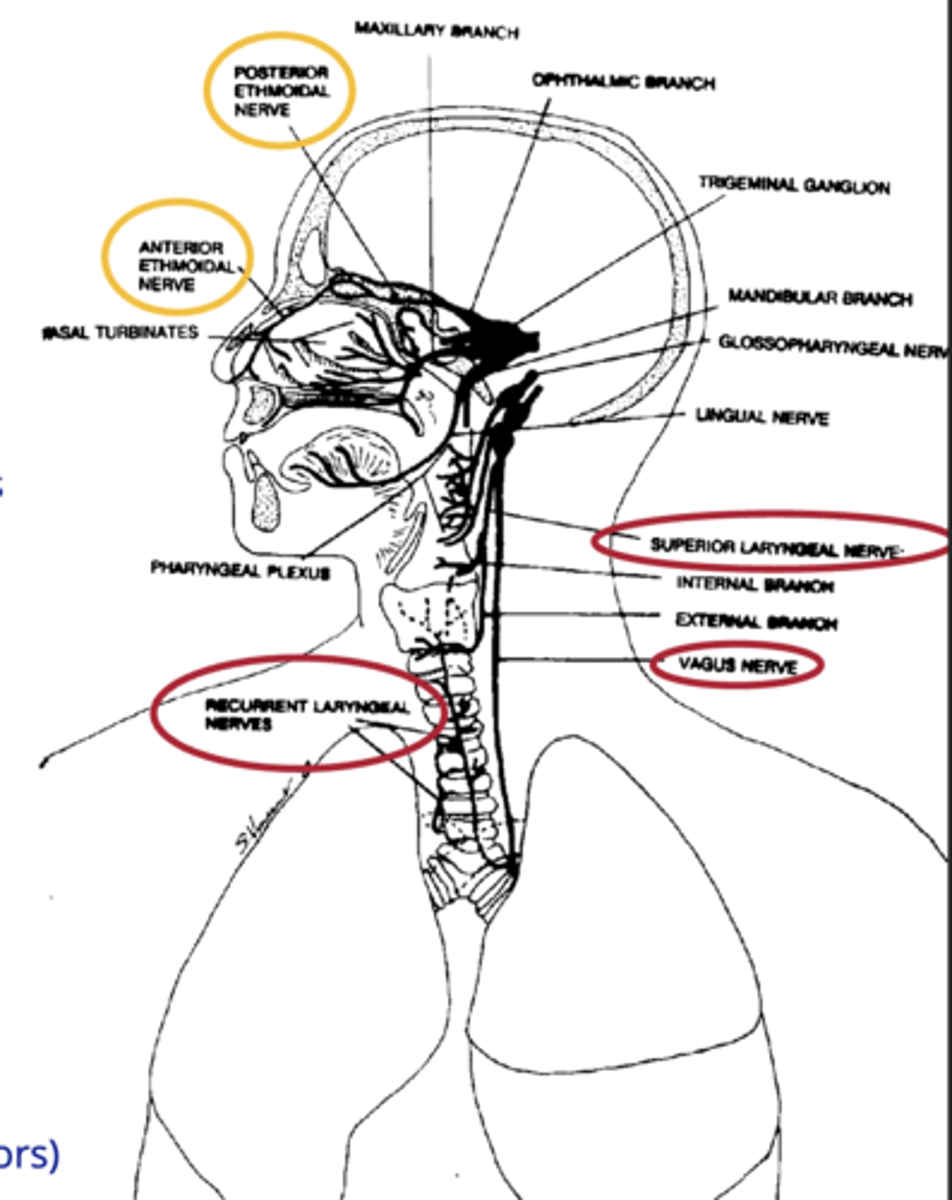
what do the upper airway afferents sense
- flow via temperature
- pressure (allows muscles to either contract or dilate to be more or less permissive of airflow)
- upper airway muscle contraction ("drive" receptors)
- smoke
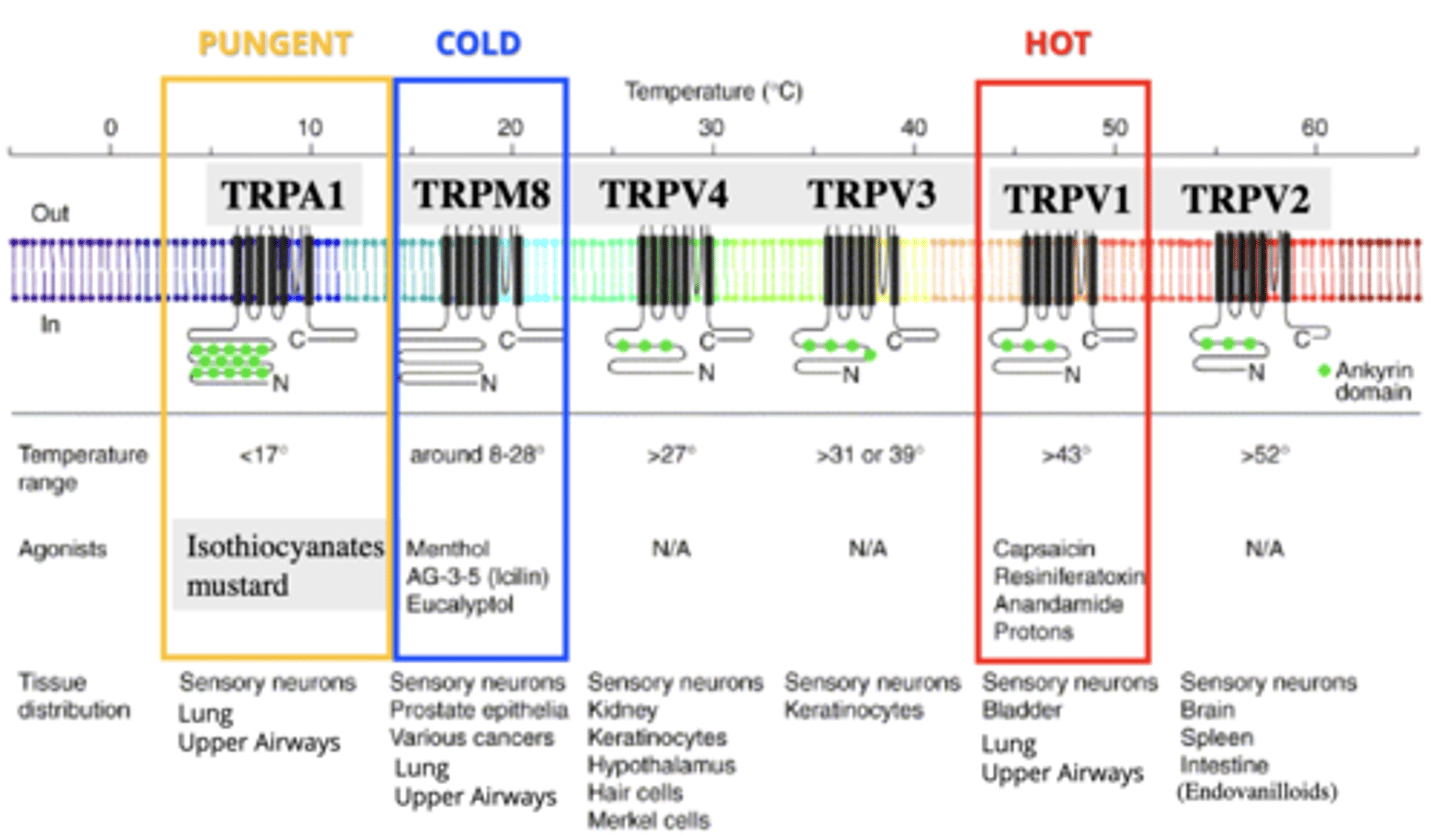
Transient Receptor Potential (TRP) Channels
Non-selective cation channels (with Ca2+ preference), respond to various stimuli.
TRP Channels can get activated by what 2 things
By either a specific temperature or an agonist specific to their channel
- remember that both have the same effect whether it was activated by one or the other
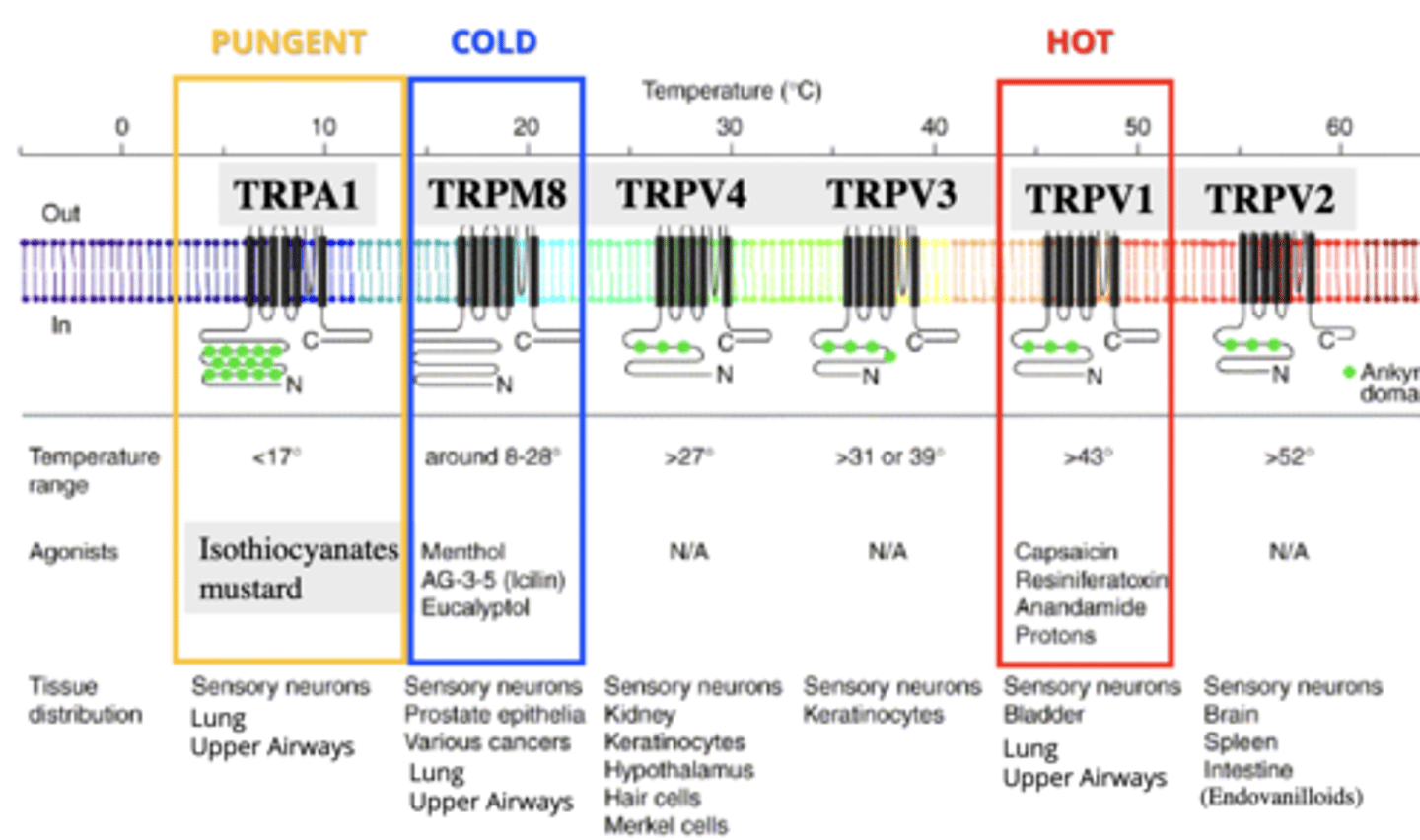
TRPM8
TRP receptor for cold stimuli, like menthol.
- 8-28 degrees
- located on sensory neurons, prostate epithelia, various cancers, lung, upper airways
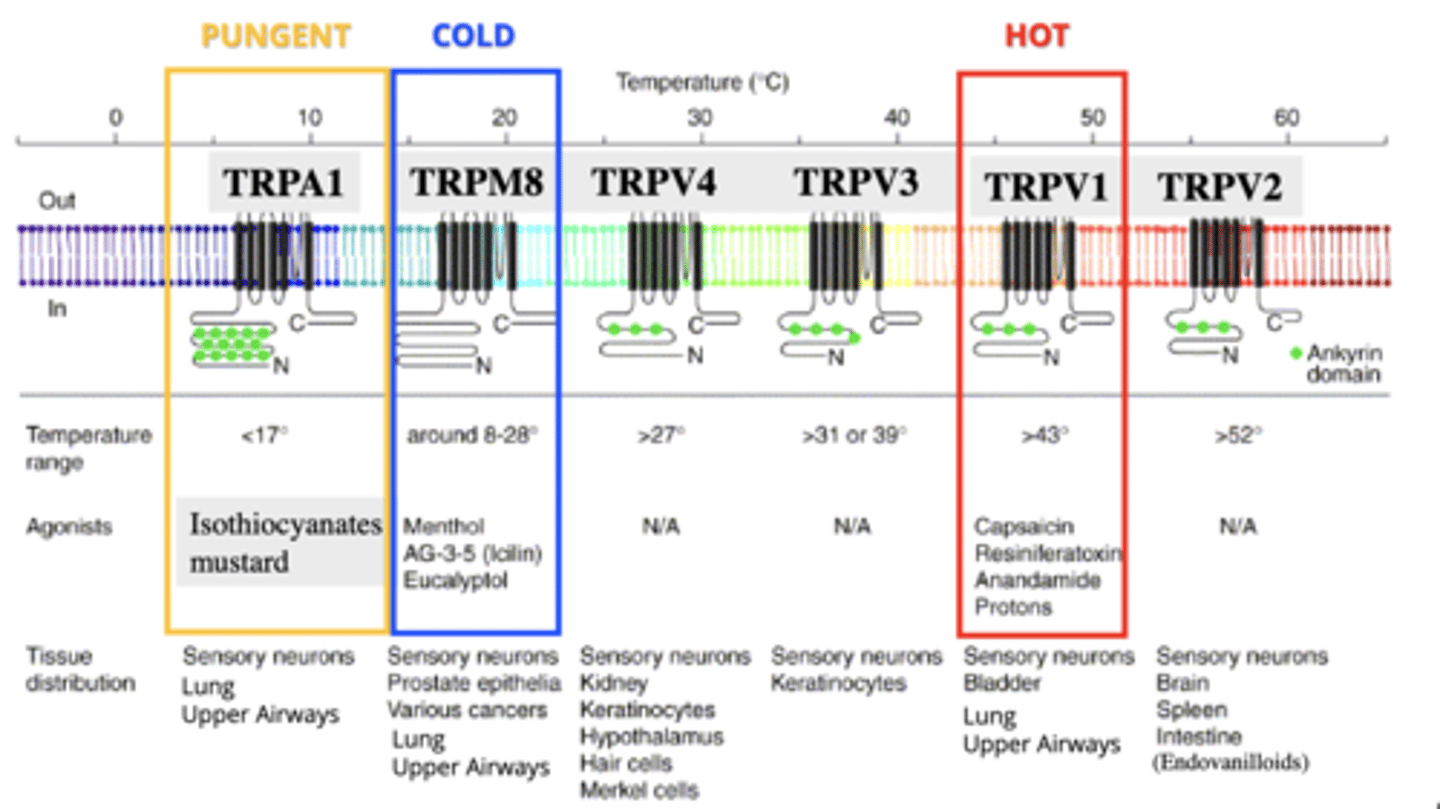
TRPA1
TRP receptor for pungent stimuli, like mustard, isothiocyanates.
- <17 degrees
- located on sensory neurons, lung, upper airways
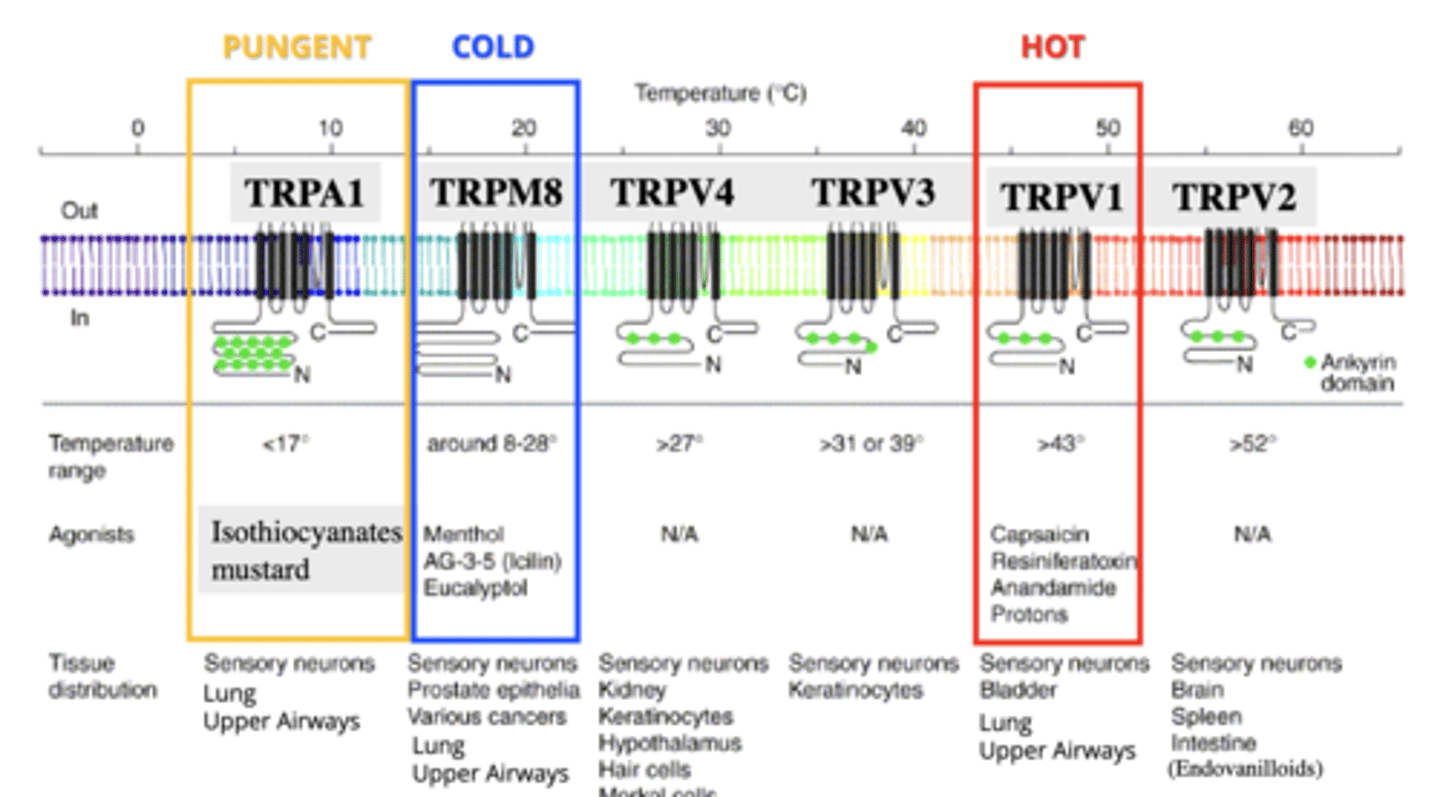
TRPV1
TRP receptor for heat and capsaicin.
- >43 degrees
- found on C fibres
- located on sensory neurons, bladder, lung, upper airways
C-fibres
respond to hyperinflation and inflammatory stimuli (autocoids, prostaglandins, histamine), lactic acid, edema.
- house TRPV1
- unmyelinated (so higher Ptp threshold decreased AP frequency)
- acid sensitive
(used to be called "j" juxtacapillary fibres)