chem bond angles
0.0(0)
Card Sorting
1/5
There's no tags or description
Looks like no tags are added yet.
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
6 Terms
1
New cards
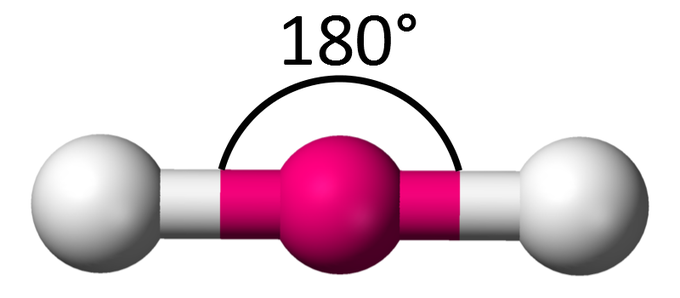
linear
sp hybridization
180°
2 bonded atoms, 0 lone pairs
2
New cards
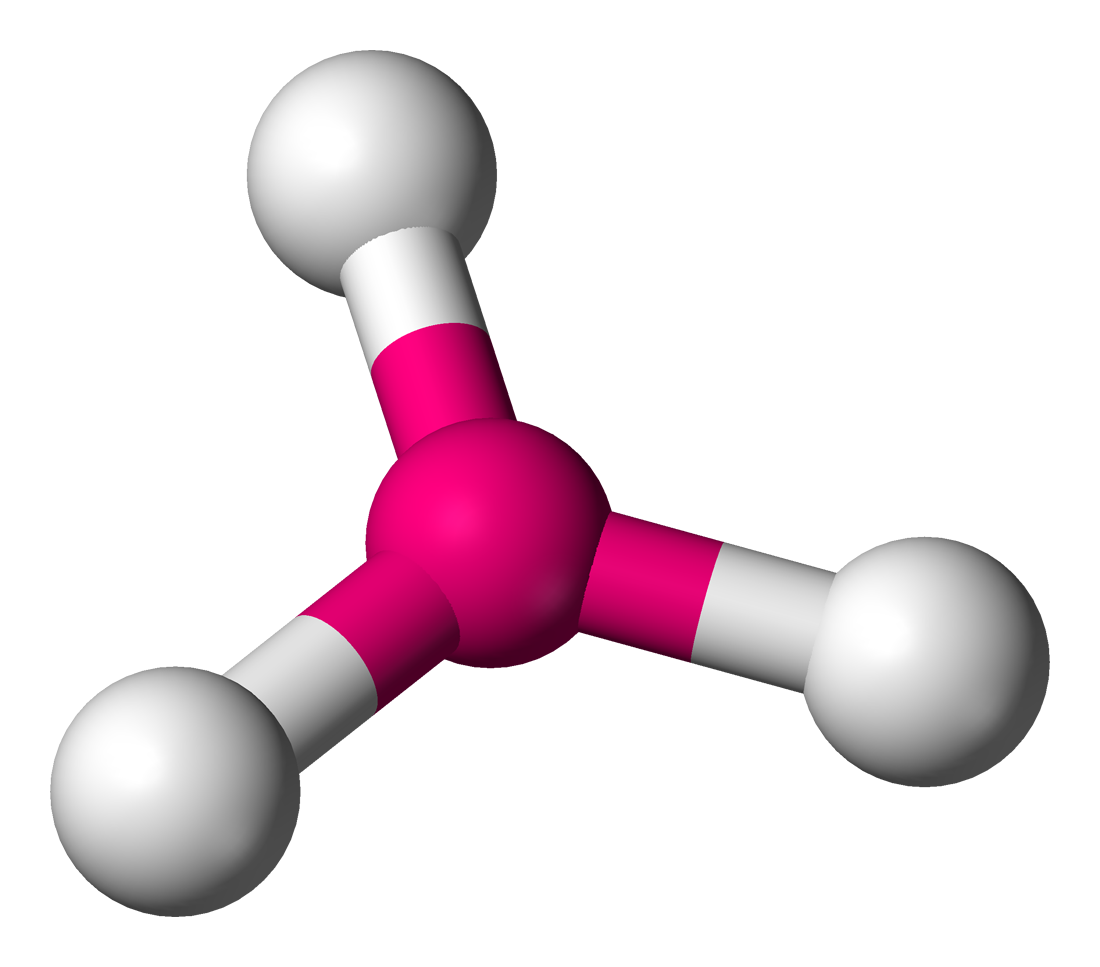
trigonal planar
sp² hybridization
120°
3 bonded atoms, 0 lone pairs
3
New cards
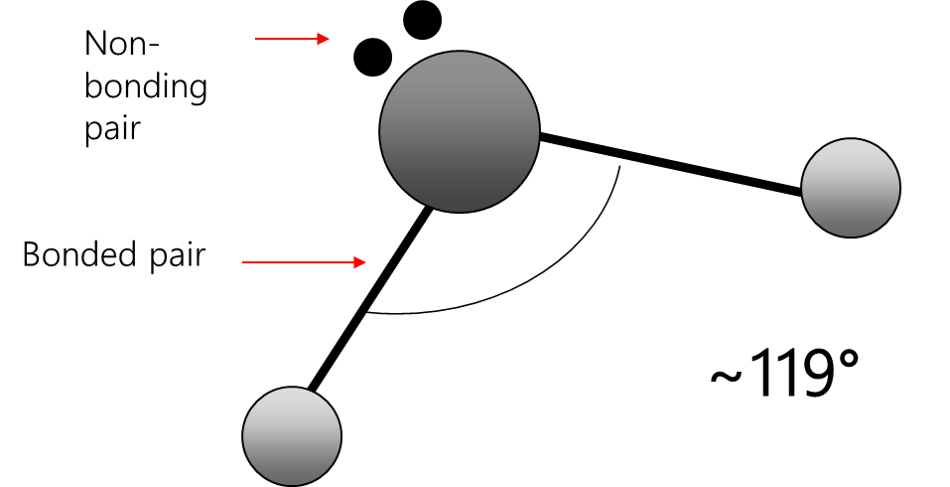
bent (1)
sp² hybridization
119°
2 bonded atoms, 1 lone pair
4
New cards
tetrahedral
sp³ hybridization
109.5°
4 bonded atoms, 0 lone pairs
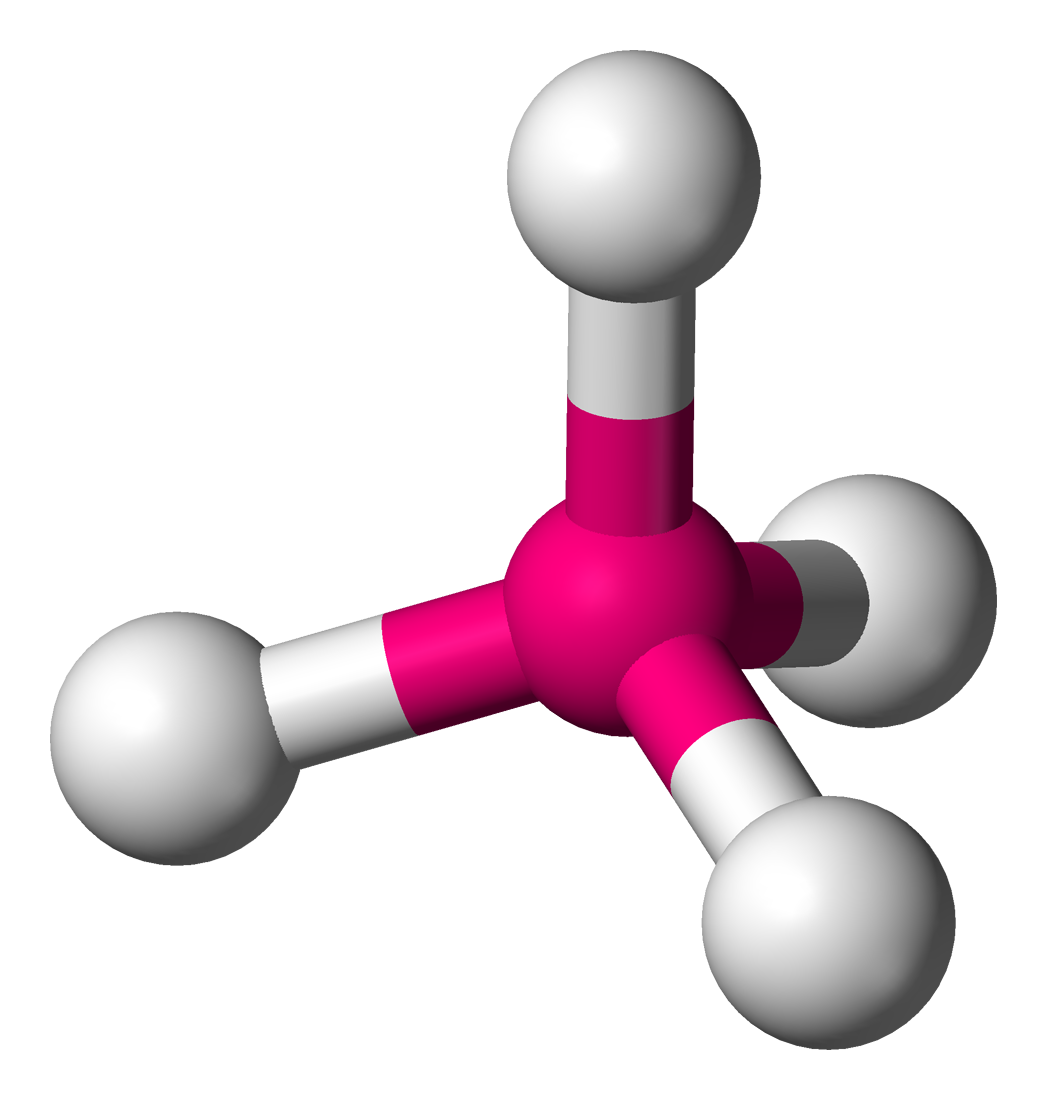
5
New cards
trigonal pyramidal
sp³ hybridization
107°
3 bonded atoms, 1 lone pair
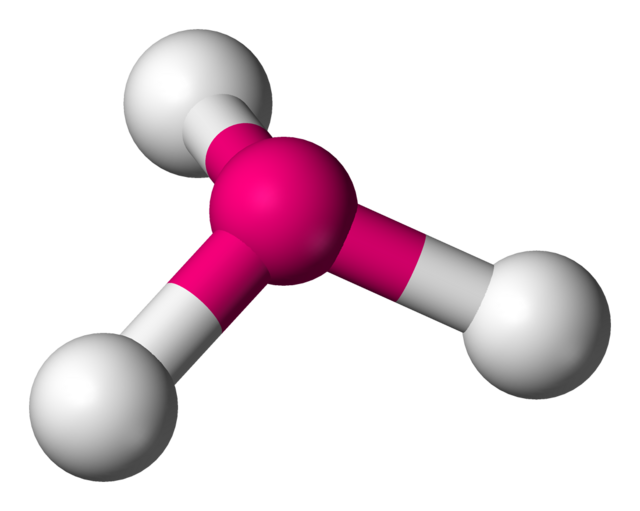
6
New cards
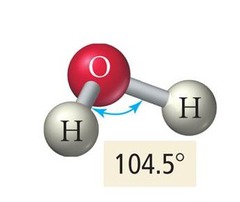
bent (2)
sp³ hybridization
104.5°
2 bonded atoms, 2 lone pairs