Energetics
1/33
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
Enthalpy of formation (ΔHf⦵ )
Enthalpy change when one mole of a substance is formed from its constituent elements with all substances in their standard states: Exothermic (-)
e.g. Na2O(s)
2 Na(s) + ½ O2(g) → Na2O(s)
Enthalpy of combustion (ΔHc⦵ )
Enthalpy change when one mole of a substance undergoes complete combustion in oxygen with all substances in their standard states: Exothermic (-)
e.g. hydrogen
H2(g) + ½ O2(g) → H2O(l)
Enthalpy of neutralisation (ΔHneut⦵ )
Enthalpy change when 1 mole of water is formed in a reaction between an acid and alkali under standard conditions: Exothermic (-)
e.g. H2SO4 + NaOH
½ H2SO4(aq) + NaOH(aq) → ½ Na2SO4(aq) + H2O(l)
(First) Ionisation enthalpy (ΔHIE⦵ )
Enthalpy change when each atom in one mole of gaseous atoms loses one electron to form one mole of gaseous 1+ ions: Endothermic (+)
e.g. magnesium
Mg(g) → Mg+(g) + e-
(Second) Ionisation enthalpy (ΔHIE⦵ )
Enthalpy change when each ion in one mole of gaseous 1+ ions loses one electron to form one mole of gaseous 2+ ions: Endothermic (+)
e.g. magnesium
Mg+(g) → Mg2+(g) + e-
(First) Electron affinity (ΔHEA⦵ )
Enthalpy change when each atom in one mole of gaseous atoms gains one electron to form one mole of gaseous 1- ions: Exothermic (-) for many non metals
e.g. oxygen
O(g) + e- → O-(g)
(Second) Electron affinity (ΔHEA⦵ )
Enthalpy change when each ion in one mole of gaseous 1- ions gains one electron to form one mole of gaseous 2- ions: Endothermic (+) as adding negative electron to negative ion
e.g. oxygen
O- (g) + e- → O2-(g)
Enthalpy of atomisation (ΔHatm⦵ )
Enthalpy change when one mole of gaseous atoms is produced from an element in its standard state: Endothermic (+)
e.g. iodine
½ I2(s) → I(g)
Hydration enthalpy (ΔHhyd⦵ )
Enthalpy change when one mole of gaseous ions become hydrated (dissolved in water): Exothermic (-)
e.g. magnesium ions
Mg2+(g) → Mg2+(aq)
Enthalpy of solution (ΔHsol⦵ )
Enthalpy change when one mole of an ionic solid dissolves in an amount of water large enough so that the dissolved ions are separated and do not interact with each other: Varies
e.g. magnesium chloride
MgCl2(s) → Mg2+(aq) + 2 Cl-(aq)
Bond dissociation enthalpy (ΔHBDE⦵ )
Enthalpy change when one mole of covalent bonds is broken in the gaseous state: Endothermic (+)
e.g. I-I bond
I2(g) → 2 I(g)
Lattice enthalpy of formation (ΔHLE⦵ )
Enthalpy change when one mole of a solid ionic compound is formed from its constituent gaseous ions: Exothermic (-)
e.g. magnesium chloride
Mg2+(g) + 2 Cl- (g) → MgCl2(s)
Lattice enthalpy of dissociation (ΔHLE⦵ )
Enthalpy change when one mole of a solid ionic compound is broken up into its constituent gaseous ions: Endothermic (+)
e.g. magnesium chloride
MgCl2(s) → Mg2+(g) + 2 Cl- (g)
Enthalpy of vaporisation (ΔHvap⦵ )
Enthalpy change when one mole of a liquid is turned into a gas: Endothermic (+)
e.g. H2O(l)
H2O(l) → H2O(g)
Enthalpy of fusion (ΔHfus⦵ )
Enthalpy change when one mole of a solid is turned into a liquid: Endothermic (+)
e.g. Mg(s)
Mg(s) → Mg(l)
Mean bond enthalpy
Energy needed to break one mole of covalent bonds in the gaseous state
Averaged over a range of different compounds/molecules
State the assumptions made during calculations in calorimetry. (3 marks)
All heat released from the reaction was absorbed by the solution
Reaction goes to completion
Specific heat capacity of solution is 4.18 J-1 g-1
Mass of solid is ignored
Solution has a density of 1 g cm-3
State the temperature at which the standard entropy of aluminium is 0 J K–1 mol–1 (1 mark)
0 K
The enthalpy of combustion determined experimentally is less exothermic than that calculated using enthalpies of formation.
Give four possible reasons for this. (4 marks)
Heat escaped to surroundings
Incomplete combustion of (substance)
Evaporation of (substance) between weighing and setting light to it
Didn’t stir to make temperature the same throughout
The student considered using a glass beaker on a tripod and gauze instead of the clamped copper calorimeter. Suggest two disadvantages of using a glass beaker on a tripod and gauze. (2 marks)
Glass is a poorer conductor than copper
Tripod and gauze would reduce heat transfer
Tripod and gauze would have a fixed height above the flame
Suggest why chemists usually aim to design production methods
with fewer steps
with a high percentage atom economy
(2 marks)
Less energy used OR Better yield
Less waste OR Less pollution
Define the term enthalpy change
Heat energy change at a constant pressure
Suggest how the students’ method, and the analysis of the results, could be improved in order to determine a more accurate value for the enthalpy of reaction. Justify your suggestions.
Do not refer to the precision of the measuring equipment. Do not change the amounts or the concentration of the chemicals. (6 marks)
Stage 1 : Improved insulation
Insulate the beaker or use a polystyrene cup or a lid
To reduce heat loss
Stage 2 : Improved temperature recording
Record the temperature for a suitable time before adding the metal
To establish an accurate initial temperature
Record temperature values at regular time intervals
To plot the temperature results against time on a graph
Stage 3 : Improved analysis of results
Extrapolate the cooling back to the point of addition
To establish a (theoretical) maximum temperature OR temperature change
Suggest and explain another change to the experiment that would decrease the percentage uncertainty in the use of the same thermometer. (2 marks)
Increase magnitude of temperature change
by increasing the concentration of the acid/alkali
The enthalpy of hydration of Ca2+(g) is –1650 kJ mol–1 Suggest why this value is less exothermic than that of Mg2+(g) (2 marks)
Ca2+ (ion) bigger/lower charge to size ratio (than Mg2+)
weaker attraction/bond to (Oδ- in) water
State why the heat change calculated from the bomb calorimeter experiment is not an enthalpy change. (1 mark)
pressure not constant in bomb calorimeter
Explain why the enthalpy of hydration becomes less exothermic from Li+ to K+. (2 marks)
(From Li+ to K+ ) size (of ion) increases
Electrostatic attraction between metal ion and Oδ– of water decreases
Explain why the enthalpy of lattice dissociation for sodium oxide is greater than the enthalpy of lattice dissociation for sodium chloride. (2 marks)
Oxide ions have higher negative charge OR smaller size OR higher charge density/higher charge/size ratio than chloride ions
stronger attraction between O2- and Na+ ions
Give a reason why data books do not contain a value for the enthalpy of solution of sodium oxide. (1 mark)
It reacts with water OR It reacts to form a solution of NaOH
Explain why the enthalpy of hydration of fluoride ions is more negative than the enthalpy of hydration of chloride ions. (2 marks)
Fluoride ions/F− ions are smaller OR F− has a higher charge density
Stronger attraction of fluoride ion to δ+ on H/ electron deficient H in water
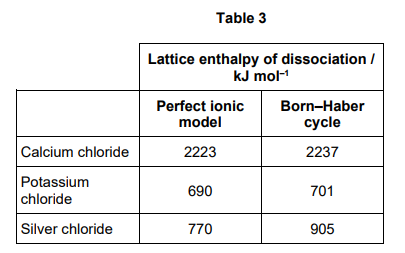
Table 3 shows lattice enthalpies based on a perfect ionic model and lattice enthalpies from Born–Haber cycles for three metal chlorides.
Discuss the values in Table 3. In your answer you should
compare the three values based on a perfect ionic model
compare the values based on a perfect ionic model to the values from a Born–Haber cycle for each compound.
(6 marks)
Stage 1: comparing values from perfect ionic model
Value for CaCl2 is larger OR Values for KCl and AgCl are similar OR Values for CaCl2 > AgCl > KCl
Ca2+ has a larger charge/ is a smaller ion OR Ag+ and K+ have smaller charge or larger ions
CaCl2 has stronger ionic bonds or stronger attraction between + and - ions (Ca2+and Cl- ) OR AgCl and KCl have weaker ionic bonds or weaker attraction between + and - ions (Ag+/ K+ and Cl- )
Stage 2: similarities in the perfect ionic model and Born-Haber cycle
CaCl2 has similar values (between the perfect ionic model and Born-Haber cycle)
KCl has similar values (between the perfect ionic model and Born-Haber cycle)
CaCl2 and KCl have (almost) perfect ionic bonding or + ions are point charges/(perfectly) spherical
Stage 3 difference in the perfect ionic model and Born-Haber cycle values
AgCl has larger difference in values (between the perfect ionic model and Born-Haber cycle)
AgCl contains (some) covalent character
Ag+ more polarising/distorts electron cloud more
Suggest a change to the student’s method, using the same apparatus, that would reduce the percentage uncertainty in the temperature change. Give a reason for your answer. (2 marks)
use a larger mass/amount of NH4NO3 / solid
so temperature change/decrease is greater OR final temperature is lower
A calculation of the enthalpy of lattice formation of silver iodide based on a perfect ionic model gives a smaller numerical value than the value calculated in Question 1.2 Explain this difference. [2 marks]
AgI contains covalent character
Forces/bonds (holding the lattice together) are stronger
Anhydrous magnesium chloride, MgCl2, can absorb water to form the hydrated salt
MgCl2.4H2O MgCl2(s) + 4H2O(l) → MgCl2.4H2O(s)
Suggest one reason why the enthalpy change for this reaction cannot be determined directly by calorimetry. (1 mark)
Not possible to prevent some dissolving