Chemistry: Kinetics and Equilibrium (7)
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
23 Terms
Particles need ________ and _________ to react
Energy and orientation
Increased collisions indicate _________ reaction rate
Increased
What effect does increased concentration have on reaction rate?
This means more particles will collide causing more collisions and increasing reaction rate.
What effect does increased temperature have on reaction rate?
This increases kinetic energy of particles and increases collisions and REACTION RATES.
What effect does increased surface area have on reaction rate?
This causes more particles to be exposed to reactions and INCREASES reaction rate.
Which of these will react faster: Powdered Mg or a pellet of Mg?
Powdered Mg
How does the nature of reactants impact reaction rate?
Ionic solutions will have faster reaction rates than molecular/covalent solutions.
What effect do catalysts have on reaction rates?
They speed them up by lowering the activation energy need for the reaction
Dynamic Equilibrium has _____ rates of forward and reverse reactions.
Equal
Dynamic Equilibrium has _____ concentrations
Constant
What are examples of physical equilibrium?
Closed systems (stoppered bottles), solutions, and phase equilibrium
According to Le Chatelier’s Principles, increase _____ ______
Shift away
According to Le Chatelier's Principle, decreases __________ __________
Shift Away
Pressure changes only affect which state of matter?
Gas
Increases in pressure will shift towards…?
side with lower amount moles of gas and vice versa
Anything in the direction of the shift will?
Increase
Anything opposite the direction of a shift will?
Decrease
What are reasons that reactions go into completion (→)?
Precipitates and Open Systems
∆H Formula
P.E. Products - P.E. Reactants
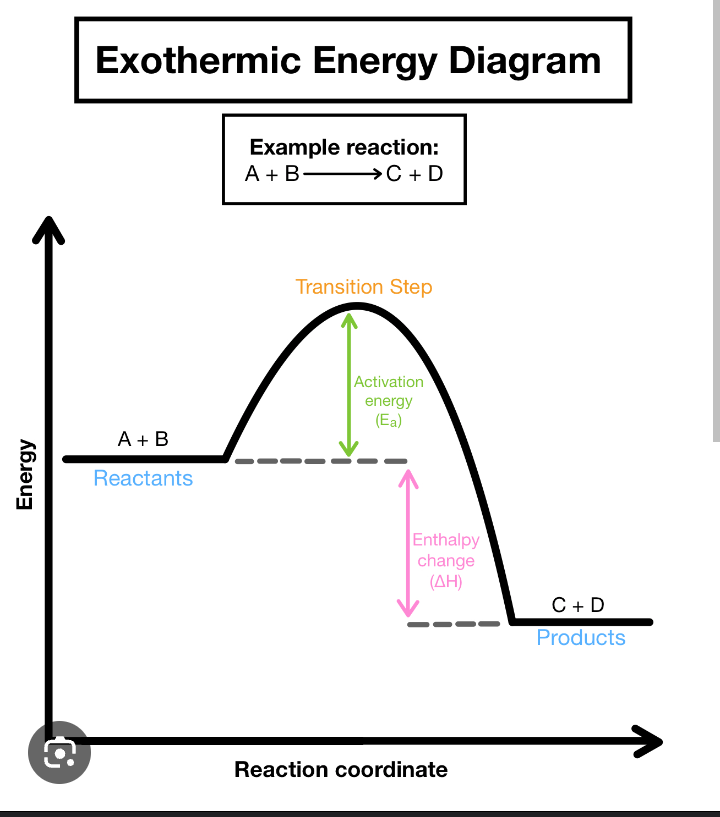
Exothermic
WARM: Releases Energy
∆H is Negative and on the Right
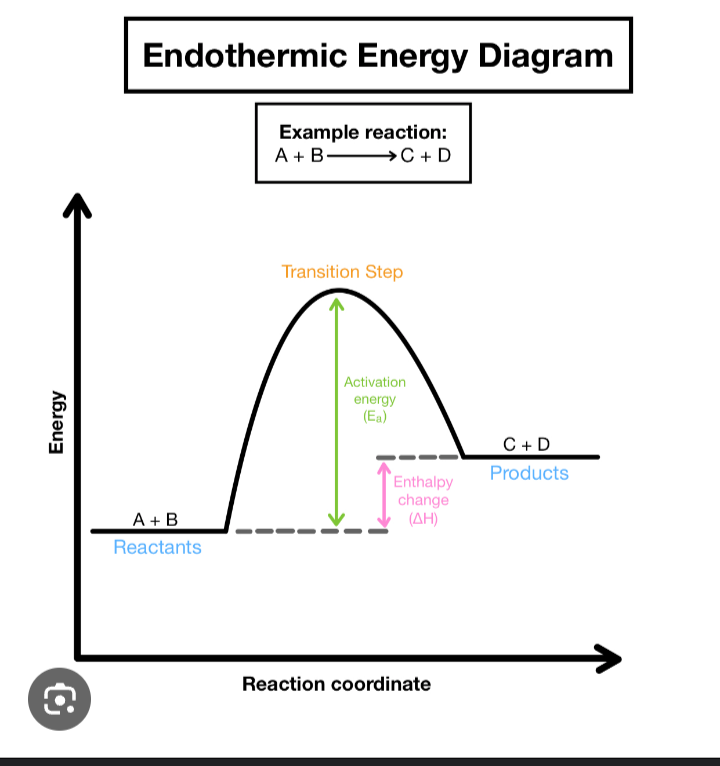
Endothermic
COLD: absorbs energy
∆H is Positive and is on the Left
What effect do catalysts have on the concentration of products or reactants or ∆H?
No effect
Nature favors ______ entropy (chaos) and ______ energy (temperature).
High entropy, and low energy