Physics 30: Unit B - Chapter 10.1 (THEORY)
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
21 Terms
Electrostatics
the study of static electricity
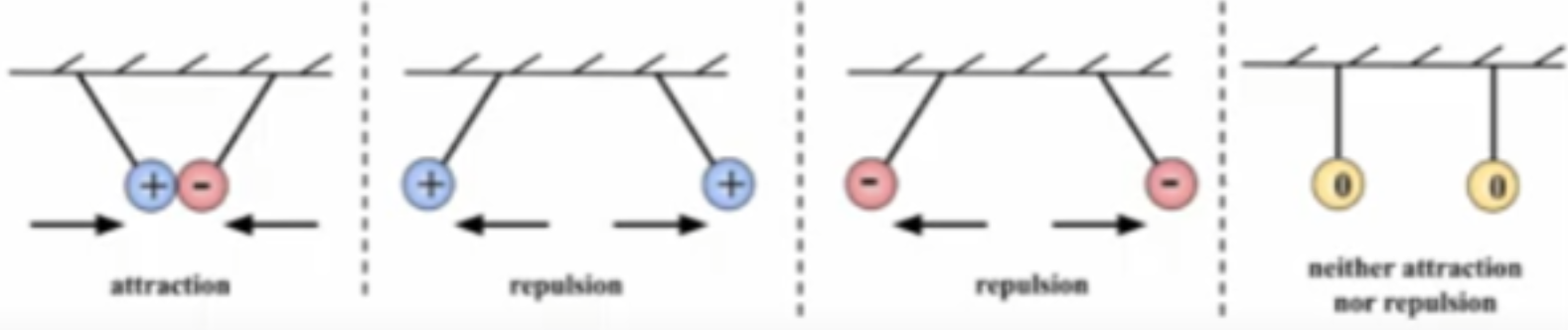
The Law of Charges
LIKE charges REPEL
OPPOSITES ATTRACT
Two types of charges: positive & negative
charge is SCALAR: “+” or “-” just communicates they are opposite types!
Objects that have no overall charge are neutral
Law of Charges (pictures)
Ionized
assumed to mean an electron has been removed
Atomic Theory (all matter contains electric charge)
all matter is made of atoms
atoms are made of protons (+), electrons (-) and neutrons (0)
protons and neutrons are fixed in the nucleus
electrons are loosely held around the nucleus in energy levels
check formula sheet for charges and masses of each!
Neutral Charges DO contain charges
they just have equal amounts of protons (positive) and electrons (negative)!
Movement of Charges
protons do NOT move within a solid → only the electrons can
free positive charges that are NOT CONFINED within a solid are of course capable of moving
if electrostatic forces are strong enough, the entire object itself can be moved, bringing the positive charges with it.
Why can neutral objects be attracted and only attracted, never repelled to charged objects?
bring (-) near → e- “flee” → leaving p+ closer
bring (+) near → e- moves towards, e- closer
(like repel, opposites attract)
Charge
charge of an object is determined by its excess (more) or deficit (fewer) of electrons compared to protons
about how many more/fewer electrons it has than protons

Conductors
materials that ALLOW charge to move freely
Insulators
materials that DO NOT allow charge to move freely
Semiconductors
materials that are:
good conductors under certain conditions
good insulators under other conditions
Superconductors
material that has zero electrical resistance
General Rule of Conductors and Insulators
metals are good conductors
non-metals are good insulators
the more freely that electrons are able to move through a material → the more conductive it is
Electroscope
device used to detect the presence of electrical charge
does NOT indicate the type of change

Law of Conservation of Charge
net charge of an isolated system is conserved
charge may be transferred, total charge stays constant
any transfers of charge are due entirely to the exchange of electrons, protons dont transfer
Static Discharge
electrons can move spontaneously if the potential difference between two objects is great enough
Three Ways to Change an Objects Net Charge
Conduction
Friction
Induction
Conduction (conductors, gain like charges)
When multiple charged objects are in contact, their electrons are free to move from one to the other.
The electrons will begin to move to the object that is more attractive (or less repulsive) to them.
As this happens, the charges of the two objects “even out”
The objects the electrons are leaving is becoming more attractive/less repulsive to the electrons
By losing negative electrons, it is becoming more positive → (opposites attract)
The object the electrons are going to is becoming less attractive/more repulsive to the electrons
By gaining negative electrons, it is becoming more negative → (like repel)
Friction (insulators, gain equal but opposite charges)
Not all nuclei are equally attractive to the electrons bound to them - some hold on to their electrons more tightly than others.
Rubbing objects “shakes up” the electrons
The energy put into the rubbing does work to rip off electrons from the nuclei.
They will then return to the one with a tighter hold on electrons.
This results in one material “stealing” electrons from the other
The final charges of two neutral materials charged by friction will always end up with opposite charges!
One gains a negative electron
One loses a negative electron
Induction (gain opposite charges)
charged object is brought near a neutral object
the neutral object is grounded: connected to unlimited storage reservoir for charge
electrons have a path to enter or leave the initially neutral object when the charged object is brought near
charged object (+) → electrons from the ground will be attracted and flood into the neutral object
charged object (-) → electrons in neutral object will be repelled and exit through the ground
ground must be removed before the charged object is taken away
cuts off path for electrons to enter/exit the neutral object (now charged)
if not removed, electrons will move through it and return the system into its initial charge distribution