AP Chemistry Units 1-5 Study Guide
1/144
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
145 Terms
Molar Mass
The conversion factor between grams (g) and moles (mol), found by multiplying the number of atoms by the masses of each atom and adding for the total.
Atomic Mass Unit
Unit for the mass of an atom, abbreviated to amu and based on C-12, which is 12 amu.
Avogadro’s Number
Used to find the number of atoms in a mole of a substance: conversion is 6.022 × 10²³
Atomic Number
Proton count in the element
Mass Number
The combined number of protons and neutrons in an element
Isotope
Specific formation of an element defined by how many neutrons there are
Average Atomic Mass Formula
∑(abundance of isotope n) × (mass of isotope n). Uses isotope’s weighted average.
Mass Spectra/Spectrometry
Measures percent abundances and masses of different isotopes. The diagrams are sorted by ratio of mass to charge (m/z), and the height indicates the abundance.
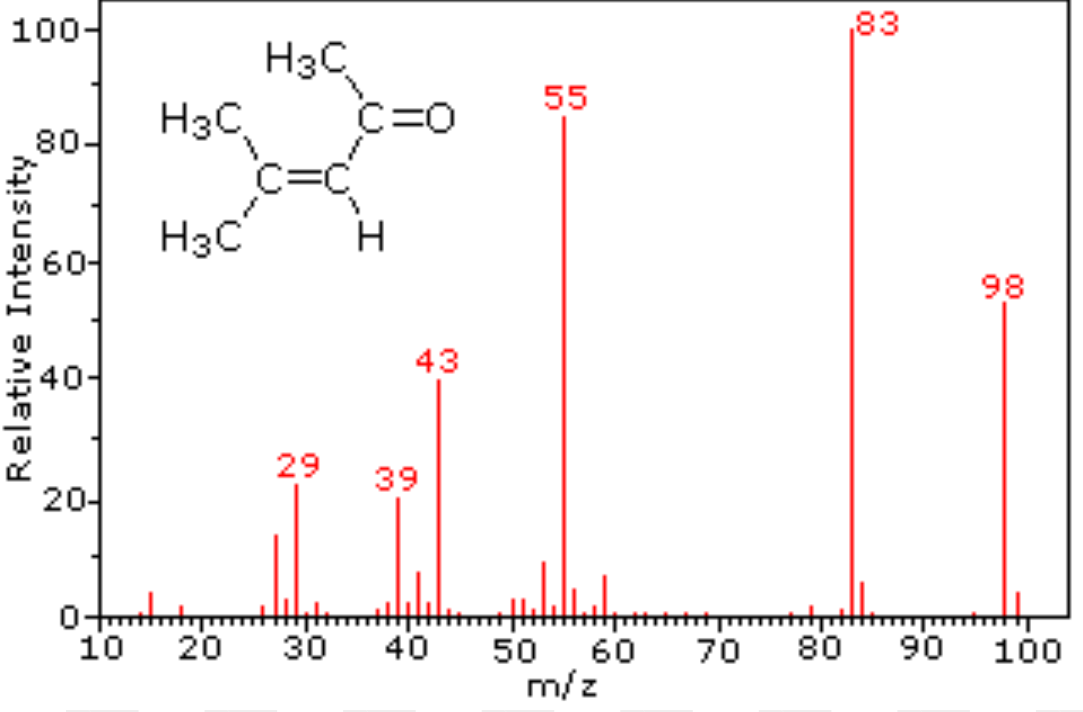
Percent Composition
The percentage of how much of an element is in a compound. Found by using the formula ((molar mass of element × element count) / (molar mass of compound)) × 100%. Essentially follows a similar pattern to stoichiometry, new/old.
Empirical Formula
Version of a composition that condenses the formula down to its simplest ratio. To find it, you can use the masses of each element (convert to moles, divide by smallest mole value, round or multiply to find whole number ratio) or the percentages of each element (‘assume you have 100g’ and use percentages as masses).
Molecular Formula
Version of a composition that demonstrates the stable/“normal” ratio of every element in the compound. Can be found by using the molar masses of each element (find the mass of the empirical formula, then compute molar mass/empirical mass to find your factor which you multiply all the subscripts by).
Mass Percent
The percentage of how much of an element or compound is in a mixture. Can be found by using the formula (mass of compound / mass of mixture) × 100%.
Contamination
Mixture with an unwanted chemical included. Typically caused by leftover reactants, accidental addition, etc. These can distort expected results in mass percent calculations; for instance, if mass percent ≠ percent composition, and it can be guessed based off a differing margin.
Coulomb’s Law
Proportional relationship of a charge’s attraction/repulsion showing how force increases as charge increases but decreases as radius increases. Expressed mathematically using F = k(Q₁Q₂/r), or relationally without the constant k using F ∝ Q₁Q₂/r (where F = Coulombic force of attraction/repulsion, Q₁ = Charge of protons in entire atom, Q₂ = Charge of a single electron, and r = radius between protons and electrons).
Orbital
Range of which an electron has the highest probability of existing at any given moment around the atom. There are s orbitals, p orbitals, d orbitals, and f orbitals. Each has 1 to 7 subshells. Ones further out have less energy due to their larger radii as they are on a higher energy level, and thus further from the nucleus of its atom.
Suborbital
Orientation of an orbital. For instance, s orbitals have one, and p orbitals have 3. Each can hold two electrons at a time.
Energy Level
Areas around the atom which 1 to 4 orbitals exist, detonated by principal quantum number and separated by nodes.
Aufbau Principle
Electrons fill whatever orbital requires the least amount of energy to fill first.
Hund’s Rule
Electrons fill each suborbital with one electron before adding the second. So, if an orbital has more than one suborbital, every suborbital will get one electron before a second is added to any of them.
Pauli Exclusion
No two electrons can have the same quantum number, so the electrons paired in a suborbital together must have opposite spins.
Valence Electron
Electron that sits on the outermost energy level of an atom, and are the easiest to remove from an atom.
Core Electron
Electron that is closer to the nucleus of the atom than its valence electrons. They have smaller radii than valence electrons, and they effect coulombic force by shielding valence electrons from the nucleus (Use Zeff instead of Z to represent protons in case this causes significant impact).
Photoelectron Spectroscopy
Graph recording the binding energy of each electron in any given element, which is helpful for displaying electron configuration and energy levels.
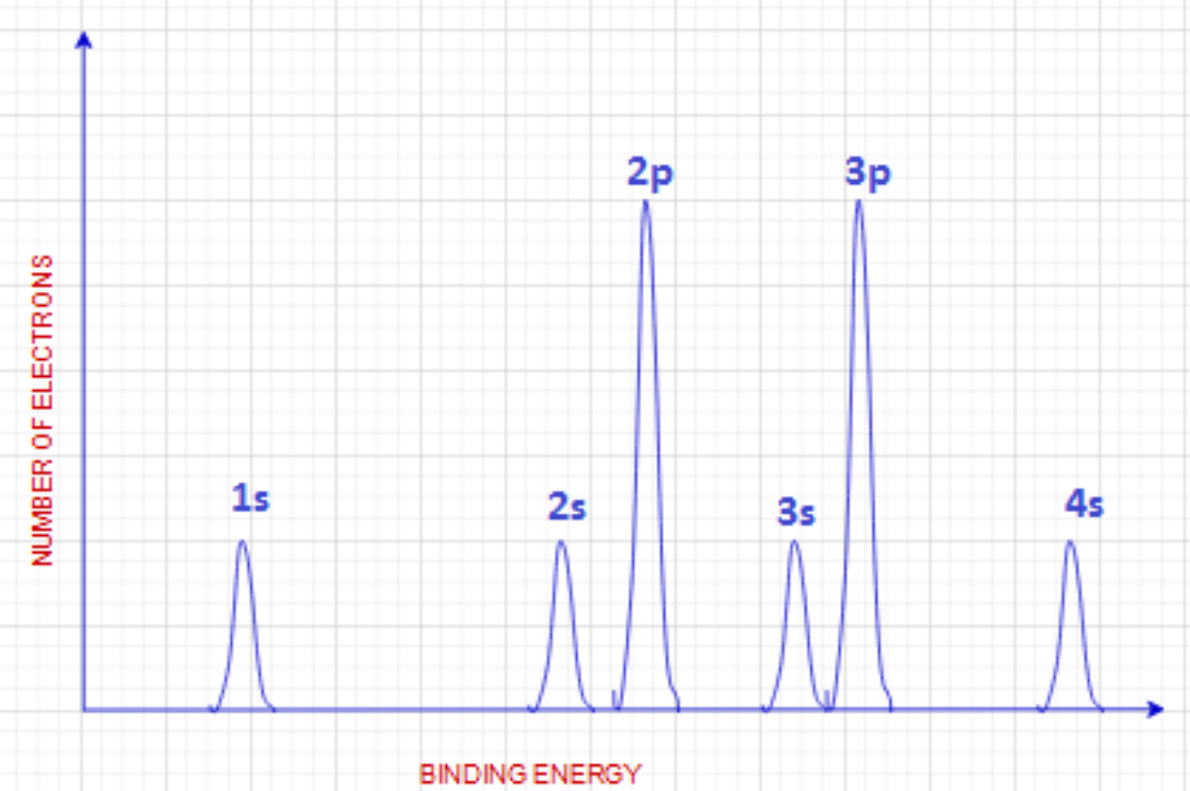
Binding Energy
Energy required to eject an electron with radiation. It tends to increase the more protons the atom has, as the larger positive charge will pull more on the elements. Can be found using the equation E = hv (where h is Planck’s constant [6.626 × 10⁻³⁴ Js], and v is the frequency of the photon used for the radiation).
Photon Frequency Equation
To convert from a photon’s wavelength to frequency or vice versa, use the equation c = λv (where c is the speed of light [2.998 × 10⁸ m/s], λ is the photon’s wavelength, and v is the photon’s frequency).
Periodic Law
Electron orbitals cause atomic properties to follow specific trends that align with the periodic table. These trends are mostly contingent on the increase of protons and energy levels as the table goes on.
Ionic Radius
The radius of an ionized element. Since cations lose electrons during the ionization process, and as a result can drop the atom a whole energy level, their radii trend downwards and to the left. However, since anions gain electrons, which provide added repulsion forces from extra electrons, their radii trend downwards and to the right.
Atomic Radius
The valence electron’s distance from their atom’s nucleus. Trends downward due to the larger orbitals and to the right due to the pull of more protons (increasing towards Cs). This trend can be demonstrated mathematically with Coulomb’s law.
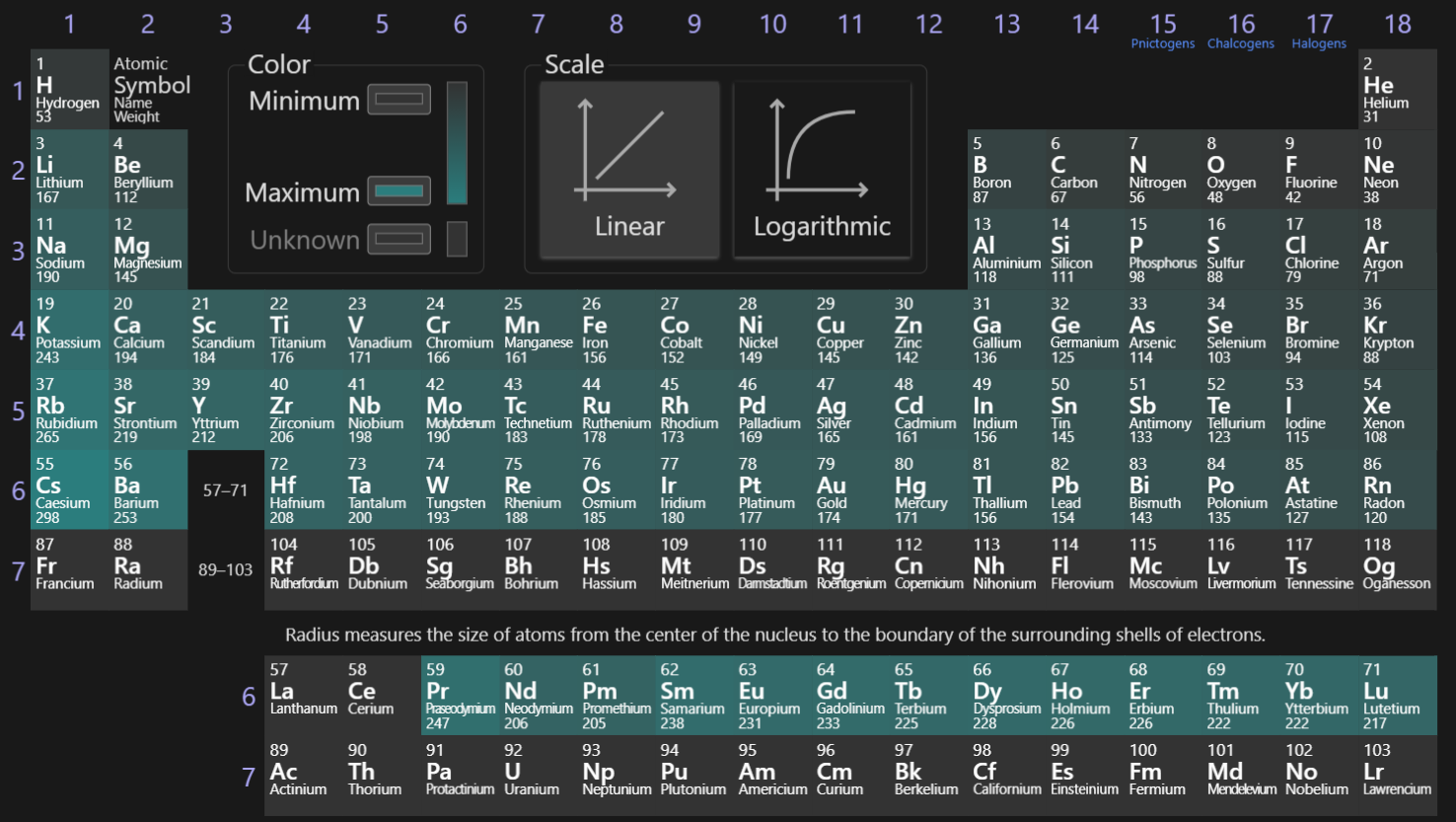
Ionization Energy
The energy needed to remove an electron from an element, or to ionize it. Can be stacked, and each stack gives the amount of energy needed to remove another electron on top of the ones previously removed (1st ionization energy, 2nd ionization, 3rd, 4th…). Jumps in orbitals can be useful to find these ions. Due to Coulombic forces, this trends upward due to smaller radii, and to the right due to more protons being added (increasing towards He).
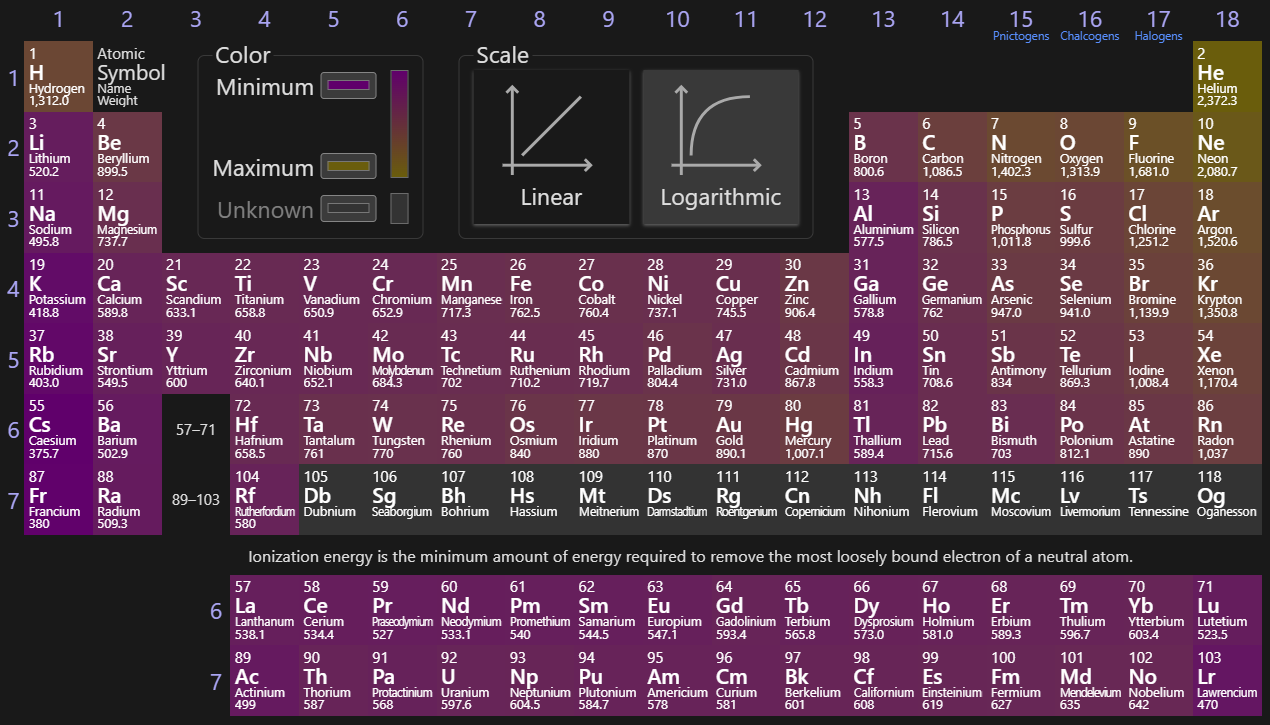
Electron Affinity
The amount of energy gained by an atom when it gains another electron. For noble gasses, this will always be 0 k/mol. But since it is affected by the strength of the attraction between the nucleus and new electron as described in Coulomb’s law for all other elements, it trends upward and to the right as (increasing towards Cl).

Electronegativity
The strength by which the nucleus of an atoms pulls on its electrons. Noble gasses do not have this, since they fill every electron slot available in their energy level. This factor determines what type of chemical bond forms in a compound containing any given element. This trends upward and to the right due to the greater pull of the nucleus on its electrons (increasing towards F).
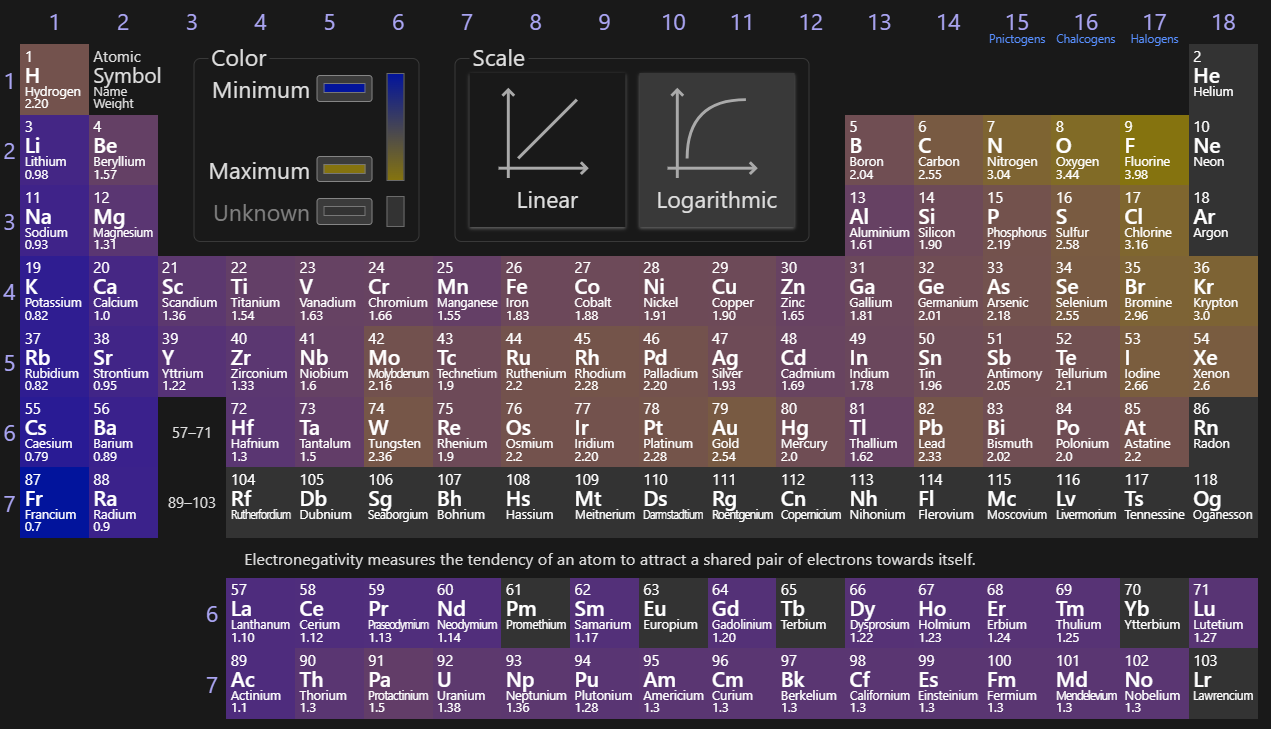
Electrostatic Force
The force which holds ionic bonds together. To be kept neutral, the electrons the least electronegative species loses must be equivalent to the number of electrons gained by the most electronegative species (this transfer happens so that the species can achieve a complete valence shell). This will form a predictable pattern across elements in similar groups, and these will form AₓBᵧ analogous compounds, where A is a metal and B is a nonmetal.
The Chemical Bond Spectrum
Different types of forces between atoms are determined by the properties of the involved species. While some elements such as Bi can engage in more than one type of bond, some are restricted to just one form on interaction with other atoms. Each bond lies on the spectrum with respect to electronegativity average and difference.
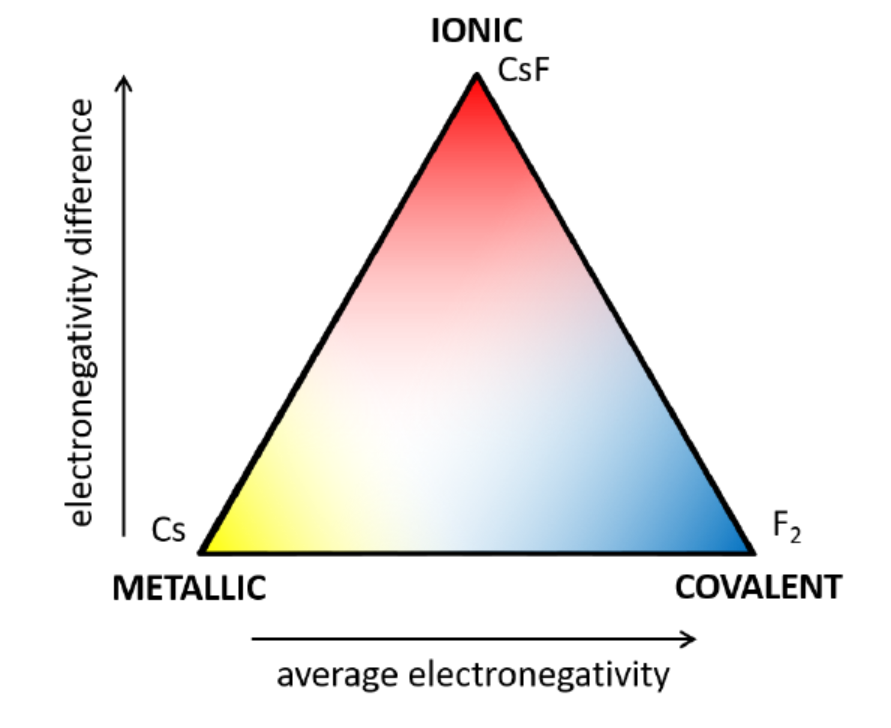
Ionic Bond
Attractions which involve cations attracting to anions (which can be preexisting or be formed when the bond is made) as governed by Coulomb’s law. The force here depends on the magnitude of each charge (-2, -1, 1, 2, etc.) and the size of the ion, as larger charges have more force and larger ions have weaker forces. While solids made of these bonds have low conductivity, liquids and dissolved compounds can conduct better.
Covalent Bond
Attraction which atoms share electrons. The orbitals of each atom overlap, situating the valence electrons in the same region. The distribution of these electrons is contingent on electronegativity. These can be made up of “single” 2-electron bonds, “double” 4-electron bonds, and “triple” 6-electron bonds. Some general things to remember about these attractions is that F will always be the most negative element, H, C, P & S are always more positive, Cl & O are always more negative (unless, of course, with F), and N, Br & I are typically negative as well.
Metallic Bond
Attraction which metals with low electronegativity allow their electrons to move freely through the metal “sea of electrons." Metals get their malleability and conductivity properties due to this, as the electrons that make up the metal can move much easier than in other bond types.
DEN Range
The spectrum which determines a bond’s polarity based on the involved species’ difference in electronegativity. A 1.7 or above will indicate an ionic bond, while anything below calls for a covalent or metallic bond. Then, 1.69 - 0.9 is very polar, 0.89 - 0.4 is moderately polar, and 0.39 - 0 is nonpolar. If the species involved have similar electronegativities, they are more likely to form a nonpolar covalent bond, but if they are strikingly different, you’ll probably see a polar covalent or even ionic bond form instead.
Bond Energy Diagram
Graph which shows the potential energy of chemical bonds, helping people predict how long and strong the chemical bond shown is. The potential energy well shows the most stable point of the bond at the trough of the graph. The bond can always exist at that state, and here it has the least amount of potential energy. The lower the trough, the shorter and stronger the bond formed there is. Bonds can be stronger because they are of a stronger bond type (triple being strongest) or made of smaller atoms as smaller bonds are stronger.
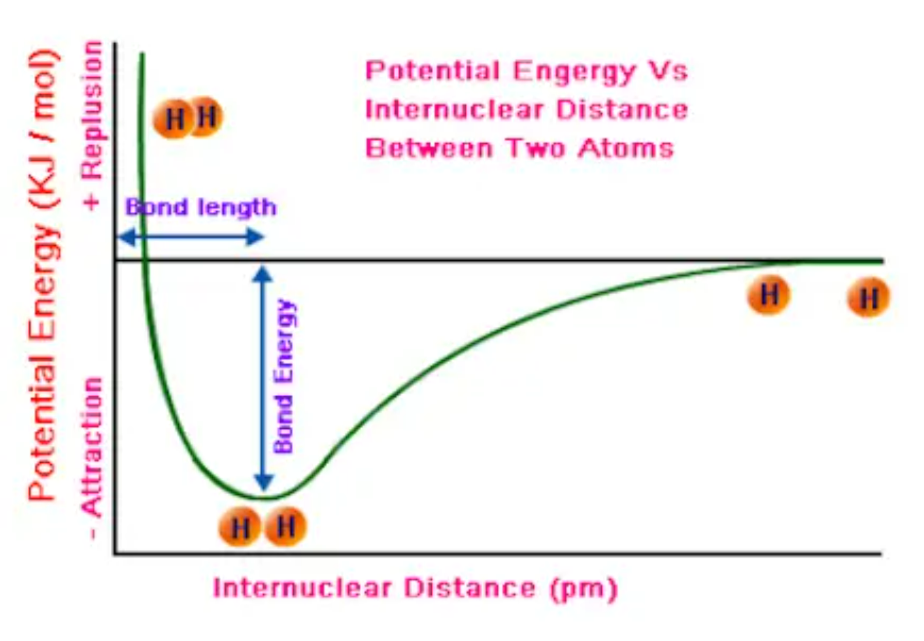
Lattice Energy
Energy caused by Coulombic forces between ions in ionic bonds. Stronger energies can be caused larger ionic charges and smaller ionic radii.
Isothermic
Maintaining a constant temperature during a process
Delocalized Electron
Electrons which are not bound to orbitals and therefore can flow freely. They have Coulombic forces with metal cations, which forms metallic bonds. An element has more if they have more valence electrons in turn, resulting in larger changes and smaller radii as well.
Alloy
Homogenous mixture (technically) formed by the metallic bonds of multiple elements at any ratio, which are typically stronger than regular metals. Due to this nature, they are customizable and used for many different purposes. As solids, they can be referred to as “solid state solutions” as well.
Substitutional Alloy
Alloy formed when some of the atoms that would typically make up the metal’s structure are replaced with the atoms of the add-in. This is common when the radii of both elements are similar, although there will still be some irregularities; in fact, these slight differences in atomic radii can make it harder for atoms to slip past each other in the alloy, thus making it stronger.
Interstitial Alloy
Alloy formed when the atoms of a smaller element, usually a nonmetal, squeeze between the metal cations that make up the original metal. This new element is typically added in small percentages, because otherwise they would lose tensile strength. This addition also increases the overall strength of the alloy from the extra bonds and electrons that are now incorporated.
Lewis Diagram
Drawing which shows the shape of a chemical, which can be helpful when predicting reactions. They can be drawn by making note of how many valence electrons each element has, connecting each atom with a single bond, and then finish filling in octets working inwards using lone pairs. When electrons run out, double/triple bonds can be used to take care of any lone pairs in need. If you’re drawing an ion, bracket the entire ion and write the charge in the top right.
Octet Rule
Atoms will strive to fill their valence molecular orbital shell, which required eight electrons, when bonding with other atoms. There are a few exceptions, however: H only needs two electrons to fill its valence shell, Be & B have such low electronegativities that they will never hit eight (instead, Be is stable at four electrons and B is stable at six), and d orbitals will allow more than 8 in what is called an expanded octet. Expanded octets will rarely be the central atom of the diagram, unless dealing with Xe, S or P.
Bond Order
Numerical representation of a bond in a Lewis diagram: single bonds are valued at 1, double bonds at 2, and triple bonds at 3. The number of electrons involved in the bond is double this value.
Radical
Dangerous and unstable chemical which has unpaired electrons in its valence layer. Atoms that don’t have full pairs avoid this by forming one bond per each unpaired electron; although, some molecules and polyatomic ions don’t do this. These bonding pairs form an overlap in orbitals where the unpaired electron pairs with that of the other atom, thus preventing both atoms from being unstable.
Delocalized Bond
Bond which could connect to any atom in a Lewis Structure
Resonance
Accurate representation of delocalized bonds when drawing a Lewis Structure. These structures satisfy octet rules and are drawn by showing every way in which the structure can be drawn. Each version is drawn so that the average properties can be seen. In this case, the bond order would evaluate to the average of the bonds (bound count / electron domain count). While some resonance structures don’t have any one most likely version as they all have the same formal charge, if these formal charges vary than the most likely structure is the one which minimizes them.
Formal Charge
Result of the comparison of expected valence electron counts to actual valence electron counts. Since bonds are shared, they count as 1 electron while lone pairs count as 2 when making this comparison. This can determine a molecule’s charge, as formal charges which don’t cancel will result in an ion with the remaining positive or negative charge. Can be found by using the formula FC = V - L - B/2 (where V is the expected number of valence electrons, L is the number of lone pairs, and B is the number of bonds on the element).
Valence Shell Electron-Pair Repulsion Theory (VSEPR)
Theory which predicts the geometry of molecules, founded on the idea that valence electrons repel from one another, so they will be as far away from each other as possible in a molecule’s shape. Although this is neglectable, the more delocalized lone pairs there are, the more they will repel. The angle of an electron’s repulsion will depend on the amount of electron domains present. Also, while lone pairs don’t physically contribute to molecular geometry, their repulsion still does.
For different electron and molecular geometries, go here: https://knowt.com/flashcards/841bf0c5-ebab-461b-8672-ab2f158e957b
Electron Domain
A region containing a bond or lone pair around a central atom
Electron Geometry
Geometry of a molecule based on how many electron domains surround the central atom
Molecular Geometry
Geometry of a molecule based on how each bond or lone pair impacts the overall structure of the molecule, and the angle by which electron domains separate.
Sigma (σ) Bond
Bond type which can form as orbitals overlap during the formation of a covalent bond. This classification is a hybridized orbital which can overlap head-on, be any shape, and are typically stronger than their counterpart. Single, double, and triple bonds all contain one of these.
Pi (π) Bond
Bond type which can form as orbitals overlap during the formation of a covalent bond. This classification is a unhybridized orbital which is weaker than and always comes with its counterpart bond type. Double bonds have one of these, and tripe bonds have two of these.
Hybridization
When the orbitals of an atom shift as it forms a molecule for stability. Here, s and p orbitals will combine to make a new orbital, so bonding orbitals are even. Only valence electrons will undergo this process. The hybridized orbital count is dictated by electron domain count, as every electro dense space will have one hybrid orbital/sigma bond. These orbitals can hold any present lone pairs. For instance, a CO atom has two electron domains, so it will have two hybrid orbitals (and two which are not) with a hybridization of sp.
Effective Nuclear Charge
Charge of protons on electrons impacting nuclear radii and represented by Zeff.
Intermolecular Force (IMF)
Coulombic forces between molecules which require positive and negative charges and usually involves polar bonds. This is not the same as a bond, but can act like one as two molecules are attracted to one another, and it would take some force to separate them.
Partial Charge
Charge of an atom deviating from neutrality due to the random movement of electrons in around an atom or the electronegativity of one atom attracting electrons from another towards it, which can cause an atom to have a slightly more positive/negative charge, called a dipole. Written as δ+ if positive and δ- if negative.
London Dispersion Forces (LDFs)
Random dipoles caused by the unpredictable movement of electrons as stated by quantum mechanics. At random, they can temporarily form positive or negative partial charges which may cause atoms with opposite partial charges to attract to one another and become more polar very briefly. This occurs in every molecule. While it is typically regarded as the weakest intermolecular force, if the molecule is big enough it can be up there as the strongest because more electron domains will result in more random partial charges at a higher frequency.
Dipole-Induced Dipole Interactions
Dipole interactions caused by one polar molecule inducing a dipole in the other nonpolar molecule due to the electronegativity of the polar molecule. This can occur between any polar molecule and any other nonpolar molecule which can be influenced by the dipole. Have about the same weakness level as and only rank slightly higher than LDFs.
Dipole-Dipole Interactions
Dipole interactions between two polar molecules, each attracting towards the other’s opposite partial charge. This can occur between two polar molecules. These interactions are stronger than LDFs and Induced Dipole interactions, but not as strong as IMFs can get.
Hydrogen Bonding
Dipole-Dipole interaction between H and either N, O, or F (increasing in strength in that order). Since the difference in electronegativity is so stark in this combination, it is much stronger than a typical dipole-dipole IMF. Only possible between one molecule with a H and one with N, O, or F; elements like Cl wouldn’t work though since they’re too big.
Ion-Dipole Interactions
Dipole interactions between one ion and one polar molecule oriented with the opposite partial charge, creating the strongest possible IMF. Can happen with any ion and any polar molecule.
Polarizable
Atom with a larger electron cloud, which can make more random partial charges, and thus is easier to make “polar” for any brief moment.
Melting/Boiling Points
Properties involving a change in state of matter from solid to liquid and from liquid to gas, respectively. The stronger the substance’s IMF’s, the higher this value will be, as it will take more energy to separate the molecules from one another.
Vapor Pressure
Property involving molecule entering the vapor phase, which is when they become gaseous before they reach their boiling point. The weaker the IMFs, the higher this value will be as it will be easier for molecules to break away from others to become vapor.
Electrical Conductivity
Property involving the travel of electricity through a substance. Needs freely moving charged particles for the electricity to travel though.
Solid
Coldest state of matter where the atoms that comprise it are close to each other, thus allowing it to conduct heat better than other states of matter. They are very similar to liquids; however, liquids have more IMFs. Crystalline solids are compact due to IMFs, while amorphous solids are more spread out.
Ionic Solids
Solid made of large crystalline lattice structures, which have high melting points and low conductivity as electrons cannot move freely in ionic bonds. They are brittle (when atoms align, they repel causing a clean break) and insoluble in nonpolar solvents. They also often maintain a packed structure alternation between positive and negative ions to maximize ionic attractions, which goes on for infinity.
Network Covalent Solids
Solid made of bonds that intersect ionic and covalent, which are typically comprised of nonmetals and/or metalloids. Like ionic solids, they can extend their pattern of atom alignment to prioritize IMFs for infinity. They have high melting/boiling points, low vapor pressure, and cannot conduct (with the exception of graphite, as it has delocalized electrons). 2D ones are brittle as they are made of layers of covalent bonds, while 3D ones are hard and rigid.
Molecular Solvents
Solvent which doesn’t have charged particles, and therefore cannot conduct. While melting/boiling points and vapor pressure depend on the molecule’s polarity, when compared to other types of solids their melting/boiling points are typically lower while vapor pressure is higher.
Large Biomolecule/Polymer
Typically used by life forms, these molecules are so large that they can have dipole-dipole interactions with themselves, and they have lots of LDFs.
Ideal Gas Law
Basic assumption of how gases operate based on the equation PV=nRT (where P = pressure in atm, V = volume in L, n = moles, R = gas constant [0.08206], and T = temperature in K). This equation can be modified to solve for any variable, and when one side of the equation remains constant it can be used to find the change in a variable on the other side. For example, if nRT is unchanging, then P₁V₁=P₂V₂.
Kinetic Molecular Theory of Gasses
Gasses consist of tiny particles that are spread far apart from one another, and do not retain any attraction or repulsion among one another inherently. They are in constant random motion, and when they collide, those collisions are elastic. Since there are so many particles, statistics apply.
Solution
Homogeneous mixture where a solute is dissolved in a solvent, usually being water.
Molarity
The concentration of a solution as it is directly related to moles, represented by the equation M = n × V (where M is molarity, n is moles of solute, and V is volume of solvent in liters).
Dilution
Process by during which one changes the molarity and volume of a solution by adding more of a solvent to reduce the molarity of the solvent in the solution. Since the moles of the solvent stay the same, here M₁V₁ = M₂V₂.
Separation of Mixtures
When you have a mixture of a solid and a solid, it can be separated easily by simple manual pulling apart of the solids. For a solid and a liquid, filtration is the best route for separation (essentially evaporation). Finally, for liquid with liquid mixtures, separate them using distillation.
Distillation
Process where a mixture of a liquid and a liquid is set at a specific temperature so that one of the components is boiled (and since each liquid has a different boiling point, the other won’t if set correctly) and the gas travels through a cooling tube to condensate. Then, the reformed liquid flows down into a separate flask to complete the separation.
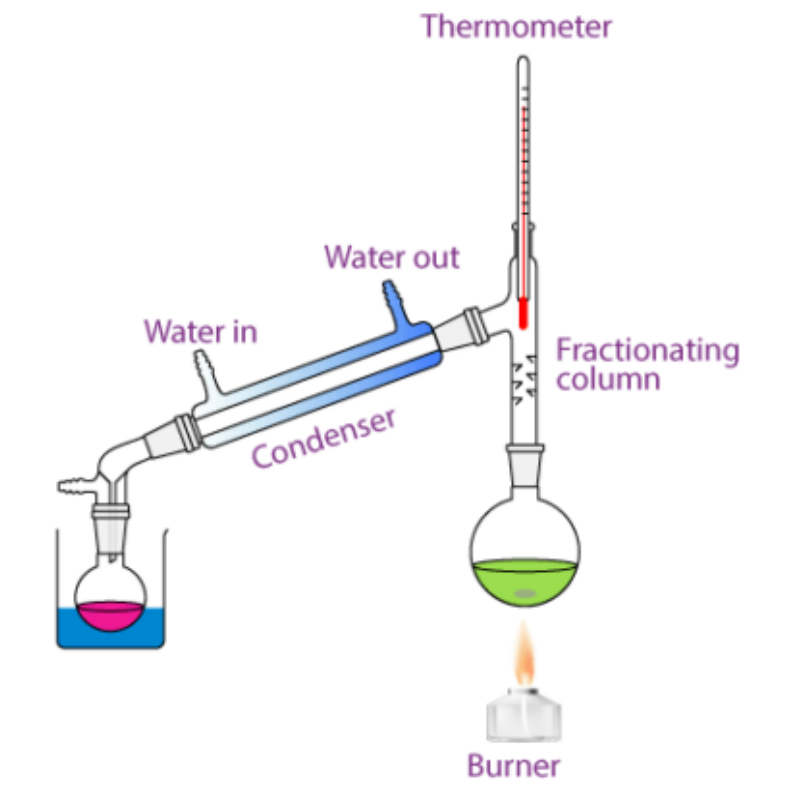
Chromatography
Separation of chemicals based on their polarities using a mobile phase and a stationary phase.
Mobile Phase
A typically nonpolar, solvent, liquid medium which during chromatography travels up to force the samples to move along with it. Assuming it is nonpolar, it can be said that things that don’t travel high are polar as they won’t want to associate with nonpolar molecules because they disrupt IMFs, however things that do travel high are less polar as the nonpolar molecule could retain them longer.
Stationary Phase
A typically polar chemical which stops things from moving fast if they are too polar. It stays in one place and attracts compounds in the mobile phase but only follows them so long as their polarity complies.
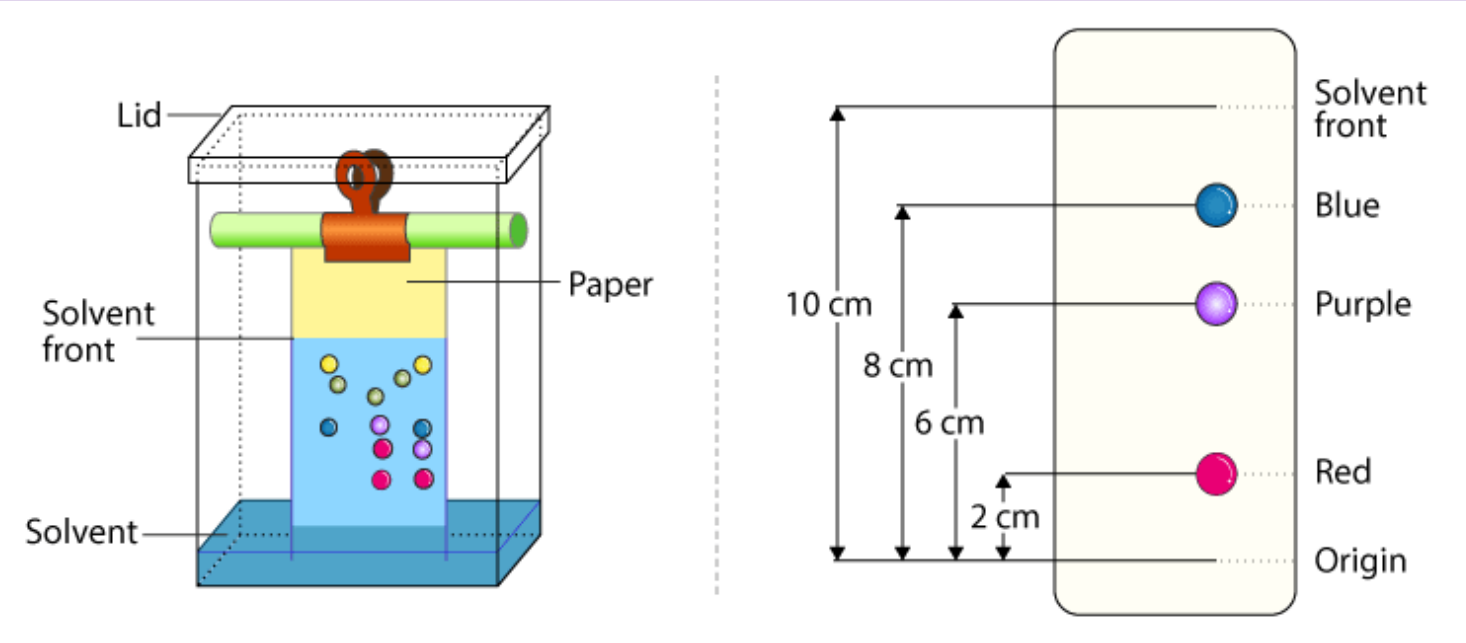
Paper Chromatography
Chromatography experiment where dye(s) is drawn on a long piece of paper, and the paper is hanged above the beaker by a piece of wood and touches the solvent at the bottom of the beaker, typically water, just beneath the dye dot. Then, as the solvent creeps up the paper, it will take the dye with it, and each molecule will separate based on when the mobile phase of the solvent is no longer able for them to cling on to. This works as paper, aka cellulose, is polar and has lots of hydrogen-bonding opportunities.
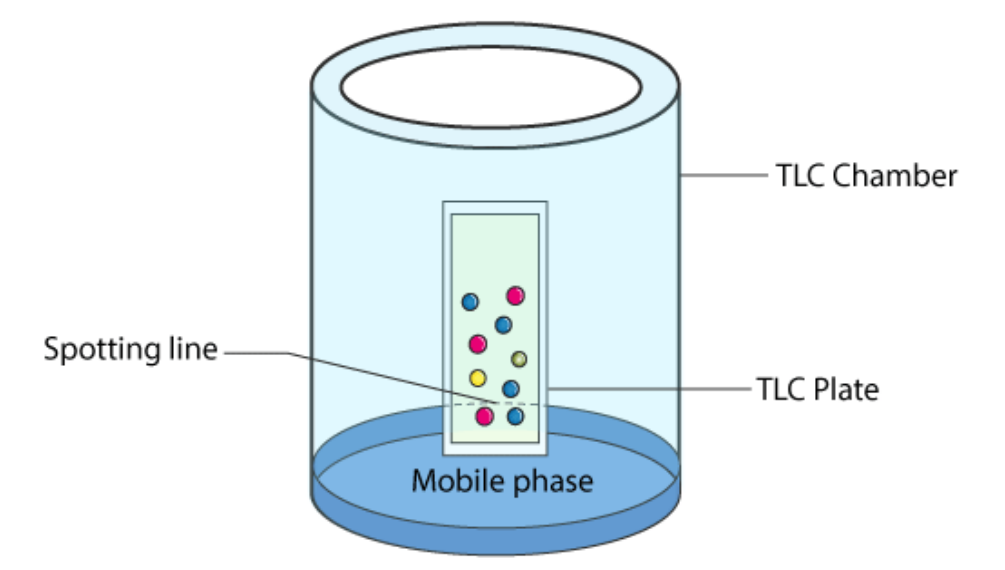
Thin-Layer Chromatography (TLC)
Chromatography experiment similar to paper chromatography but is designed to separate molecules faster. It is commonly used in labs to clearly distinguish every different part of the mixture in question.
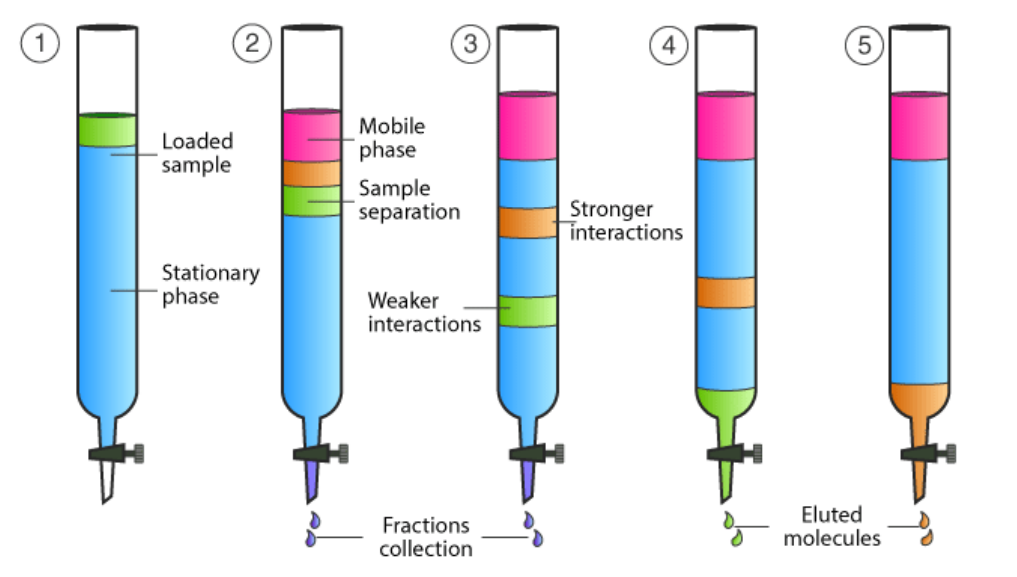
Column Chromatography
Chromatography experiment which allows for not only separation of different molecules in a sample, but also the collection of each output by having the stationary phase inside of a column, then the sample, then the mobile phase on top so that the sample travels through the stationary phase and gets separated by it, but then at the end is dispensed out of the column and can be retrieved using a beaker.
Retention Factor
Comparison of stationary phases distances, which is essentially the distance of the analyte (what is being analyzed) / distance of the solvent (portion we are looking for, typically told apart by its color as a result of paper chromatography). Essentially, this value is how high up the sample goes before stopping, or how slow it descends through the column.
Solubility
Generally, to determine a molecule’s solubility in another, compare polarities. Molecules will dissolve with others that harbor similar IMFs, so polar molecules will dissolve in other polar ones, while nonpolar ones will dissolve in other nonpolar ones. However, a polar molecule won’t dissolve with a nonpolar one, as the nonpolar molecule gets in the way of dipole-dipole or hydrogen bonding of the polar molecules. Also, the dissolution of an ion breaks apart its ionic structure, a typically endothermic process, and other molecules with opposite partial charges will attract to the now separate ions. How much an ionic compound dissolves in contingent on Coulomb’s law, as the stronger the forces of the ionic bonds are, the less the substance will dissolve.
Electromagnetic Spectrum
Spectrum of [light] wave frequencies which can interact with objects in many different ways based on where they fall in the spectrum. In the following order from weakest to strongest, there are radio waves, microwaves, infrared waves, visible/ultraviolet waves, x-rays, and gamma rays. When struck with light waves, materials emit electrons, and they can be impacted in a few ways; not just what colors we see.
Microwave Radiation
When hit with this frequency of a wave, the rotational level/spin of an electron is transitioned or switched.
Infrared Radiation
When hit with this frequency of a wave, the vibrational levels of an electron are increased.
Visible/Ultraviolet Radiation
When hit with this frequency of a wave, the electron is excited and jumps up an energy level.
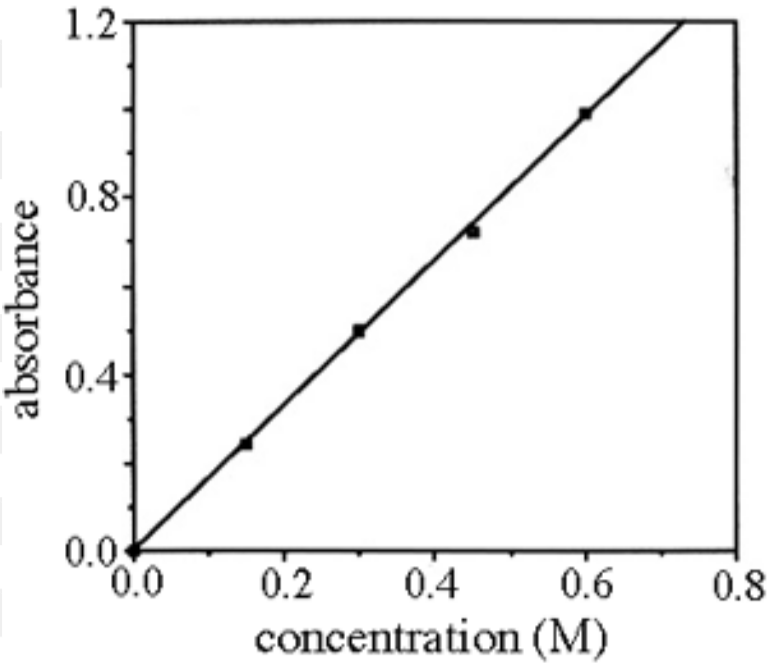
Beer-Lambert Law
The absorbance [of light waves] of a chemical is defined by the equation A = εbC (where A is absorbance often within the interval 0 < A < 1, ε is the molar absortivity constant, b is the path length of the curette in cm [if not indicated, it is assumed to be 1cm], and C is the concentration of the chemical in M or m/L). Experimentally, this can be demonstrated by placing a substance in a curette and targeting a specific frequency of light at it to measure its absorbance factor.
![<p>The absorbance [of light waves] of a chemical is defined by the equation A = εbC (where A is absorbance often within the interval 0 < A < 1, ε is the molar absortivity constant, b is the path length of the curette in cm [if not indicated, it is assumed to be 1cm], and C is the concentration of the chemical in <em>M</em> or m/L). Experimentally, this can be demonstrated by placing a substance in a curette and targeting a specific frequency of light at it to measure its absorbance factor.</p>](https://knowt-user-attachments.s3.amazonaws.com/4da36820-bbe6-4284-bf72-372256d4d592.png)
Percent Transmission
How much light makes it to the other side of the curette after the rest is absorbed by the chemical.
Curette
Container used to measure absorbance which has all clear sides, but sometimes will have two frosted sides. If this is the case, it is important to use the clear side to shoot the light through, as using the frosted side will absorb more light than it should and mess up your data. Similarly, on the side you use to target light ensure there are no fingerprints or other obstructions that could absorb light and produce flawed results.
Molar Absortivity Constant
Constant in Beer’s Law which determines the relationship between absorbance and concentration, which varies for every chemical and is represented by ε. It can be found by running a Beer’s Law experiment for many different trials/dilutions and using the data collected to form a line of best fit; if b = 1, then the slope of this line is ε. If not, then divide the slope of the line of best fit by b first to get ε.
Intensive/Intrinsic
Chemical property (any, need to be observed with a reaction) or physical property that isn’t affected based on the substance’s quantity. Some examples of this are color, hardness, and temperature.
Extensive/Extrinsic
Physical property only that is contingent on the substance’s quantity. Some examples of this are volume, length, and mass.