Bioprocessing: Cell Growth Kinetics and Yield Principles
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
Reaction kinetics
Rate of reaction i.e., how fast or quickly the reactants are converted into products.
Monod equation
A model that describes the rate of cell growth in a bioreactor.
Specific growth rate (𝜇)
Defined as 𝜇= 𝜇"#$[𝑆]/(𝐾% + [𝑆]), where 𝜇"#$ is maximum velocity or rate (s-1) and [S] is substrate concentration (mol m-3).
Rate or speed of reaction (𝑟!)
Defined as 𝑟! = 𝑉"#$[𝑆]/(𝐾" + [𝑆]), where 𝑉"#$ is maximum velocity or rate (mol m-3 s-1) and [S] is substrate concentration (mol m-3).
Michaelis constant (𝐾%)
A constant that represents the substrate concentration at which the reaction rate is half of 𝜇"#$.
Growth rate (𝑟$)
Defined as 𝑅$ = (𝑋2 - 𝑋1)/(𝑡2 - 𝑡1), where 𝑋1 and 𝑋2 are biomass concentrations at times 𝑡1 and 𝑡2.
Doubling time (𝑡H)
The time taken to double the initial cell concentration, calculated as 𝑡H = ln(2)/𝜇.
Exponential growth phase
The phase where the population grows at a constant rate, characterized by a constant specific growth rate (𝜇).
Lag phase
The initial phase of growth where cells adapt to their environment before division starts.
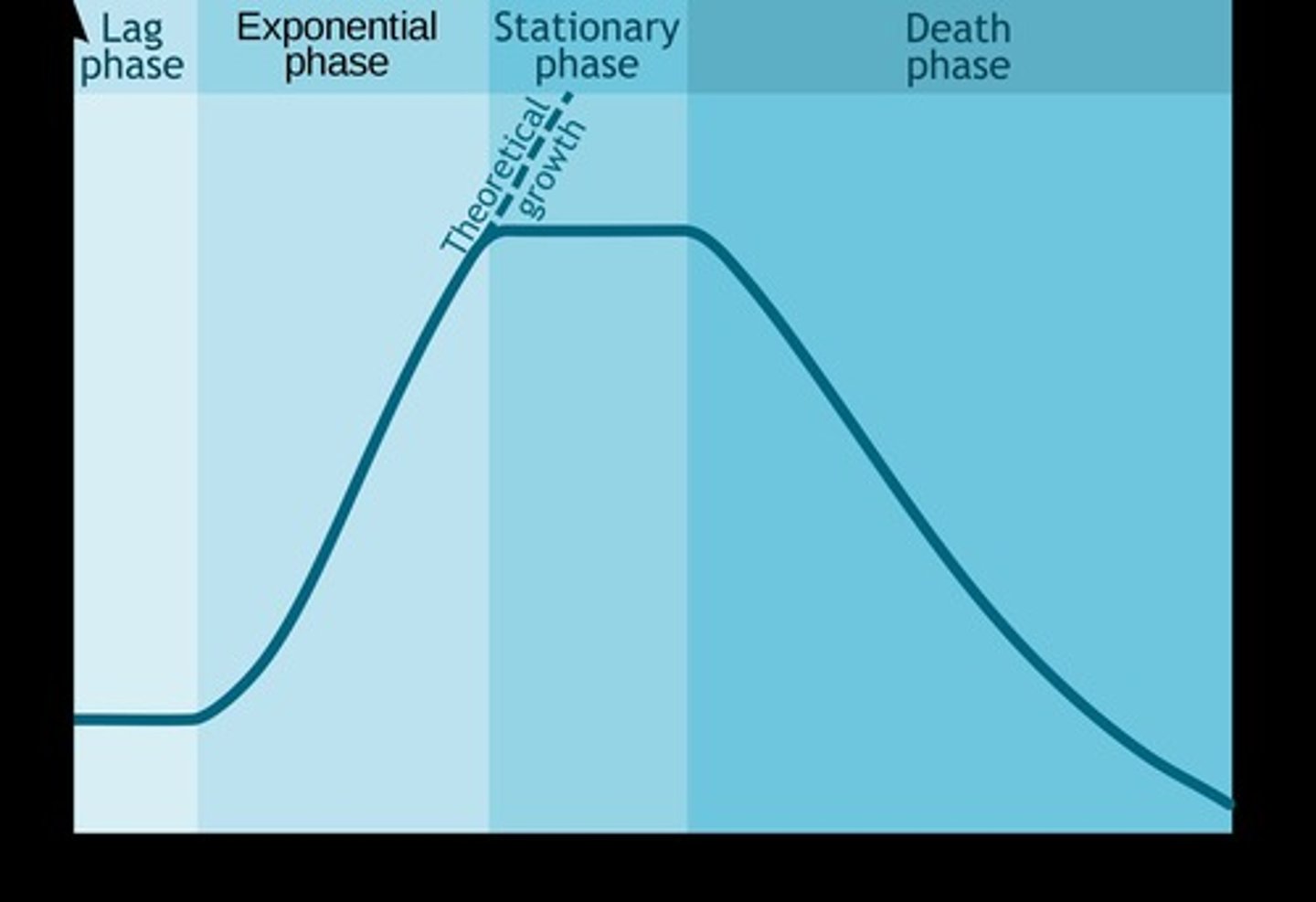
Stationary phase
The phase where the growth rate slows and stabilizes as resources become limited.
Death phase
The phase where the number of viable cells decreases due to nutrient depletion and waste accumulation.
First-order kinetics
A reaction where the rate is directly proportional to the concentration of one reactant.
Exponential growth equation
Describes growth as 𝑥 = 𝑥3𝑒^(𝜇𝑡), where 𝑥3 is the initial concentration.
Slope of ln 𝑥 vs 𝑡
The slope of the graph is equal to the specific growth rate (𝜇).
Substrate concentration ([𝑆])
The amount of substrate available for the reaction, measured in mol m-3.
Maximum velocity (𝑉"#$)
The highest rate of reaction achievable by a system at maximum substrate concentration.
Rate laws
Mathematical expressions that relate the rate of a reaction to the concentration of reactants.
Biomass concentration (𝑥)
The amount of biological material present in a given volume, measured in mol or mass per volume.
Order of reaction
The power to which the concentration of a reactant is raised in the rate law.
Proportionality in growth rate
The relationship where the growth rate (𝑟$) is directly proportional to the biomass concentration (𝑥).
Growth rate constant (𝜇)
The constant that defines the specific growth rate in relation to biomass concentration.
Exponential growth phases
Phases where the growth of the population follows an exponential pattern.
Monod equation
A mathematical model that describes the rate of cell growth in a bioreactor in relation to substrate concentration.
Specific growth rate (𝜇)
The rate of growth of a cell population, expressed in s-1.
Maximum velocity (𝜇max)
The maximum specific growth rate achievable by a microorganism, expressed in s-1.
Substrate concentration ([S])
The concentration of the substrate in the bioreactor, expressed in mol m-3.
Half-saturation constant (𝐾%)
The substrate concentration at which the specific growth rate is half of the maximum specific growth rate (𝜇max), expressed in mol m-3.
Yield coefficient (𝑌JK)
A measure of the efficiency of biomass production, calculated as the negative change in mass or moles of product (∆𝐽) produced divided by the change in mass or moles of substrate (∆𝐾) consumed.
Yield (𝑌&)
The ratio of the amount or moles of biomass (X) produced to the amount or moles of substrate (S) consumed.
Yield (𝑌$%)
The ratio of the amount or moles of product (P) produced to the amount or moles of substrate (S) consumed.
Yield (𝑌$&)
The ratio of the amount or moles of product (P) produced to the amount or moles of biomass (X) consumed.
Yield (𝑌&' )
The ratio of the amount or moles of biomass (X) produced to the amount or moles of oxygen (O) consumed.
Yield (𝑌(%)
The ratio of the amount or moles of CO2 (C) produced to the amount or moles of substrate (S) consumed.
Yield (𝑌(' 𝑜𝑟 𝑅𝑄)
The ratio of the amount or moles of CO2 (C) produced to the amount or moles of oxygen (O) consumed.
Yield (𝑌&)*"+
The ratio of the amount or moles of biomass (X) produced per kcal of heat evolved during fermentation.
Overall yields
Yields defined using the initial and final states of biomass, product, and substrate in batch culture.
Instantaneous yields
Yields quantified over shorter periods of time or at a specific time during the culture.
Reaction rate-homologue
A model that describes the specific growth rate or reaction rate following a Michaelis-Menten-like behavior.
Bioreactor
A vessel or container in which biological reactions occur, particularly for growing microorganisms.
Cell concentration
The amount of cells present in a given volume, typically measured in cells per liter.
Substrate
The substance on which an organism grows or feeds, often providing the necessary nutrients.
Biomass
The total mass of living organisms in a given volume, often measured in grams per liter.
Product
The substance produced as a result of a biological reaction or process.
Inflow and outflow
The measurable quantities of substrate entering and product exiting the bioreactor.
Lag phase
The initial phase of cell growth where cells adapt to their environment before division begins.
Exponential phase
The phase of cell growth where cells divide at a constant rate, leading to rapid population increase.
Stationary phase
The phase of cell growth where the rate of cell division slows and stabilizes due to limited resources.
Yield Coefficients
Related to rates of production and consumption.
Instantaneous Biomass Yield (Y'RS)
Rate of production of biomass divided by rate of consumption of substrate.
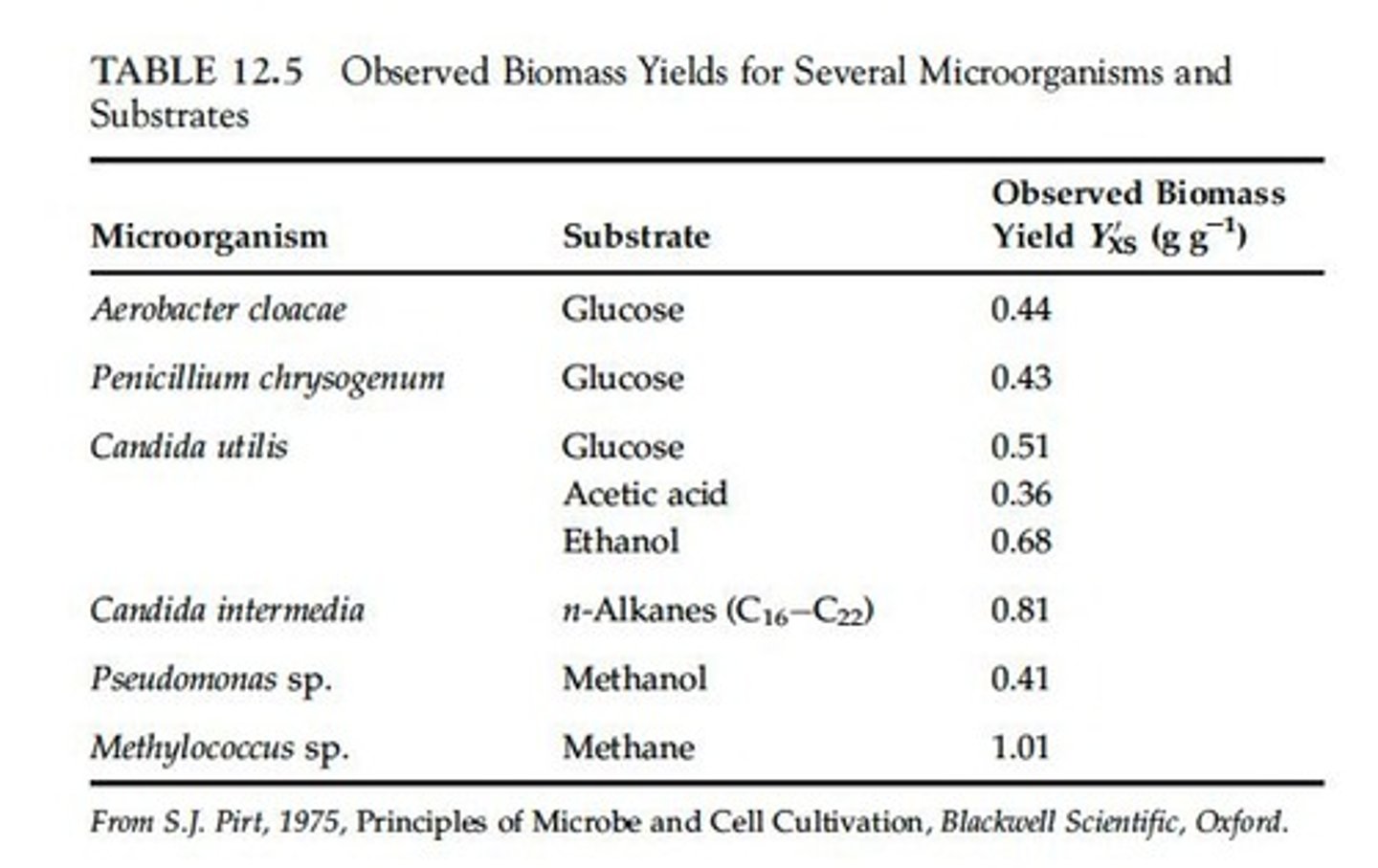
Observed Biomass Yield (Y'RS)
Calculated as -∆R/∆S, considering total substrate used for both growth and non-growth purposes.
Theoretical Biomass Yield (YRS)
Calculated as -∆R/∆S, where ∆S is substrate used for growth.
Substrate Used for Growth (∆SV)
Amount of substrate utilized for growth processes.
Substrate Used for Non-Growth Activities (∆SW)
Amount of substrate utilized for maintenance and other non-growth related processes.
Rate of Production of Product (rP)
Directly proportional to biomass concentration (x).
Specific Rate of Product Formation (qP)
Rate of production of product per unit biomass concentration.
Maintenance Factor (mP)
Represents the substrate used for maintenance of cells.
Rate of Biomass Growth (rX)
Rate at which biomass increases in a bioprocess.
First-Order Kinetics
A model where the rate of reaction is directly proportional to the concentration of one reactant.
Non-First-Order Kinetics
A model where the rate of reaction does not follow first-order behavior, often seen in secondary metabolite production.
Ethanol Utilization
Plant cells find it tougher to utilize ethanol compared to glucose.
Aerobic Production of Acetic Acid
Reaction equation: C2H5OH + O2 → CH3CO2H + H2O.
Ethanol Concentration Change
Initial concentration of 10 g l-1 reduced to 2 g l-1 after reaction.
Acetic Acid Production
7.5 g l-1 of acetic acid produced from the reaction.
Observed Yield vs Theoretical Yield
Comparison of actual yield obtained from a reaction versus the yield predicted by stoichiometry.
Bioprocess
A process that uses living cells or their components to obtain desired products.
Instantaneous Yield Calculation
YRS = rP/rC, where rP is the rate of production and rC is the rate of consumption.
Theoretical Yield Assumptions
Assumes all substrate is converted to biomass/product without losses.
Non-Growth-Related Substrate Usage
Small amounts of substrate are used for essential maintenance of cells or metabolism.
Yield Calculations
Involves determining the efficiency of substrate conversion into biomass or product.
Biomass Concentration (x)
The amount of biomass present in a given volume of culture.