Bioreactor Engineering: Cell Growth and Kinetics
1/107
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
108 Terms
Reaction kinetics
Rate of reaction i.e., how fast or quickly the reactants are converted into products.
Monod equation
Rate of cell growth in a bioreactor.
Specific growth rate (𝜇)
Defined as 𝜇= 𝑟$ ∝ 𝑥, where 𝑥 is cell concentration (mol or mass per volume).
Rate or speed of reaction (𝑟!)
The rate at which a chemical reaction proceeds.
Maximum velocity or rate (𝜇"#$)
The highest rate of reaction achievable under specific conditions.
Maximum velocity or rate (𝑉"#$)
The maximum velocity or rate expressed in mol m-3 s-1.
Substrate concentration ([𝑆])
The amount of substrate present in the bioreactor, measured in mol m-3.
Michaelis constant (𝐾%)
A constant that describes the substrate concentration at which the reaction rate is half of 𝜇"#$.
Michaelis constant (𝐾")
Another constant that describes the substrate concentration at which the reaction rate is half of 𝑉"#$.
First-order kinetics
A reaction where the rate is directly proportional to the concentration of one reactant.
Exponential growth phase
The phase where cell growth occurs at an increasing rate.
Lag phase
The initial phase of cell growth where there is little to no increase in cell numbers.
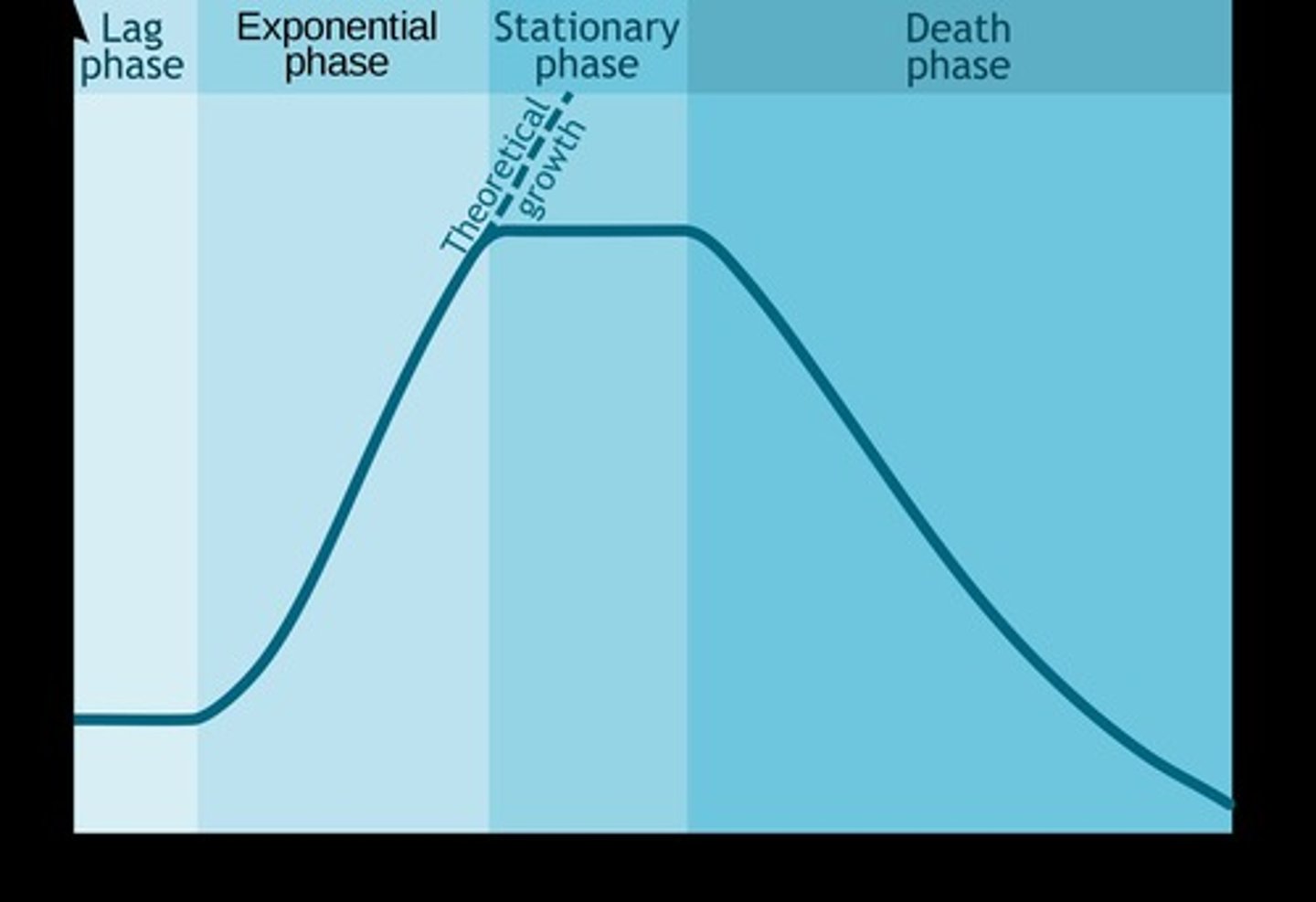
Stationary phase
The phase where cell growth rate slows and stabilizes as resources become limited.
Death phase
The phase where the number of viable cells decreases due to depletion of nutrients.
Acceleration phase
The phase where the growth rate begins to increase after the lag phase.
Yield
A measure of the amount of product formed per unit of substrate consumed.
Generic yield principles
Principles that define yields in complex reactions during cell culture.
Exponential growth equation
ln 𝑥 = 𝜇𝑡 + ln 𝑥7, where ln 𝑥 is plotted vs 𝑡.
Specific growth rate calculation
For example, if cell concentration increases from 20 gm to 40 gm, then 𝜇= (40−20)/20*5 = 0.2 min-1.
Rate laws
Laws that describe the relationship between the concentration of reactants and the rate of reaction.
Cell concentration (𝑥)
The amount of cells present in a given volume, expressed in mol or mass per volume.
Yield coefficient (𝑌@A)
A measure defined as −∆𝐽/∆𝐾, where ∆𝐽 is mass or moles of J produced and ∆𝐾 is mass or moles of K consumed.
∆𝐽
Mass or moles of J produced.
∆𝐾
Mass or moles of K consumed.
Biomass yield (𝑌%&)
Ratio of amount or moles of biomass (X) produced to amount or moles of substrate (S) consumed.
Product yield (𝑌$%)
Ratio of amount or moles of product (P) produced to amount or moles of substrate (S) consumed.
Biomass to product yield (𝑌$&)
Ratio of amount or moles of product (P) produced to amount or moles of biomass (X) consumed.
Biomass to oxygen yield (𝑌&')
Ratio of amount or moles of biomass (X) produced to amount or moles of oxygen (O) consumed.
CO2 to substrate yield (𝑌(%)
Ratio of amount or moles of CO2 (C) produced to amount or moles of substrate (S) consumed.
CO2 to oxygen yield (𝑌(' 𝑜𝑟 𝑅𝑄)
Ratio of amount or moles of CO2 (C) produced to amount or moles of oxygen (O) consumed.
Biomass per heat yield (𝑌&)*"+)
Ratio of amount or moles of biomass (X) produced per kcal of heat evolved during fermentation.
Instantaneous biomass yield (𝑌′HI)
Defined as the rate of production of biomass (𝑟$) divided by the rate of consumption of substrate (𝑟%).
Observed biomass yield (𝑌′HI)
Calculated as −∆H/∆I(K∆I), considering total substrate used for both growth and non-growth purposes.
Theoretical biomass yield (𝑌HI)
Calculated as −∆H/∆I(, where ∆𝑆L is substrate used for growth and ∆𝑆M is substrate used for non-growth activities.
Rate of production of product (𝑟!)
Proportional to biomass concentration (𝑥), defined as 𝑟! = 𝑞! 𝑥.
Specific rate of product formation (𝑞!)
The rate at which product is formed per unit of biomass.
Maintenance factor (𝑚!)
A factor that accounts for the maintenance of cells during fermentation.
Rate of biomass growth (𝑟$)
The rate at which biomass is produced in the bioprocess.
First-order kinetics
A model where the rate of reaction is directly proportional to the concentration of one reactant.
Non-growth-related substrate usage
Substrate used for essential maintenance of cells or metabolism, not contributing to growth.
Batch culture
A type of culture where overall yields are defined using initial and final states of biomass, product, and substrate.
Instantaneous yields
Yields quantified over shorter periods of time or at a particular time.
Substrate concentrations (∆𝐾)
The concentration of substrate that can change during the culture process.
𝑌!$
product yield from biomass consumed
𝑟$
rate of biomass growth
𝑚!
maintenance factor
𝑥
biomass concentration
𝑟!
rate of production of product
𝑞!
(𝑌!$ X 𝜇 + 𝑚!)
Stirred-tank reactor (STR)
Reactor configuration with a central stirrer shaft that mixes the contents.
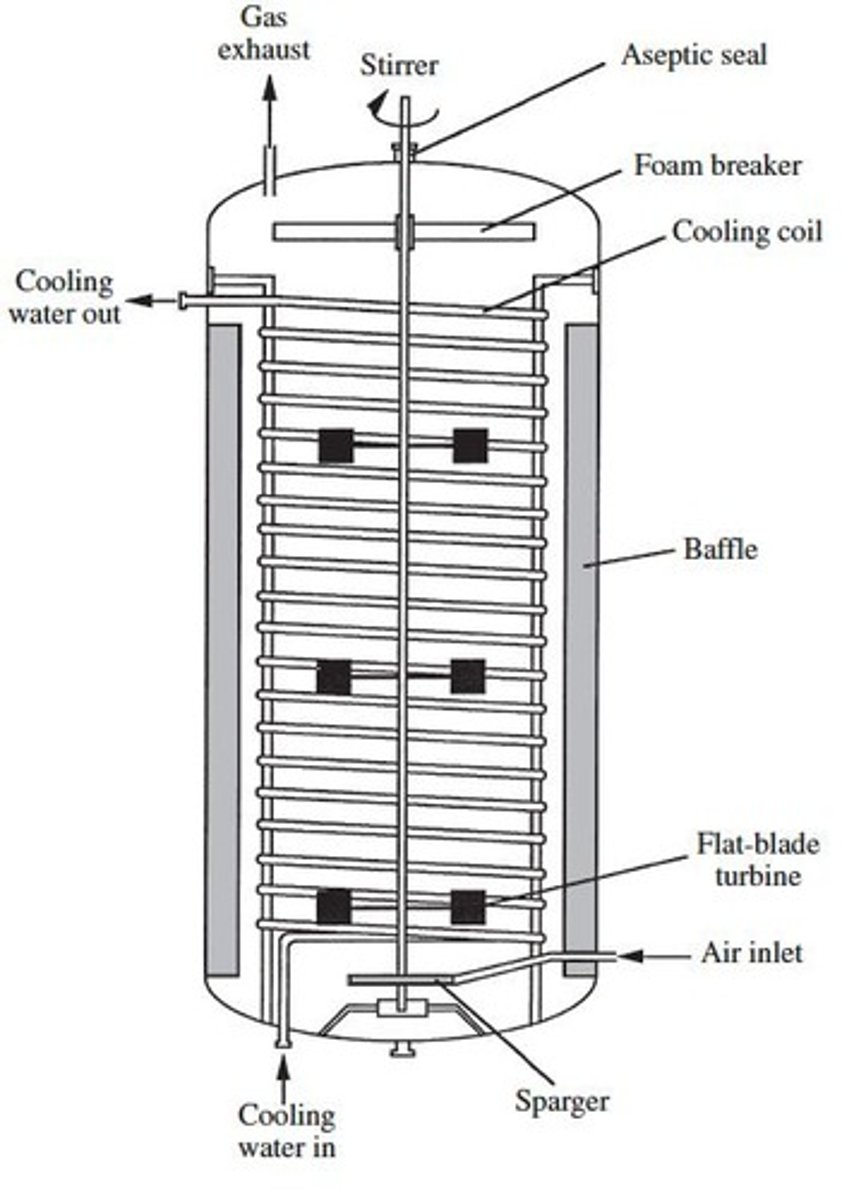
Reactor vessel material
Made from steel.
Central stirrer shaft
Contains many turbine blades at different heights that mix contents.
Liquid volume in STR
Typically, liquid volume is 60 - 70 %.
Stirrer power
High stirrer power is required for bubble dispersal.
Foam control
Necessary to manage foam during the mixing process.
Aspect ratio
Height to depth ratio, typically 1:1 or 3:1.
Oxygen dissolution
Taller reactors lead to more oxygen dissolution.
Mixing time (𝑡")
Time required to achieve less than 10% concentration differences after injection of bolus.
Reynolds number (𝑅𝑒)
Defined as 𝜌𝑁-𝐷-𝐷-/𝜇.
Stirrer speed (𝑁-)
Measured in rotation or radian/s.
Diameter of the impeller (𝐷-)
The size of the stirring device in the reactor.
Density of the liquid broth (𝜌)
Mass per unit volume of the broth.
Viscosity of the broth (𝜇)
A measure of the broth's resistance to flow.
Empirical relationship for mixing time
𝑡" = 5.9𝐷N/R/S-.
Power consumption in STR
Scale-up from 1 m³ to 100 m³ requires tremendous power increases, where 𝑃100 m³ = 2000 X 𝑃1 m³.
Batch process
Reactor is filled, substrate and biomass are added, and then closed for processing.
Fed-batch process
Substrate is added progressively while the reactor is closed to exit of biomass and media.
Continuous process
Substrate is fed in at a constant rate while biomass and toxins are removed at a constant rate.
Steady state in continuous process
At steady state, volume, concentration of substrate, and concentration of biomass/product is constant.
Concentration
𝐶#
Time
27
Generic mass balance for species I
𝑑𝑀- / 𝑑𝑡 = ̇𝑀-,-( − ̇𝑀-,&') + ̇𝑀-,V1( + ̇𝑀-,0&(%'
Bioreactor mass balance
/W- /) = ̇𝐹-(𝐶-,-( − ̇𝐹&')𝐶-,&') + V𝑟-,V1( − V𝑟-,0&(%, where ̇𝐹-( : Influx flow rate, ̇𝐹&') : Efflux flow rate, 𝐶-,-( : Inlet concentration, 𝐶-,&') : Outlet concentration, 𝑟-,V1( : rate of production or generation, 𝑟-,0&(% : rate of consumption, V : Volume
Batch process bioreactor
/W- /) = ̇𝐹-(𝐶-,-( − ̇𝐹&')𝐶-,&') + V𝑟-,V1( − V𝑟-,0&(%, where ̇𝐹-( = 0; ̇𝐹&') = 0; 𝑟-,V1( = 0; 𝑟-,0&(% = Michaelis-Menten
Michaelis-Menten equation
⇒𝑑(𝑠𝑉) / 𝑑𝑡 = 0 + 0 + 0 −𝑉( 𝑣XYZ[𝑠] / (𝐾" + [𝑠])
Rearranging variables
)[). %/ 𝐾X + [𝑠] ? 𝑑𝑡= ? 𝑣"#$[𝑠] 𝑑𝑠
Batch time equation
⇒𝑡,= A0 / G123 ln(%# / %/ + G123 ,
Enzyme production example
An enzyme is used to produce a compound used in the manufacture of sunscreen lotion. 𝑣"#$ for the enzyme is 2.5 mmol m-3 s-1; 𝐾" is 8.9 mM. The initial concentration of substrate (𝑠7) is 12 mM.
Biomass production in batch process
/W- /) = ̇𝐹-(𝐶-,-( − ̇𝐹&')𝐶-,&') + V𝑟-,V1( − V𝑟-,0&(%, where ̇𝐹-( = 0; ̇𝐹&') = 0; 𝑟-,0&(% = 0; 𝑟-,V1( = 𝜇𝑥
Biomass growth rate
⇒/($Q) / ) = 0 + 0 + 𝑉𝜇𝑥−0, where 𝜇= 8123[I] / A4K[I]
First-order kinetics
In a batch process 𝑠≫𝐾% ⟹ 𝜇= 𝜇"#$.
Batch time for biomass production
⇒𝑡,= 6 /
/ $/ $#, where 𝑡, is the batch timeComplex substrate consumption
Rate of Substrate consumption 𝑟-,0&(% = substrate used for biomass + substrate used for product + substrate used for maintenance
Substrate consumption equation
⇒𝑑𝑠 / 𝑑𝑡= −𝜇"#$𝑥 / 𝑌$% / 𝑌!% / 𝜇"#$ : Maximum specific growth rate of biomass, 𝑌$% : Biomass yield coefficient, 𝑌!% : Product yield coefficient, 𝑚% : maintenance coefficient
Batch time for product production
ln(1 + 𝜇"#$) / 𝑡, = 1 / (𝑝+ −𝑝7) / (𝜇"#$ / 𝑥7𝑞!)
Summary of batch time expressions
Depending on the situation we have four expressions for the batch time: 1. 𝑡, = A0 / G123, / G123 ln(%# / %/ + 2. 𝑡, = 6 /
/ $/ $#, % #5%/ , 3. 𝑡, = 6 / $ / 67 / 8123 ln(1 + 534K / 81239574K 14 / 8123 $#, 4. 𝑡, = 6 / 8123 ln(1 + 8123 / $ #`7 𝑝+ −𝑝7 ,Zymomonas mobilis fermentation example
Zymomonas mobilis is used to convert glucose to ethanol in a batch fermenter under anaerobic conditions. The yield of biomass from substrate (?) is 0.06 g g-1; the yield of product from biomass (? ) is 7.7 g g-1. The maintenance coefficient (?) is 2.2 g g-1 h-1; the specific rate of product formation due to maintenance is (?) 1.1 h-1. The maximum specific growth rate (?) of Z. mobilis is approximately 0.3 h-1. Five grams of bacteria are inoculated into 50 litres of medium containing 12 g L-1 glucose (?).
YXS
Yield of biomass from substrate, 0.06 g g-1
YPX
Yield of product from biomass, 7.7 g g-1
ms
Maintenance coefficient, 2.2 g g-1 h-1
mp
Specific rate of product formation due to maintenance, 1.1 h-1
𝜇"#$
Maximum specific growth rate of Z. mobilis, approximately 0.3 h-1
Batch culture time for 10g biomass
Determine the time required to produce 10g of biomass
Batch culture time for 90% substrate conversion
Determine the time required to achieve 90% substrate conversion
Batch culture time for 100g ethanol
Determine the time required to produce 100g of ethanol using 𝑞! = (𝑌!$ X 𝜇"#$ + 𝑚!)
Chemostat culture
Continuous fermenter operation under anaerobic conditions
Feed concentration
12g L-1 of glucose in continuous mode
Ks
Substrate saturation constant for the organism, 0.2 g L-1
Flow rate for steady-state substrate concentration
Determine the flow rate required for a steady-state substrate concentration of 1.5 g L-1
Cell density at flow rate
Determine the cell density at the specified flow rate