Physical Properties of Solutions: Types, Intermolecular Forces, and Colligative Properties in Chemistry
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
What is a solution?
A homogeneous mixture of two or more substances, consisting of a solvent and one or more solutes.
What is an unsaturated solution?
A solution that contains less solute than the solvent can dissolve at a specific temperature.
What is a saturated solution?
A solution that contains the maximum amount of solute that can dissolve in a solvent at a specific temperature.
What is a supersaturated solution?
A solution that contains more dissolved solute than is present in a saturated solution and is generally unstable.
What is solvation?
The process when solute molecules are separated from one another and surrounded by solvent molecules.
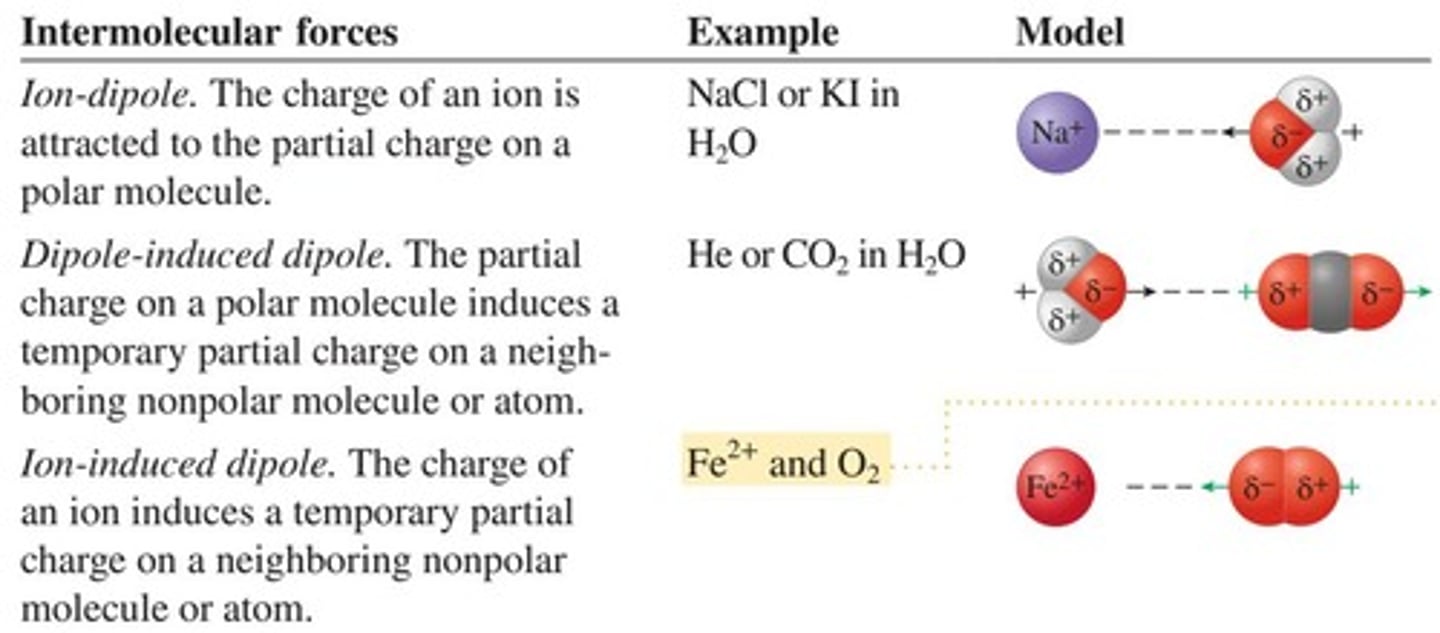
What types of interactions are involved in solvation?
Solute-solute interactions, solvent-solvent interactions, and solute-solvent interactions.
What are dispersion forces?
Attractive forces present in all substances.
What are dipole-dipole forces?
Attractive forces present in polar substances.
What is hydrogen bonding?
A strong type of dipole-dipole force exhibited by molecules with O-H, N-H, or F-H bonds.
What are ion-ion forces?
Attractive forces present in ionic substances.
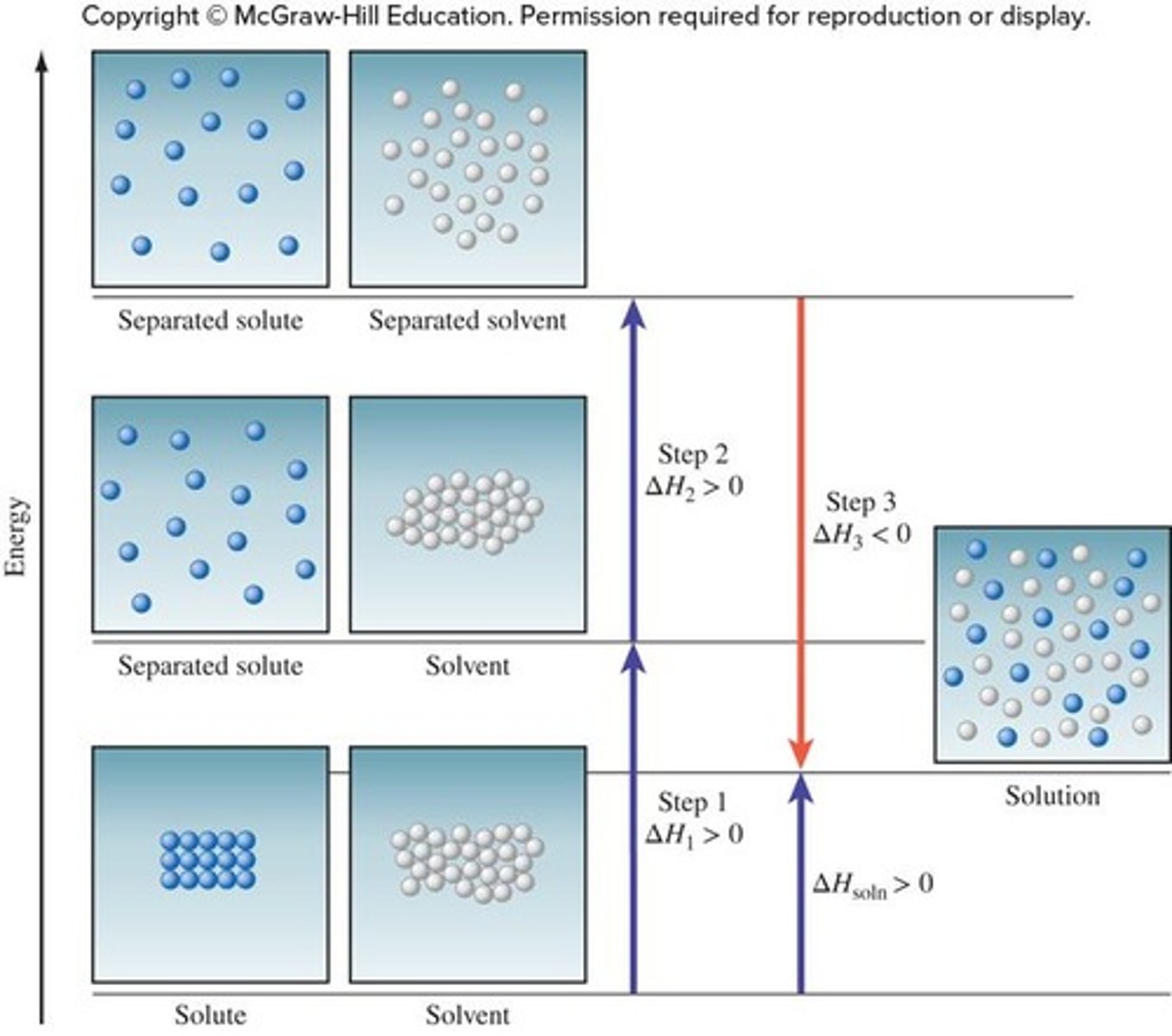
What is the overall solution-formation process when ΔHsolution < 0?
The process is exothermic, meaning the energy given off in mixing is greater than the energy required to separate solute and solvent.
What is the condition for a spontaneous process in terms of Gibbs free energy?
The change in Gibbs free energy (ΔG) must be negative.
What does 'like dissolves like' mean?
Two substances with similar types and magnitudes of intermolecular forces are likely to be soluble in each other.
What is molality?
The number of moles of solute dissolved in 1 kg (1000 g) of solvent.
What is percent by mass?
A concentration unit that expresses the mass of solute as a percentage of the total mass of the solution.
What is a mole in chemistry?
A unit used to measure the amount of a substance, defined as 1 mole = 6.022 × 10²³ particles.
What is the significance of Avogadro's number?
It defines the number of particles in one mole of a substance, which is 6.022 × 10²³.
What factors affect solubility?
Temperature and pressure.
What are colligative properties?
Properties that depend on the number of solute particles in a solution, such as vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
What is vapor pressure lowering?
A colligative property where the vapor pressure of a solvent decreases when a solute is added.
What is boiling point elevation?
A colligative property where the boiling point of a solvent increases when a solute is added.
What is freezing point depression?
A colligative property where the freezing point of a solvent decreases when a solute is added.
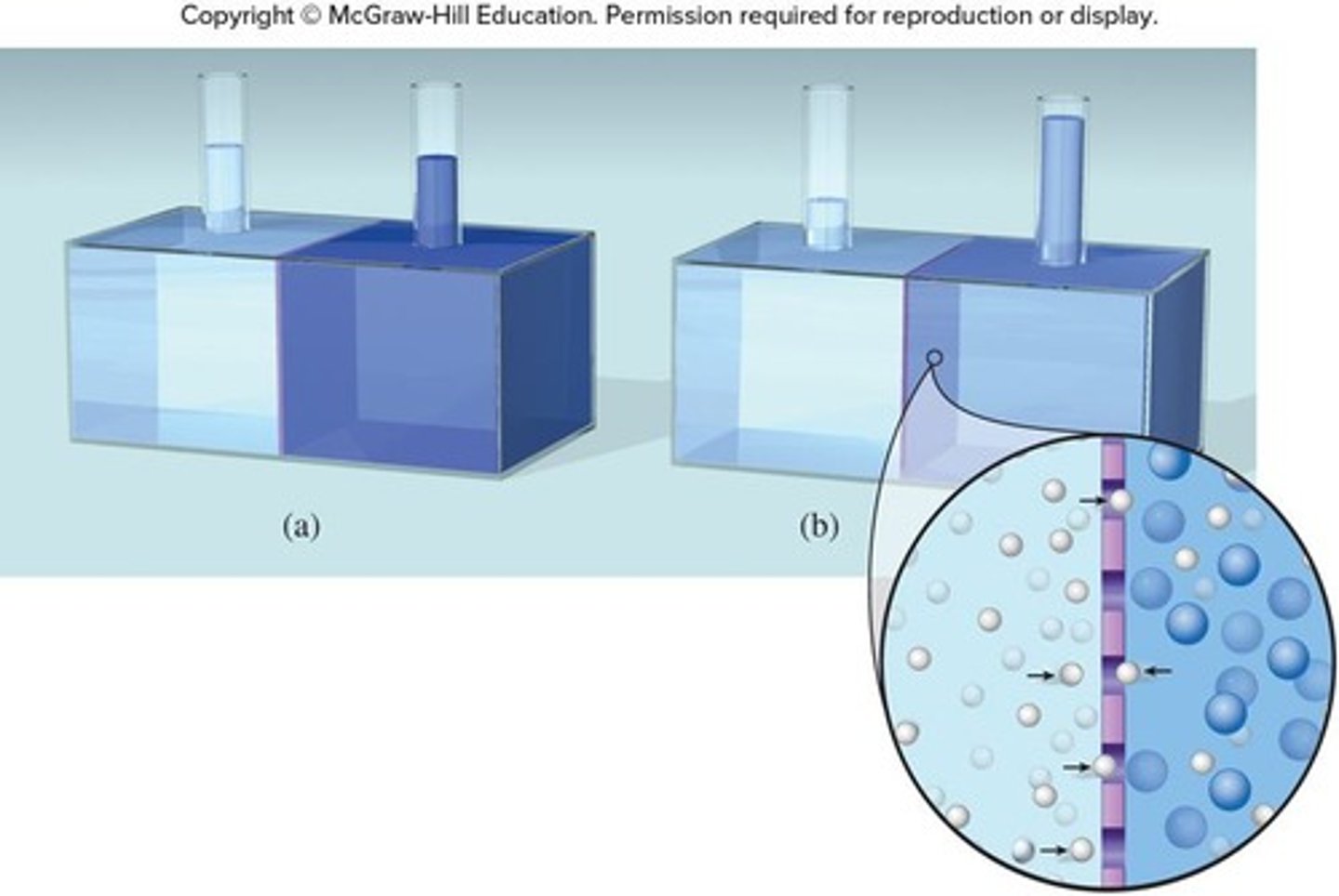
What is osmotic pressure?
The pressure required to stop the flow of solvent into a solution through a semipermeable membrane.
What is the relationship between energy and entropy in spontaneous solutions?
For a solution to be spontaneous, the increase in entropy (TΔS) must outweigh the change in enthalpy (ΔH).
What is the molar mass of glucose (C6H12O6)?
180.2 g/mol
How do you calculate the mass of water in a solution when given the mass of glucose and the total mass of the solution?
Subtract the mass of glucose from the total mass of the solution.
What is the formula for molality?
Molality (m) = moles of solute / kg of solvent
What is the formula for percent by mass?
Percent by mass = (mass of solute / mass of solution) × 100
What is the formula for parts per million (ppm)?
ppm = (mass of solute / mass of solution) × 10^6
What is the density of the glucose solution given in the example?
1.062 g/mL
How does temperature affect the solubility of most substances?
Temperature generally increases solubility, but not for all substances.
What does Henry's law state about gas solubility?
The solubility of a gas in a liquid is proportional to the pressure of the gas over the solution.
What is the relationship between molarity and molality in dilute aqueous solutions?
Molarity and molality are similar or the same only for very dilute solutions.
List the four main colligative properties.
1. Vapor pressure lowering 2. Boiling point elevation 3. Freezing point depression 4. Osmotic pressure
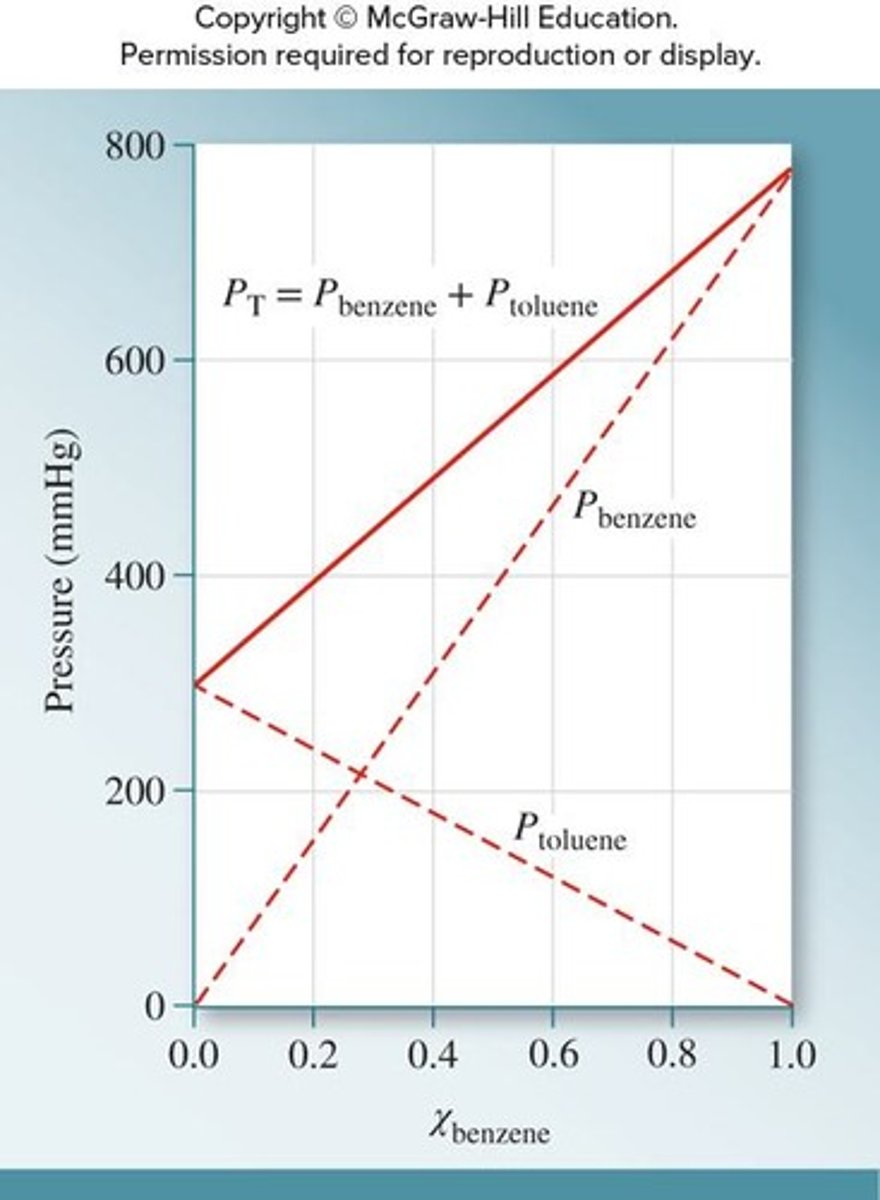
What does Raoult's law describe?
The partial pressure of a solvent over a solution is the vapor pressure of the pure solvent times the mole fraction of the solvent.
What is the boiling point elevation formula?
ΔTb = Kb m, where ΔTb is the boiling point elevation, Kb is the boiling point elevation constant, and m is molality.
What is the freezing point depression formula?
ΔTf = Kf m, where ΔTf is the freezing point depression, Kf is the freezing point depression constant, and m is molality.
What is the molar mass of isopropyl alcohol (C3H7OH)?
60.09 g/mol
What is the density of the rubbing alcohol solution mentioned?
0.79 g/mL at 20°C
What is the van't Hoff factor (i)?
A factor that accounts for the dissociation of electrolytes in solution.
How do you calculate the vapor pressure of a solvent over a solution?
Use Raoult's law: partial pressure = vapor pressure of pure solvent × mole fraction of solvent.
What happens to the vapor pressure of a solution if both components are volatile?
The vapor pressure of the solution is the sum of the individual partial pressures.
What is the effect of temperature on the solubility of gaseous solutes?
Most gaseous solutes become less soluble in water as temperature increases.
What is the effect of pressure on the solubility of gases according to Henry's law?
Increasing pressure increases the solubility of the gas in the liquid.
What is the concentration of carbon dioxide in a soft drink bottled under a pressure of 5.0 atm?
Use Henry's law to calculate the molar concentration at that pressure.
What is the freezing point of a solution containing 685 g of ethylene glycol in 2075 g of water?
−9.89°C
What is the boiling point of a solution containing 685 g of ethylene glycol in 2075 g of water?
102.7°C
What is the van't Hoff factor (i) for nonelectrolytes?
The van't Hoff factor (i) is 1 for all nonelectrolytes.
What does the van't Hoff factor (i) represent for strong electrolytes?
For strong electrolytes, i should equal the number of ions produced.
Why is the van't Hoff factor (i) usually smaller than predicted?
It is usually smaller due to the formation of ion pairs, which consist of cations and anions held together by electrostatic forces.
What effect does concentration have on the experimentally measured van't Hoff factors?
Concentration affects the measured van't Hoff factors, with lower concentrations leading to more complete dissociation.
How is the experimental van't Hoff factor for potassium iodide (KI) calculated?
It is calculated using the osmotic pressure formula: i = π / (MRT), where π is osmotic pressure, M is molarity, R is the gas constant, and T is temperature.
What was the experimental van't Hoff factor for KI at 0.0100 M concentration?
The experimental van't Hoff factor for KI at this concentration is 1.90.
What is percent dissociation in the context of electrolytes?
Percent dissociation is the percentage of dissolved molecules that separate into ions in a solution.
How does concentration affect the percent dissociation of strong electrolytes?
The percent dissociation of strong electrolytes is more complete at lower concentrations.
What is a colloid?
A colloid is a dispersion of particles of one substance throughout another substance, with particles larger than normal solute molecules.
What are the two categories of colloids based on their interaction with water?
Colloids can be categorized as hydrophilic (water-loving) or hydrophobic (water-fearing).
What role do hydrophilic groups play in colloids?
Hydrophilic groups on the surface of a large molecule stabilize the molecule in water.
What is emulsification?
Emulsification is the process of stabilizing a colloid that would otherwise not stay dispersed.
What is the Tyndall effect?
The Tyndall effect is the scattering of light by colloidal particles, which can help distinguish colloids from true solutions.
What is Raoult's Law?
Raoult's Law states that the vapor pressure of a solvent in a solution is directly proportional to the mole fraction of the solvent.
What is the relationship between boiling point elevation and molality?
Boiling point elevation is directly proportional to the molality of the solute in the solution.
How can colligative properties be used?
Colligative properties can be used to find solution molarity, molality, and/or a solute's molar mass.
What is the significance of the molecular formula of quinine in the context of molar mass calculations?
The molecular formula of quinine (C20H24N2O2) indicates its molar mass is 324.4 g/mol, which can be used to verify calculations.
What happens to the percent ionization of weak acids as concentration decreases?
For weak acids, the lower the concentration, the greater the percent ionization.
What is the formula for calculating molar mass using osmotic pressure?
Molar mass can be calculated using the formula: Molar mass = mass of solute / moles of solute.
What is the effect of ionization on the concentration of particles in solution?
The total concentration of particles in solution includes the concentration of intact molecules and the ions produced from ionization.