MCAT Kaplan General Chemistry Chapter 4: Compounds and stoichiometry
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Compounds
pure substances composed of 2 or more elements in a fixed proportion. Smallest unit is a molecule for covalent compounds.
Molecules
two or more atoms held together by covalent bonds
formula unit
the lowest whole-number ratio of ions in an ionic compound
formula weight
the sum of the atomic weights of the atoms in the formula unit of an ionic compound. Unit is amu
molecular weight
the sum of the atomic weights of all the atoms in a molecule. Unit is amu
molar mass
the mass of one mole of a pure substance (g/mol)
Equivalents
how many moles of the thing we are interested in (like e- or H+) will one mole of a given compound produce. Ex: Sodium donates one equivalent of e- but magnesium donates 2 equivalents.
Equivalents=mass of compound/gram equivalent weight
gram equivalent weight
mass (in grams) that provides one equivalent of the particle of interest. equals molar mass/n, where n is the number of particles (like H+ or OH-) produced per molecule
normality (N)
equivalent of solute per liter of solution, often denoted by N.
1 N of HCl is 1 M HCl, but 1 N of H2CO3 is 0.5 M H2CO3 because it is diprotic. Molarity=Normality/n, where n is the number of equivalents of thing of interest a molecule produces.

Law of Constant Proportions
law that states that, in chemical compounds, the ratio of the elements is always the same
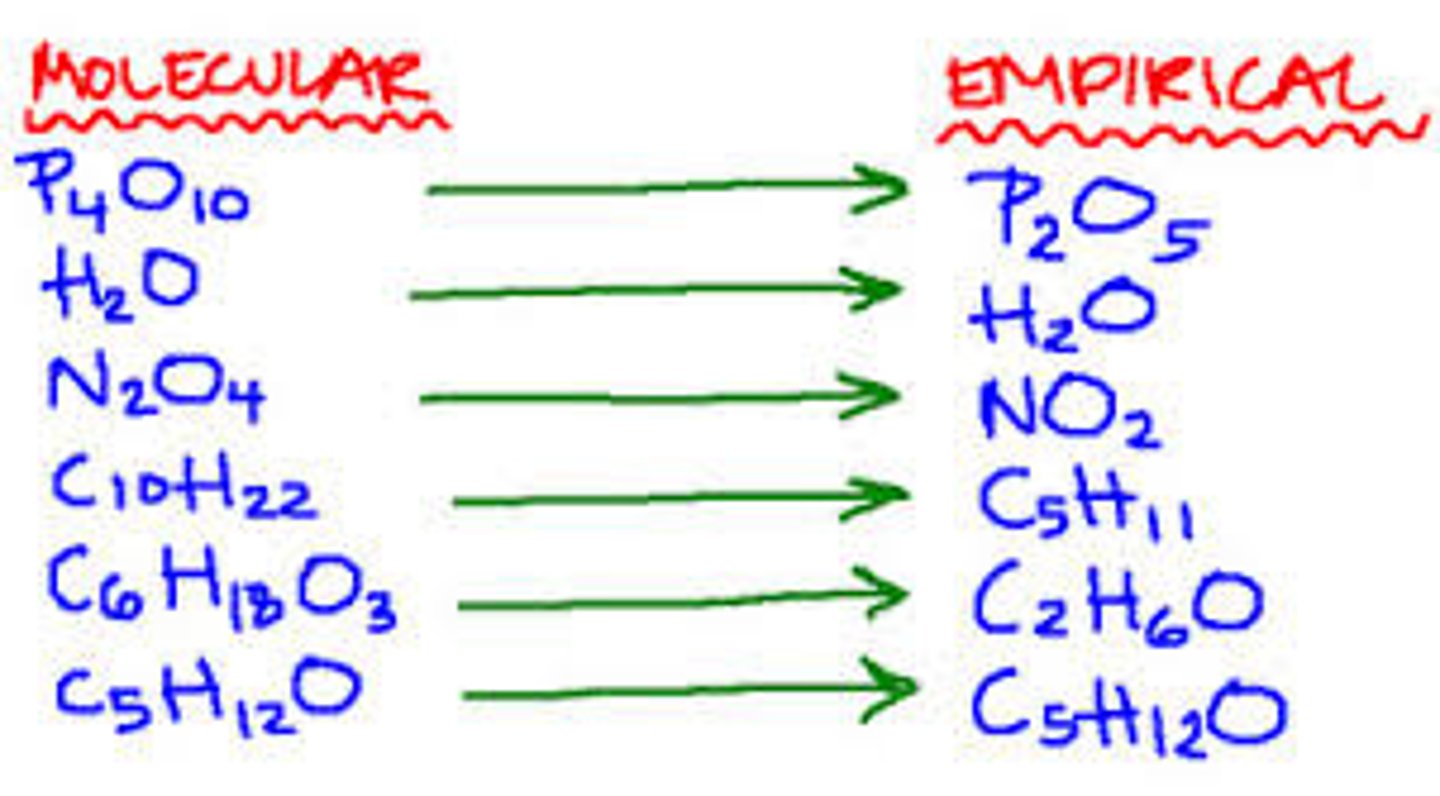
empirical formula
a formula with the lowest whole-number ratio of elements in a compound
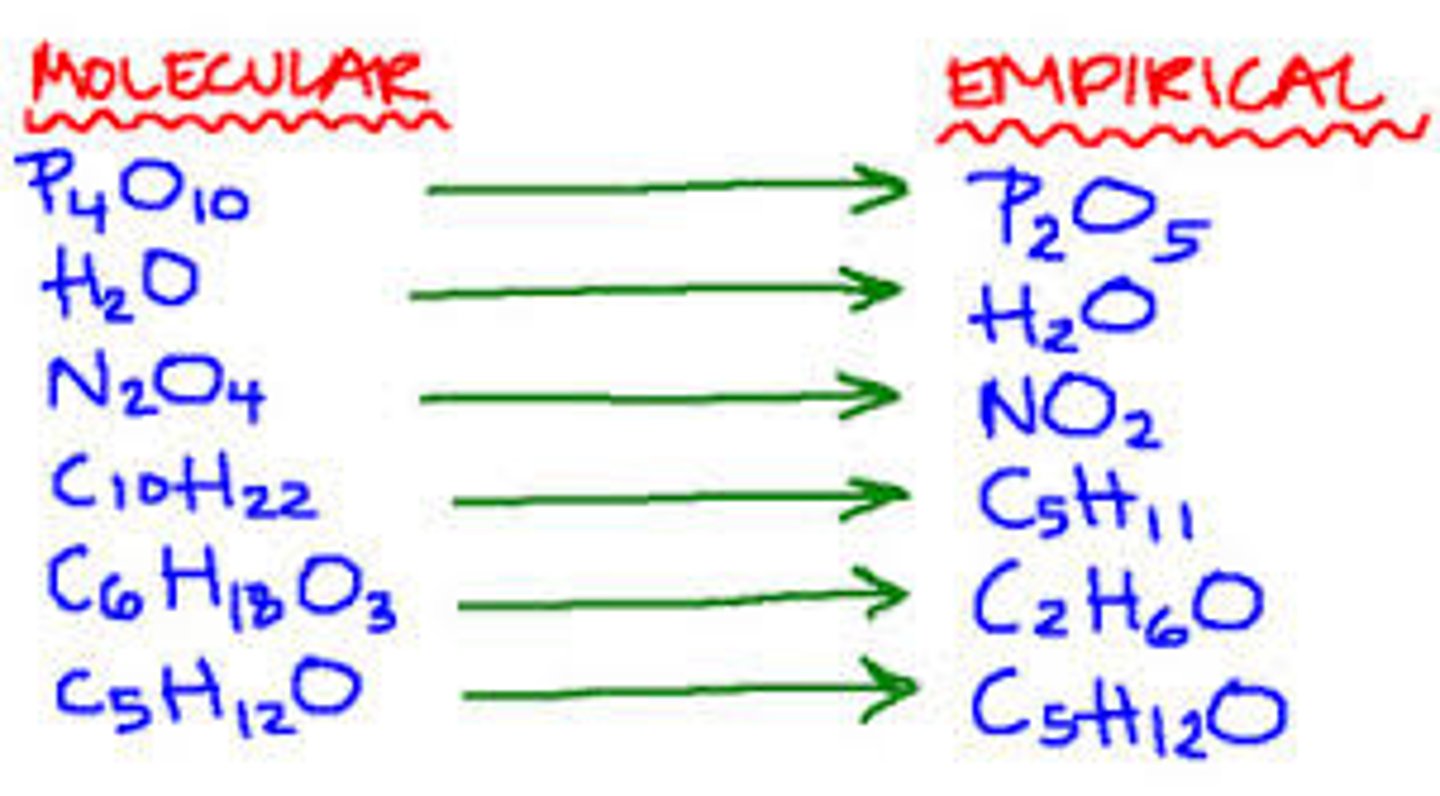
molecular formula
A chemical formula that shows the exact number and kinds of atoms in a molecule, but not the arrangement of the atoms.
percent composition by mass (for an element in a molecule)
molar mass of element/molar mass of molecule x 100%
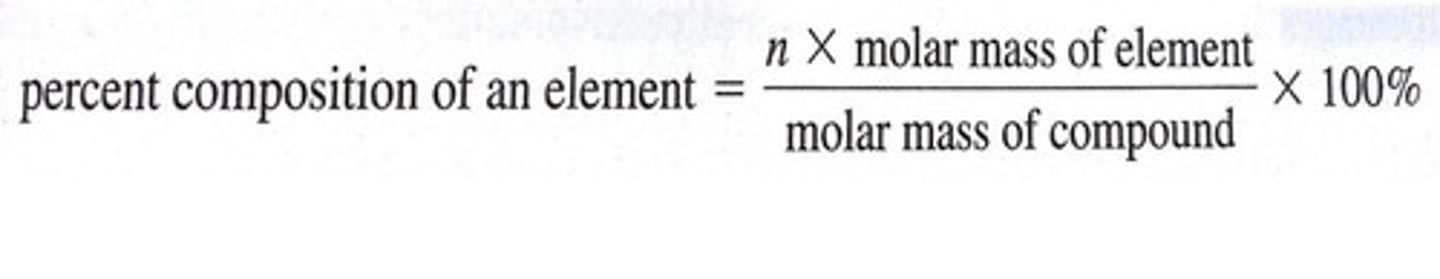
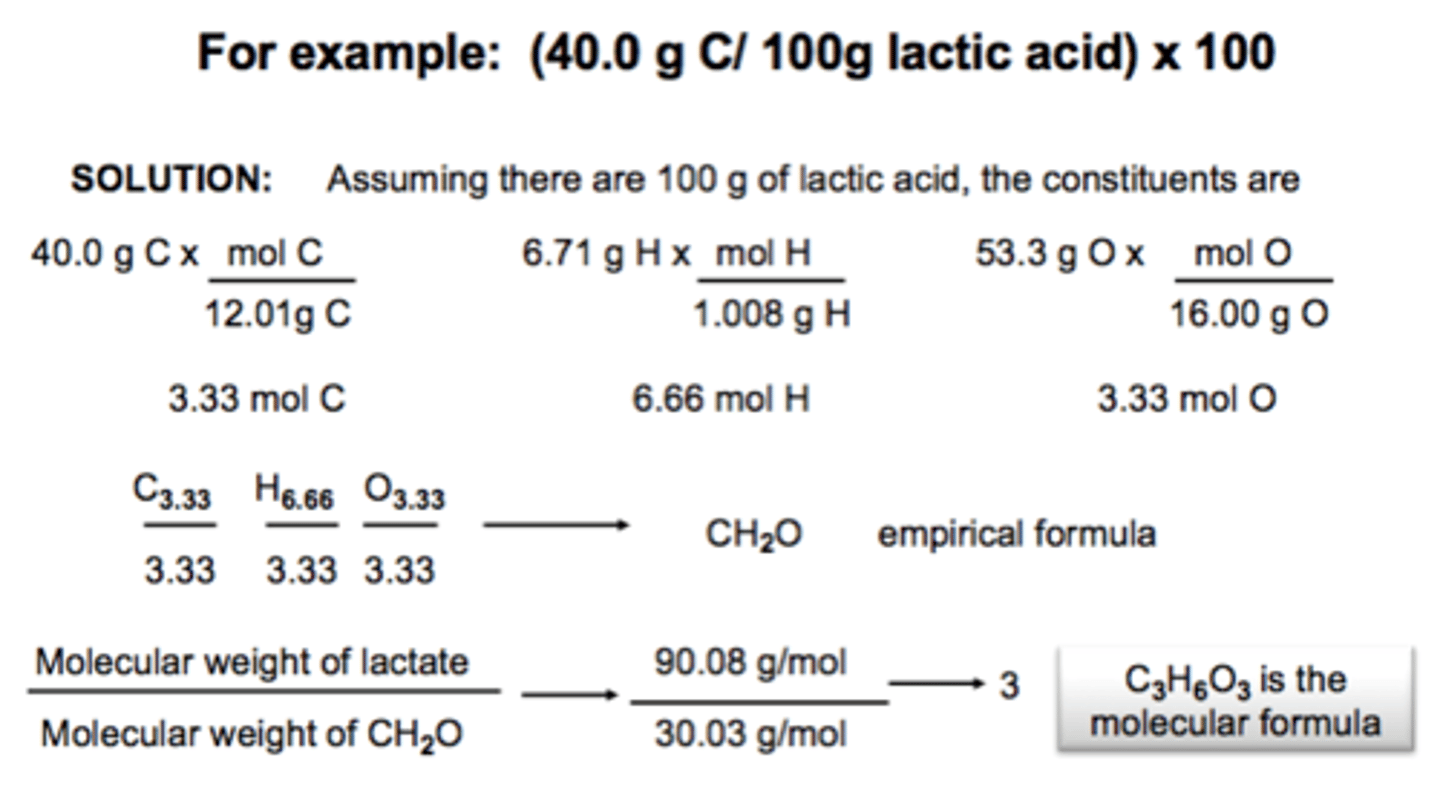
Finding Molecular Formula given molar mass and percent of each element
40.9% carbon, molar mass of compound 264 g/mol
moles C=(0.409)*(264 g/mol)/(12 g/mol)
Alternative method shown in image.
combination reaction
a chemical change in which two or more substances react to form a single new substance
decomposition reaction
a reaction in which a single compound breaks down to form two or more simpler substances
Combustion
reaction involves fuel and oxidant, usually hyrdocarbon and oxygen. Fuel can also be sugar or sulfur. Form carbon dioxide and water.
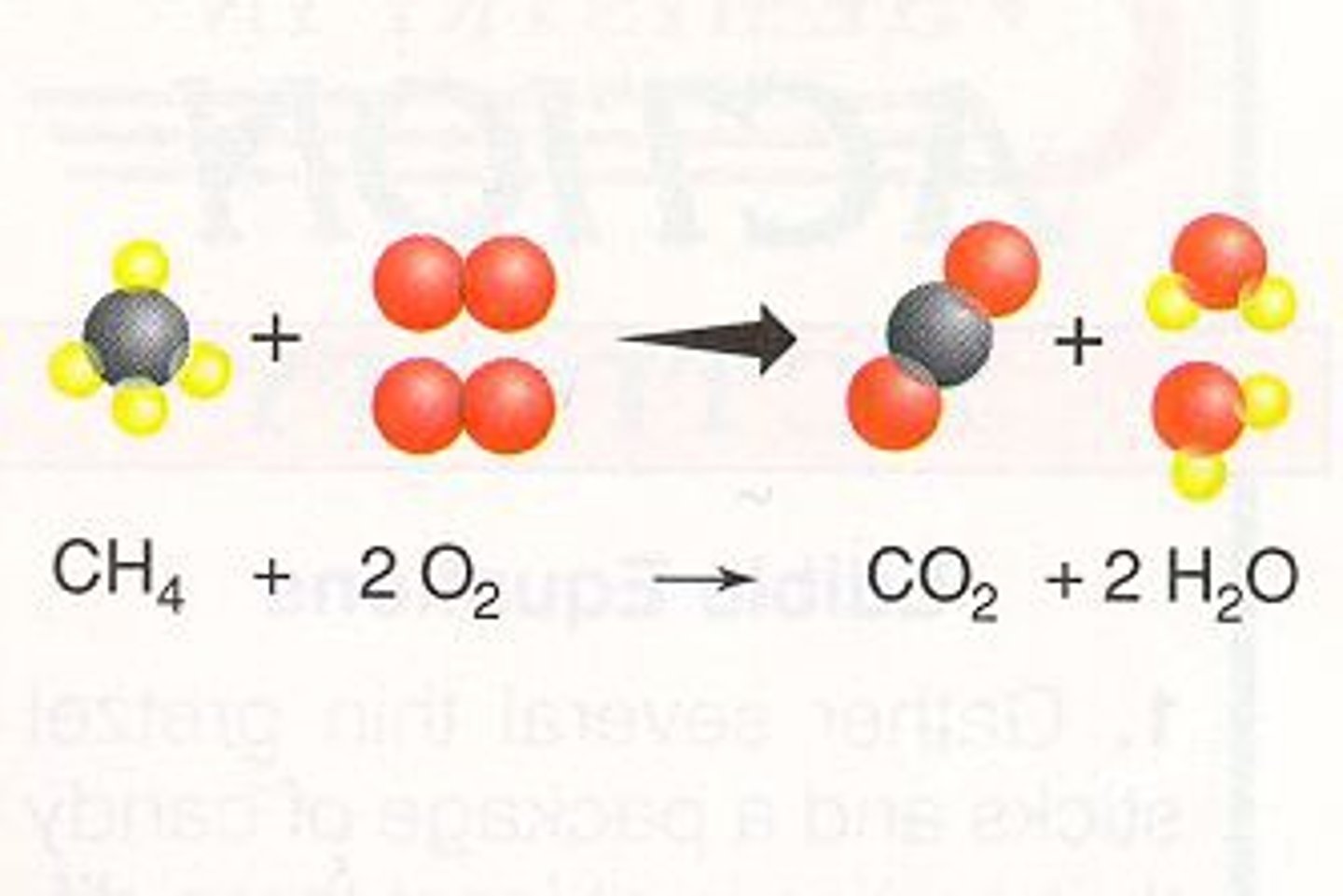
single-displacement reaction
atom or ion in a compound is replaced by another. Often redox.
Ex: Cu+AgNO3-> Ag +CuNO3
double replacement reaction (metathesis)
elements from two different compounds swap places
Ex: CaCl2 + 2 AgNO3-> Ca(NO3)2+2AgCl
neutralization reaction
special type of displacement where acid and base react to form a salt.
laws of conservation of mass and charge
mass/ charges of the reactants consumed must equal the mass/charge of the products produced
stoichiometric coefficients
indicate # of moles of a given species involved in rxns
whole numbers
limiting reactant
the substance that controls the quantity of product that can form in a chemical reaction
excess reagents
reactants present in quantities greater than necessary to react with the quantity of the limiting reagent
theoretical yield
the maximum amount of product that can be produced from a given amount of reactant based on stoichiometry
actual yield
the amount of product that forms when a reaction is carried out in the laboratory
percent yield
actual yield/theoretical yield x 100
-ous
suffix used to represent ions with the lesser charge
Ex: ferrous (Fe2+) vs ferric (Fe 3+)
-ic
suffix used to represent ions with the greater charge
Ex: ferrous (Fe2+) vs ferric (Fe 3+)
-ide
The suffix added to a negative ion's name
Oxyanions
polyatomic ions that contain oxygen;
"-ite" for oxyanion with less oxygen, "-ate" for oxyanion with more oxygen
an anion that has one fewer oxygen atom than the "-ite" anion is given the prefix "hypo-".
An anion that has one more oxygen atom than the "-ate" anion is given the prefix "per-".

Bi-
used to indicate addition of a single hydrogen to anion
Ex: bicarbonate
HCO3-
hydrogen carbonate (bicarbonate)
HSO4-
hydrogen sulfate (bisulfate)
NH4+
Ammonium
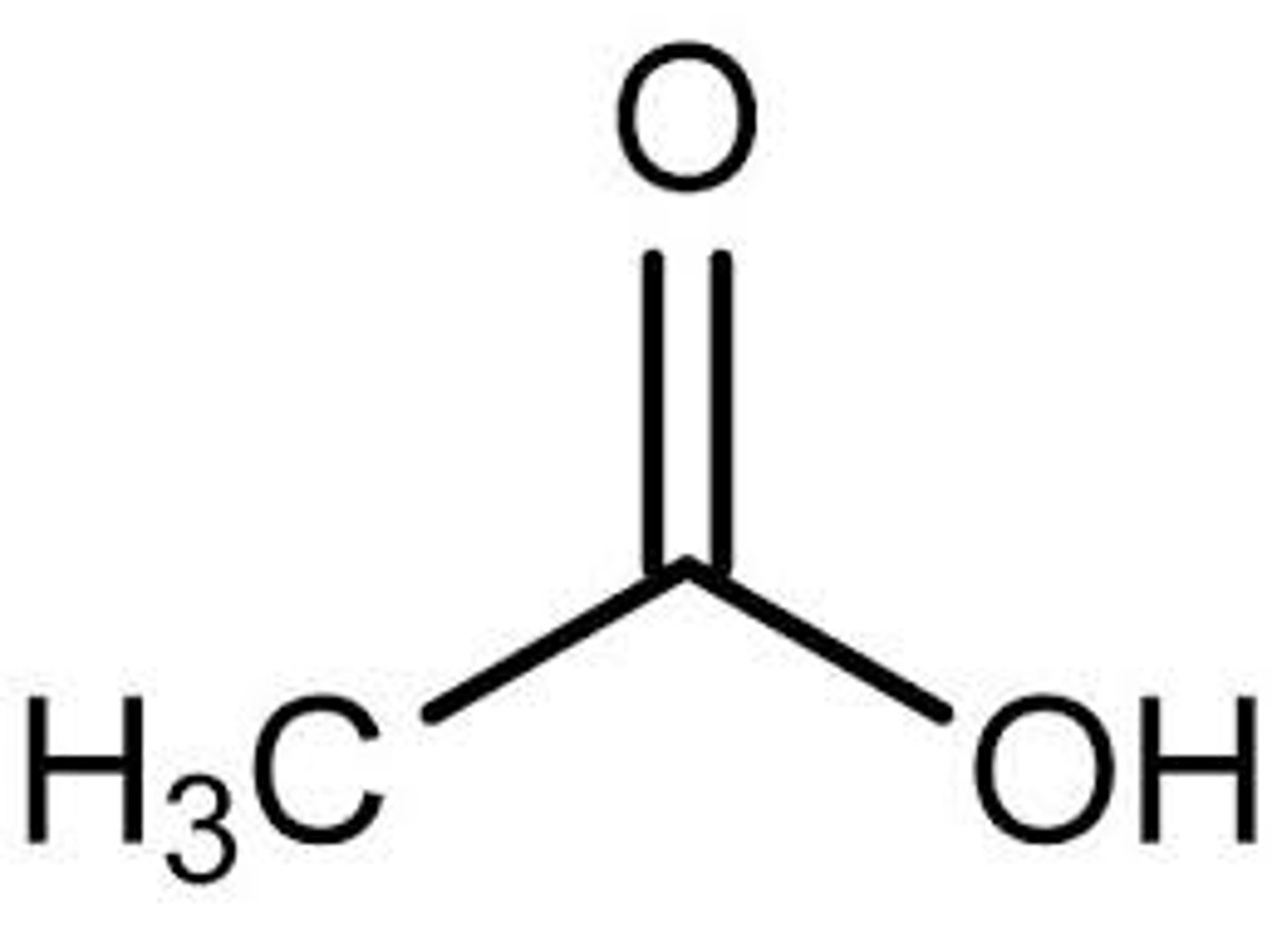
C2H3O2-
Acetate
CN-
Cyanide
MnO4-
Permanganate
SCN-
Thiocyanate
CrO4 2-
Chromate
Cr2O7 2-
Dichromate
BO3 3-
Borate
Electrolytes
-solutes that enable solutions to carry currents
- electrical conductivity is governed by presence & concentrations of ions in solution
strong electrolytes dissociate into ions completely. Weak ones dissociate incompletely, like Hg2I2 and acetic acid. Nonpolar gases are nonelectrolytes
solvation
the act of dissolving