Organic Chemistry - Organic Compounds
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
Alkene
Double carbon covalent bond
Unsaturated hydrocarbons
More reactive than alkanes due to carbon double bond
useful starting materials for making other organic compounds/polymers
can be part of addition reactions due to double bond
Functional Group
group of atoms in a molecule that determines how it reacts
Bromine water test
add orange bromine water to unknow alkane or alkene and shake
if a saturated compound like alkane is present no reactions happen at it remains bright orange
if a unsaturated compound like akene is present a reaction occurs and bromine water is decolourised (colourless)
Alkene DRAW
DRAW
Drawbacks of incomplete combustion
smoky yellow flame
less energy released than from complete combustion
produced pollutants carbon + carbon monoxide + carbon dioxide + water
How do addition reactions work?
two molecules combine to form a single molecule
carbon carbon double bond breaks and opens up (meaning alkenes can be found in them)
new atom is added to each carbon
Hydrogenation
- hydrogen reacts with an alkene in the presence of a catalyst
- which forms an alkane
- this get rid of double bond and adds an hydrogen to EACH carbon atom
Alcholos from alkenes
react with steam in the presence of a catalyst
(water is added across double bond and alcohol forms)
breaks the double bond hydrogen goes on one side OH on the other
then have to separate out the alcohol (OH goes to O - H)
alcohol produced depends on the position of the C=C in the chain
Halogens - Alkenes
chlorine/iodine/bromine
double bond breaks
halogen added to each carbon
di - prefix normally
Polymerisation + conditions
lots of small molecules (monomers)
join together to form a large molecule (polymer)
high temp/pressure
presence of catalyst
How to draw repeating unit of polymer
Draw the bonds of the monomer vertically up and down.
draw to single bonded carbons instead of double bond
fill in rest of the atoms
branch out lines and put brackets in the middle
write n on the outside on the right
Alcohols General Formula +DRAW first four
CnH2n+1OH
functional group - OH
ends in -ol
always show OH when writing general/structural
Properties of alcohols (ONLY FOR THE FIRST FOUR)
Flammable - produce co2 and h2o
souble in water and dissolve to forn neutral solutions with ph7 (longer chain less soluble)
react with sodium to form hydrogen
oxidised by oxidising agent to form carboxylic acids
Uses of alcohol
Alcoholic drinks - ethanol mainly used
Solvents -(methylated spirit - ethanol+chemicals which is posionous to drink so purple dye added)/perfumes
Fuels - ethanol used as a fuel in spirit burners
Ethanol + 4 Uses
C2H5OH
Chemical feedstock to produce other chemicals
Solvent/Fuel? Alcoholic drinks
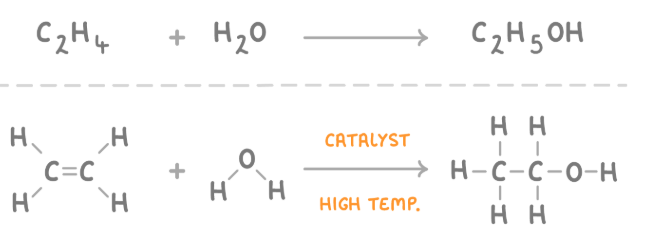
Hydration of ethene
Addition react as water is being added to ethene
high temperature 300'C
6-70 atm
phosphoric acid catalyst H3PO4
ethene+steam—> ethanol
Pros/cons of hydration
100% atom economy as pure ethanol due to one product
Ethene/Reaction is cheap/efficient
however it is non renewable resource made from crude oil
requries catalyst
high cost for process
Fermentation of glucose for ethanol
anaerobic respiration of sugar by yeast cells
carried out in fermentation tanks requires yeast cells (with naturally occuring enzymes) to catalyse the reaction
glucose for yeast to respire
anaerobic conditions
30-40 degrees celsius (optimum temp)
glucose—>ethanol+carbon dioxide
Pros/Cons of fermentation
glucose is a renewable resource and cant run out (yeast is easy to grow)
slow process
not pure ethanol need to undergo fractional distilltion
batch process
Carboxylic acid + DRAW
COOH functional group
end in -anoic acid
double bond with lone oxygen atom and single bond with an oxygen atom in an OH group
show COOH clearly in formulas
Carboxylic acids with water
dissolve in water but dont complete ionise and release H+ ions
form weak acids
higher ph (less acidic) that aqueous solutions of strong acids with the same concentration
Carboxylic acids with carboonates
produce carbon dioxide
a salt
water
NAME BY : adding -anoate to the end eg. Sodium Butanoate
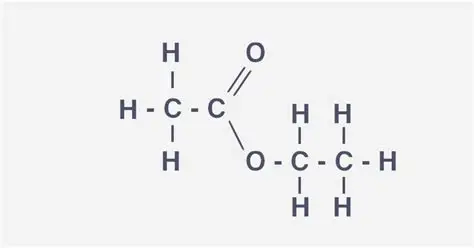
Esters
COO functional group
Can be called an ester linkage as the link the carboxylic acid and alcohol
alcohol + carboxylic aic —> ester + water (in presence of strong acid catalyst) conc H2SO4
used in perfumed because they are volatile/food flavourings
have a sweet and fruity smell
Ethyl ethanoate
Condensation polymerisation
monomers form to create polymer releasing small by products such as water
two different monomers with two different functional groups
one monomer with two functional groups
Polyesters
diol+dicarboxylic acid —> polyester + water
Formation of condensation polymer
dicarboxylic acid (carboxylicc acid with 2COOH groups) gives over its OH
diol (alcohol with two oh groups) gives away its H
this forms one water molecules
the leftover carbon then joints up to the diols oxygen and forms and ester link
repeat this to get 2nH20
Differnces between condensation and additional polymerisation
in addition only one mononer type containing a C=C bond
in condition there are two monomer types each containting two of same function group or one with two different groups
in addition only one product the polymer is formed
in condensation polymer and small molecule is formed
in additional the repeating unit is a hydrocarbon
whereras in condesation has an ester linkage
addtion produced from alkene whereas polyester from a dicarboxylic acid and diol/alcohol
Amino Acids
two function groups in one molecule
Amino Group NH2
Carboxyl group (COOH)
can form polypeptides via condensation polymerisation
Proteins
one or more long chain of polypeptides
fold up to form protein
DNA
deoxyribosenucleic acid
double helix of two polynucleotides
made of four different monomers
Nucleotide
phosphate backbone
deoxyribose sugar
nitrogenous base
adenosine,thymine,guanine,cytosine
Sugars
small moleucles containing carbon,oxygen,hydrogen
can form carbohydrate polymers via polymerisation reactions
polyerms such as starch and cellulose
Incomplete combustion of alkanes
Combustion without sufficient oxygen to react completely
alkene + oxygen → carbon monoxide + carbon dioxide + water
What do functional groups usually determine?
Determine how organic compounds react
eg. Alkene C=C makes it more reactive than alkanes
Harmful products of incomplete combustion
Carbon
Carbon Monoxide
Unburnt hydrocarbons
How to draw addition reactions for alkanes
Always turn double bond to single bond
Hydrogenation - Add tow hydrogens so it forms alkane
Steam - Add OH but position matters
Halogens - Add the diatomic molecule to the alkene
After addition reaction with alkene what must be done
Fractional distillation
To separate water and alcohol
Bromine water example of reaction with halogen
alkene + bromine
shake
causes dibromo -
causing orange colour to disappear (colourless)
this is because C=C reactive so reacts with bromine
Why are alkenes unsaturated compared to alkanes?
Alkene molecules are unsaturated because they contain two fewer hydrogen atoms than the alkane with the same number of carbon atoms.
What does MS 5b mean in AQA GCSE Chemistry?
MS = Mathematical Skill
You must be able to visualise 3D molecules
Draw 2D representations (displayed formulae)
Interpret structural diagrams
Convert between molecular, structural and displayed formulae
Understand molecules are 3D even when drawn in 2D
Monomer
Small repeating units that joint together to form a larger molecule
Know as a Polymer
Polymer
Larger molecule formed from lots of smaller molecules (monomers)
Polymerisation
Process by which lots of small molecules (monomers) join together to form a large molecule (polymer)
Addition Polymers
Formed from unsaturated monomers (double bonds) like alkenes
How do you name polymers
Monomer name in brackets and the phrase “poly” in front of it
Polymer of Ethene would be “poly(ethene)”
How do addition polymers form?
Carbon carbon double bonds within the monomers break
Allowing two monomer molecules to bond to each other
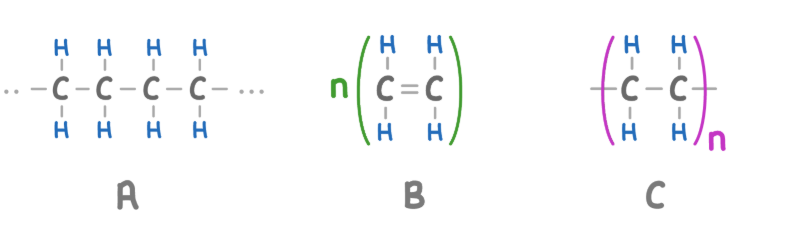
A - Polymer
B - Repeating Unit
C - Monomer
Repeating unit differences
n is on the left
double bond is still present
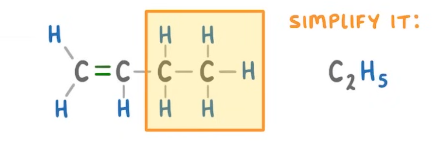
Longer molecules addition polymerisation how to draw repeating unit?
Draw the repeating unti part with the carbon
Take out the carbon double bond
Then for missing part simplify it
Add to the repeating unit (vertically) and connect carbon to carbon not hydrogen
then do the brackets branch out and n on the right
How can alkenes be used to make polymers?
Alkenes can be used to make polymers such as poly(ethene) and poly(propene) by addition polymerisation.
What happens in addition polytmerisaiton?
In addition polymerisation reactions, many small molecules (monomers) join together to form very large molecules (polymers).
Repeating unit vs monomer in addition polymerisation reactions?
In addition polymers the repeating unit has the same atoms as the monomer because no other molecule is formed in the reaction.
Why are alcohols used as solvents?
Dissolve lots of different substances including hydrocarbons oils and fats
Alcohol Table DRAW
Why are temperatures of 30-40 degrees used when making ethanol by fermentation?
catalysed by enzymes and 30-40 degrees is the optimum temperature for them to function at
What is fermentation?
Fermentation is the anaerobic respiration of sugars by yeast cells to produce ethanol and carbon dioxide.
How is ethanol made from ethene?
Ethene is reacted with steam (gaseous water) at a high temperature, high pressure, and with a phosphoric acid catalyst.
How are esters formed?
An ester is made by reacting a carboxylic acid and an alcohol in the presence of an acid catalyst (such as sulfuric acid).
Formation of ester steps
the carboxylic acid loses its OH group,
the alcohol loses the H from its OH group.
These then combine to form water (H2O).
What does biodegradable mean?
Substance that can be broken down by naturally occuring polymers
only condensation polymers are biodegradeable
Requirements for condensation polymerisation
Condensation polymerisation involves monomers with two functional groups
2 different function groups overall
When these types of monomers react they join together, usually losing small molecules such as water, and so the reactions are called condensation reactions.
Three elements that make up carbohyrdates
hydrogen
carbon
oxygen
2 Different functional groups in all amino acids
Amino group (NH2)
Carboxyl Group (COOH)
Different amino acids can be combined in the same chain to produce proteins.
How are polypeptides made
mino acids react by condensation polymerisation to produce polypeptides.
one or more long chains of polypeptides are know as proteins
Sugar Made of?
Carbon
Oxygen
Hydrogen
Sugars can form carbohydrate polymers known as polysaccharides via polymerisation reactions
Examples of Naturally Occurring Polymers
Proteins
DNA
Poly Peptides
Imporatnace and function of DNA
DNA (deoxyribonucleic acid) is a large molecule essential for life
DNA encodes genetic instructions for the development and functioning of living organisms and viruses.
Most DNA molecules are two polymer chains, made from four different monomers called nucleotides, in the form of a double helix.
What are the four naturally occurring polymers essential for life?
DNA - Nucleotides
Protein - Amino Acids
Starch - Glucose
Carbohydrates - Simple sugars (Glucose)
How does amino acids form poly peptides
Two function groups allow adjacent amino acids to join together through condensation reactions
OH and H taken out to form water
Carbon and nitrogen bonds together (amide link/bond/peptide bond)
n before polymer n around repeating unit n before water